This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the benefits of using HILIC as an ideal separation mode for glycans as well as the robustness of HILIC in an SPE format.
Glycans are highly polar biomolecules, making them amenable to Hydrophilic Interaction Chromatography (HILIC) based Solid Phase Extraction (SPE).
Reversed-phase (RP) chromatography, though universally accepted for a majority of compounds, is not particularly well suited for analytes that are hydrophilic in nature. When subjected to RP chromatography, polar compounds are often poorly retained or separated with non-optimal selectivity. Alternatively, Hydrophilic Interaction Chromatography (HILIC) can be used to successfully improve retention of very polar species, such as the glycans encountered during the characterization of protein therapeutics. There are over 40 highly cited published papers using HILIC chemistry for the separation of glycans (search on SciFinder June 2014). One reason for this is that this mode of separation is ideal for these types of compounds due to their highly polar nature. Polar analytes can be strongly and selectively retained onto a HILIC stationary phase when a low polarity mobile phase is used. The concept can be simplified by describing it as like-attracts- like.2 Specifically, the retention of glycans onto a HILIC stationary phase can be explained in terms of the hydrogen bonding as well as ionic and dipole-dipole interactions that occur while the glycans partition into an immobilized water layer.
This technique can be exploited as a form of solid phase extraction (SPE) with the goal to clean up and concentrate the analyte of interest, in this case glycans.
Through sequential load wash and elution steps, one can successfully purify glycans while removing many of the less polar “contaminants” found in deglycosylation and post-derivatization mixtures. Overall, HILIC provides many benefits as an attractive mode of separation for glycans.1
In the case of GlycoWorks SPE, a silica-based aminopropyl sorbent was chosen from several tested due to its highly polar nature. This stationary phase readily and selectively retains polar compounds such as glycans. In addition, this sorbent possesses a weakly basic surface allowing the added potential to exploit ion exchange/repulsion properties.
The eluent pH and ionic strength can impact retention on this HILIC phase and cannot be ignored. For instance, as the ionic strength of the eluent is increased, ionic interactions between the stationary phase and the solutes are disrupted, resulting in ion exchange playing a lesser role in retention.1
In addition, as the mobile phase pH is altered, the charge state of the stationary phase surface is impacted (by nature of the aminopropyl ligand and the base particles’ silanol activity). At higher pH values the ionization of basic analytes and the aminopropyl ligand is reduced, however the ionization of the surface silanols is increased. Because of this, it is critical to choose an eluent that will have a fixed, stable pH every time an assay is performed. Method development taking into account all of these factors is crucial for successful assay reproducibility.1
