In this application note, we present CCS, derived from ion mobility drift times, as a new identification parameter that can efficiently reduce the number of false detections when used in combination with conventional accurate mass and retention time information.
The SANCO/12571/2013 guidance document implemented in 2014 describes the method validation and analytical quality control guidelines to support the validity of data used for checking compliance with maximum residue limits (MRLs), enforcement actions, or assessment of consumer exposure to pesticides in the EU.1 For high resolution MS systems, the requirements for identification are that the quasi molecular ion and at least one fragment ion are obtained with mass accuracy <5 ppm. In addition isotope ratios and signal-to-noise (S/N) are considered. It is also stated that “identification relies on proper selection of diagnostic ions”. The ratio of the chromatographic retention time of the analyte to that of a suitable internal standard, i.e. the relative retention time of the analyte, should correspond to that of the calibration solution with a tolerance of ±0.2 minutes for LC. It is also stated that “different types and modes of mass spectrometric detectors provide different degrees of selectivity, which relates to the confidence in identification”. Requirements for identification such as maximum tolerances for ion ratios are provided. These requirements should be regarded as guidance criteria for identification, not as absolute criteria, to prove the presence or absence of a compound.
The current guidelines have been modified to make retention time tolerances more stringent and could result in system-specific assay parameters being generated. Retention time tolerances can be impacted by matrix shifts, system setup, and column conditions. Applying a “catch all” approach would provide a generic set of processing parameters that would allow qualitative screening methods to be developed which are applicable across a wide variety of matrices, and provide a more efficient transfer between instruments and laboratories. The application of additional filtering such as isotope pattern match and fragment ions enables less stringent screening parameters and provides increased specificity simultaneously. This approach becomes less system specific.
Pesticide residue analyses in food has become a more difficult task considering the increasing number of compounds that need to be monitored at lower concentrations using generic extraction procedures. Qualitative multi-residue methods focus only on detection (demonstration of neither recovery nor linearity is required), and can therefore alleviate the quantification process.
Full-scan high resolution MS offers a higher specificity with theoretically no limitation on the number of compounds detected, but it is still challenging to rapidly and efficiently identify targeted compounds present in a sample with a large number of co-extracted matrix components.
The key to successful implementation is the ability to efficiently identify targeted compounds present in a sample with an acceptable level of false negative results (≤5%) as outlined in the EU guidelines Although there is no requirement in the guidelines regarding the number of false detects, it is desirable to keep their number as low as possible to minimize the time required for additional investigation and hence to reduce the overall cost. The challenge is to remove false detections through careful optimization of the software screening parameters, but also to deal with very complex matrices that can produce false negative identifications.
Collision cross section (CCS) is a robust and precise physicochemical property of an ion. CCS is an important distinguishing characteristic of an ion that is related to its chemical structure and three-dimensional conformation, illustrated in Figure 1, where the shadow of the rotating three-dimensional ion represents the average collision cross section. The use of CCS data offers the potential to reduce the initial specificity requirements of applied screening parameters. Previously generated CCS data have been entered into a scientific library within the Waters UNIFI Scientific Information System. Expected and previously determined CCS values can be utilized to reduce false identifications in proficiency test samples and matrix matched calibrant series analyzed, while applying wider screening tolerance parameters. In this application note, we present CCS, derived from ion mobility drift times, as a new identification parameter that can efficiently reduce the number of false detections when used in combination with conventional accurate mass and retention time information.
|
UPLC system: |
ACQUITY UPLC I-Class |
|
Column: |
ACQUITY UPLC BEH C18 100 mm x 2.1 mm, 1.7 μm |
|
Column temp.: |
45 °C |
|
Flow rate: |
0.45 mL/min |
|
Mobile phase A: |
Water (0.1% formic acid) |
|
Mobile phase B: |
Acetonitrile (0.1% formic acid) |
|
Injection volume: |
5 μL |
|
Time (min) |
%A |
%B |
|---|---|---|
|
0.00 |
98.0 |
2.0 |
|
0.25 |
98.0 |
2.0 |
|
12.25 |
1.0 |
99.0 |
|
13.00 |
1.0 |
99.0 |
|
13.01 |
98.0 |
2.0 |
|
13.00 |
98.0 |
2.0 |
|
17.00 |
98.0 |
2.0 |
|
MS system: |
SYNAPT G2-S |
|
Ionization mode: |
ESI+ |
|
Desolvation temp.: |
550 °C |
|
Mass range: |
50 to 1200 Da |
|
Acquisition rate: |
5 spectra/sec |
|
Capillary voltage: |
1 kV |
|
Cone voltage: |
20 V |
|
Drift gas: |
N2 |
|
Collision energy ramp: |
10 to 45 eV |
|
IMS wave velocity range: |
650 m/s |
|
IMS wave height: |
40 V |
|
IMS gas flow: |
90 mL/min |
|
IMS duty cycle: |
10.8 ms |
|
Lock mass: |
m/z 556.2766 (Leucine enkephalin) |
10 g of homogenized sample was extracted with 60 mL of 20-mM ammonium acetate in methanol using an Ultra-Turrax device. the crude extract was then filtered and diluted up to 100 mL with 5-mM ammonium acetate in water before injection.