The first step to HILIC SPE is conditioning with aqeuous mobile phase, which establishes a layer of polar solvent on the stationary phase surface. It is into this aqueous layer that the glycans will partition when they are loaded under low polarity solvent conditions. Subsequently, the adsorbed glycans are washed with solvent to ensure less polar compounds are removed from the sample. Thereafter, the glycans are eluted off the stationary phase with a strongly polar solution, in this case a high concentration of water with a buffer in order to minimize the ionic interactions. The use of ammonium salts of formic acid or acetic acid (ammonium formate or ammonium acetate) are preferentially used due to their volatility. Refer to the application note "Optimization of GlycoWorks HILIC SPE for the Quantitative and Robust Recovery of N-Linked Glycans from mAb-Type Samples" and one of its corresponding figures (Figure 2) for a demonstration on how an eluent for GlycoWorks HILIC SPE was optimized for the reproducible elution of 2-AB labeled glycans.3
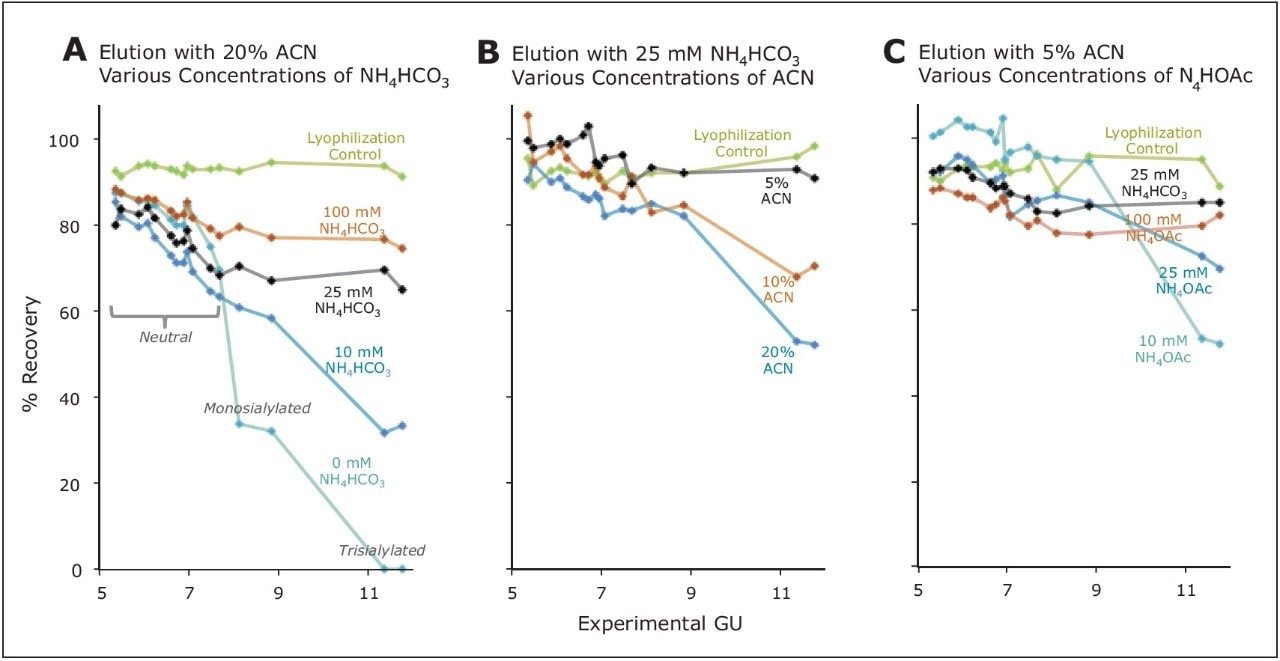
In this work, 2-AB labeled glycans were loaded onto a 96-well HILIC μElution plate according to the protocol provided in the GlycoWorks High-throughput Sample Preparation Kit Care and Use Manual. Various eluents were then employed for elution of the labeled glycans and recoveries for each major species in the test mixture were subsequently determined. These data were compared alongside the recoveries of the glycans from just the lyophilization and reconstitution steps that were performed after the HILIC SPE procedure in preparation of the samples for HILIC-FLR. By means of this development work, a 100 mM NH4OAc, 5% ACN eluent was selected as the optimal elution condition, since it is a pH stable solution and it provided high as well as relatively unbiased analyte recoveries.
Many times, due to the nature of solid phase extraction devices, especially in the case of micro-elution plates, well to well flow rate variation can occur. At times, it can be observed that one well will exhibit a faster flow than another. Such well-to-well flow rate differences can be further amplified by more viscous solvent such as highly aqueous solvents. Other variables that can contribute to flow variability include improper seating of the plate on the vacuum manifold, introduction of air when pipetting into a well and changes in operating pressures when vacuum driven SPE is performed. Optimization of individual vacuum manifolds can be critical to achieving consistent well-to-well flow rates. Nevertheless, it is almost impossible to entirely eliminate well to well flow rate differences so the question was asked, what impact does this have on the resulting reproducibility of oligosaccharide recovery? If the method is robust, this mechanical variability should be inconsequential.
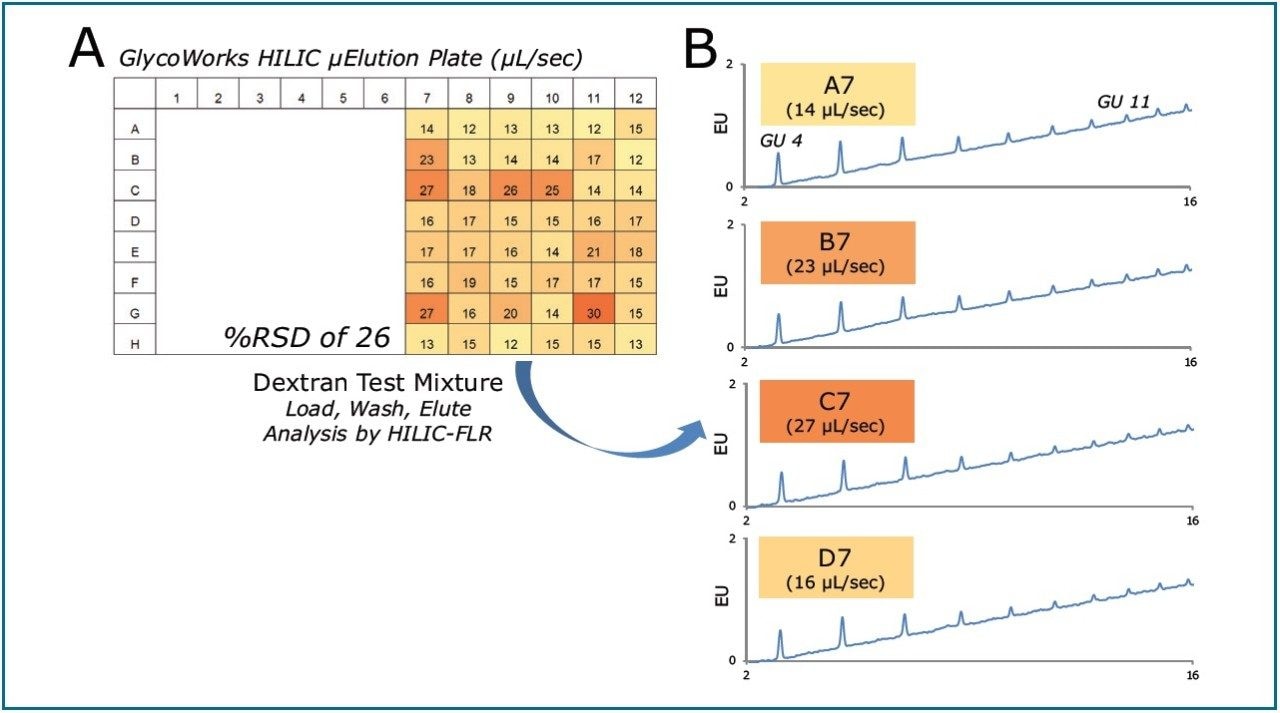
To this end, the GlycoWorks Care and Use protocol was employed along with the Dextran Calibration Ladder (PN 186006841) to test the impact of variable flow rates on sample recovery (GlycoWorks High-throughput Sample Preparation Kit Care and Use Manual).4 As an initial step, 500 μL of water was pulled through wells on a GlycoWorks HILIC SPE uElution plate. A vacuum setting of 5 inches of Hg was used to drive flow through one well at a time.