An organic mandarin sample was used to produce a matrix-matched curve and a previous European ring-test FV-13 sample was analyzed (European Commission proficiency tests for pesticide residues in fruits and vegetables. FV-13 Mandarin Homogenate, 2011).
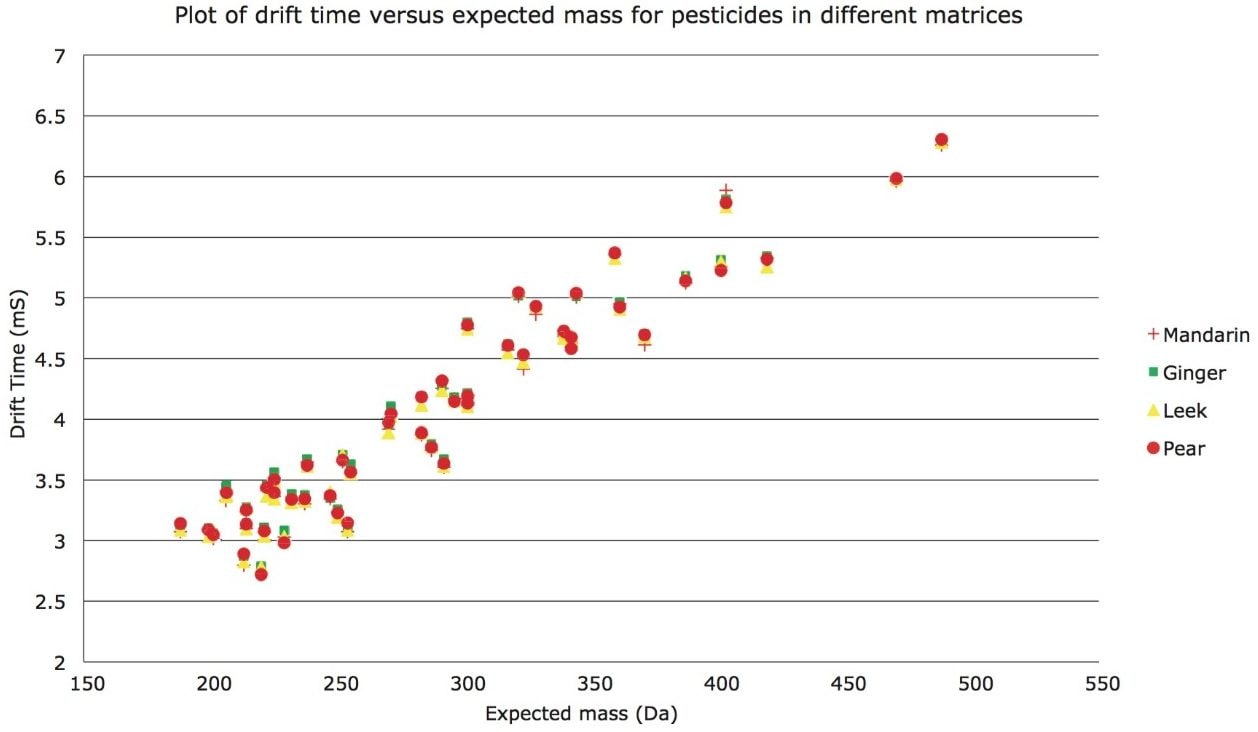
Pear, ginger, leek, and mandarin matrices were spiked with a mixture of pesticide standards and ion mobility drift time values of the pesticides in the matrix were determined. Ion mobility drift times obtained for each respective matrix were plotted against their respective m/z. From Figure 2, it can be seen that the ion mobility of the pesticide standards is independent of the matrix components and that the drift times of the pesticides in each matrix overlay each other. Therefore, ion mobility drift times can be utilized as a confirmatory parameter to increase confidence in identification and help to further reduce false detections.