Meanwhile, the time to draw the 500 μL through each well was recorded, enabling a flow rate metric for each well to be determined. Sample was thereafter processed, without the use of any special practices. Figure 3A shows the measured flow rates. The color gradient denotes faster flow rates with darker colors and slower flow rates with lighter colors.
Figure 3B illustrates the fluorescence chromatograms obtained for oligosaccharide samples processed through wells with varying flow rate characteristics.
Overall, the GlycoWorks HILIC SPE plate exhibited less than a 3-fold variation in flow rate across a set of 48 wells when operated with a vacuum manifold (Figure 3A).
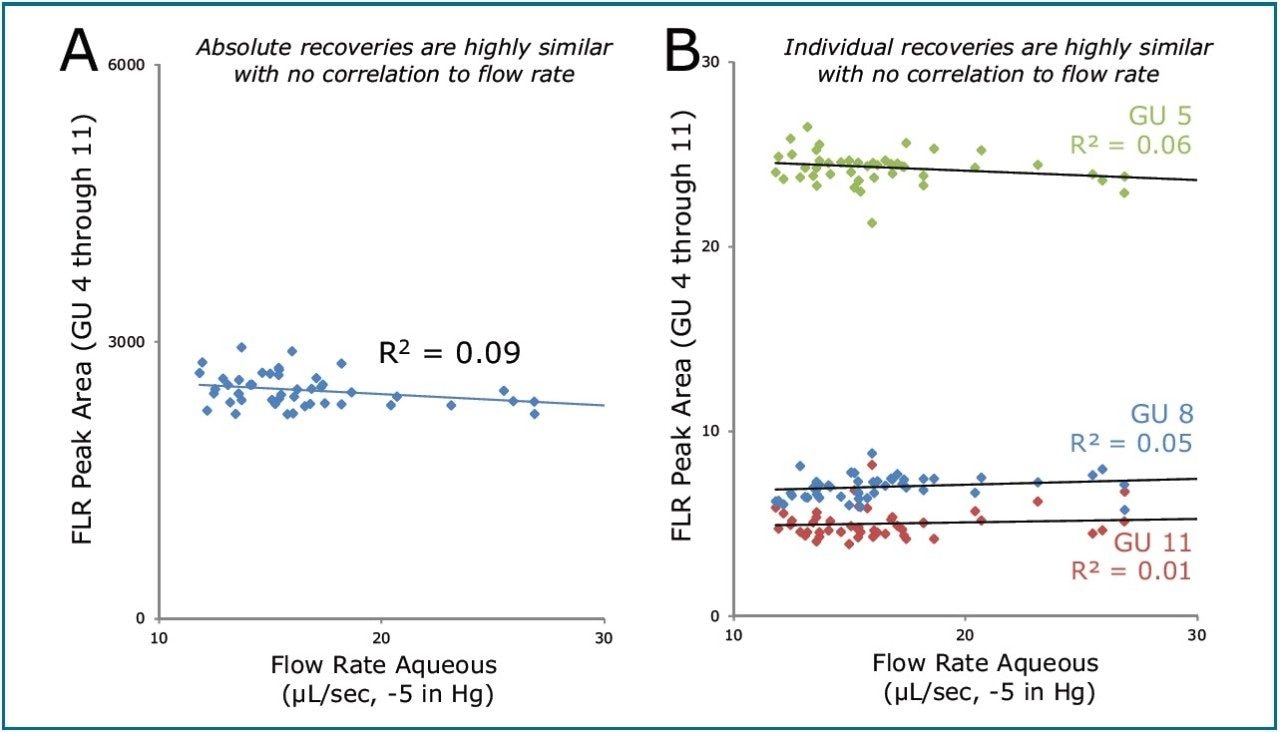
When these data were viewed in the context of oligosaccharide recovery, however, it became clear that any differences that do exist in flow rate characteristics from well-to-well do not negatively impact recovery, whether absolute or relative, as demonstrated in Figure 4.
GlycoWorks HILIC SPE offers a robust, reliable solution for cleanup and concentration of glycans from complex matrices. Its application during the sample preparation of 2-AB labeled glycans helps ensure that an analyst reliably obtains successful results when studying protein glycosylation.
720005116, September 2014