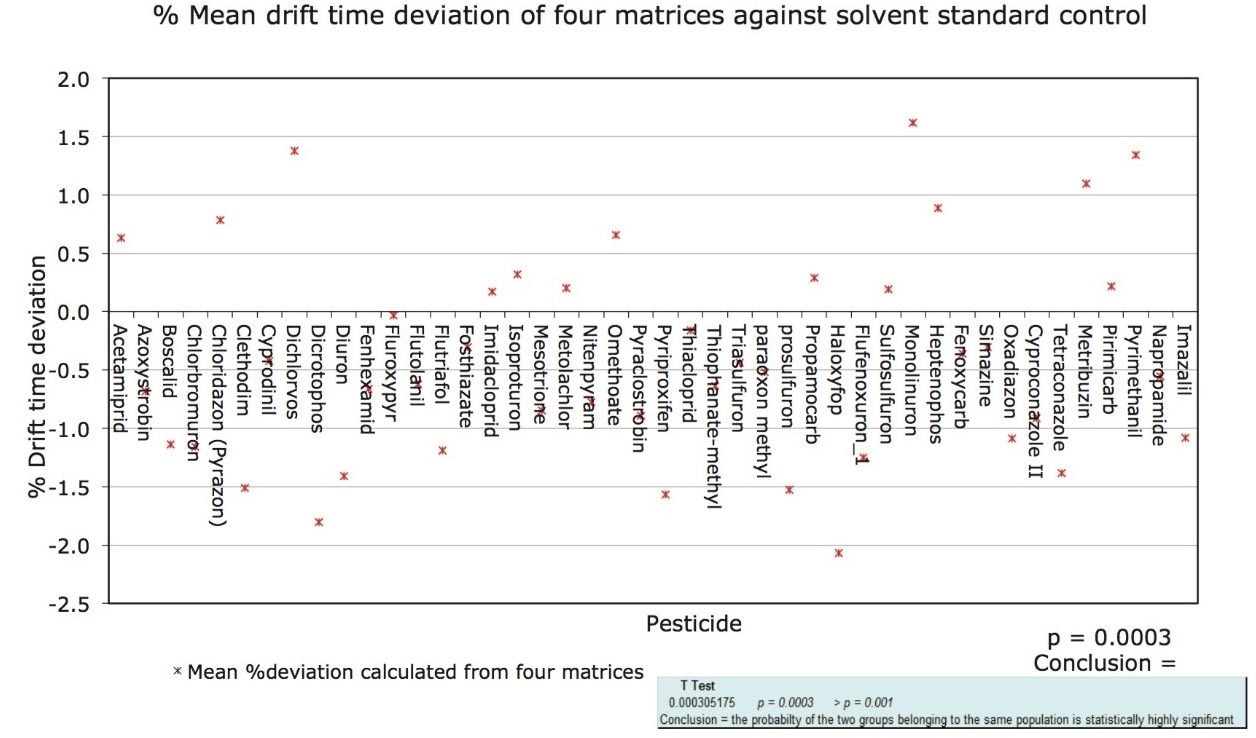
Ion mobility drift times, from the pesticide solvent standards and the analyzed pesticide matrix matched samples were shown to statistically belong to the same population, as shown in Figure 3. A one-tailed t test was performed and a probability that the two groups belonged to the same population was highly significant. Based upon these initial studies software development was undertaken, which enables ion mobility drift times and hence collision cross sections to become part of a routine screening workflow.
For the first time, estimated CCS values were generated and employed in a conventional workflow within the UNIFI Scientific Information System, which has been specifically designed for non-targeted accurate mass screening applications. The feasibility of using UPLC separations and ion mobility MS with a generic precursor ion and fragment ion acquisition has been explored.
Data were initially acquired using MassLynx Software for a series of solvent standard mixtures. Subsequently the corresponding set of data was acquired for the pear, ginger, leek, and mandarin matched matrix calibration series and then for the EU-RL proficiency test samples. These were utilized to generate mobility separated single component precursor ion and fragmentation spectra for the [M+H]+ or adducted species. The MassLynx data was processed using the software within UNIFI. Estimated CCS values, precursor ion, fragment ions, and retention time values were determined for the pesticide standards. This information was entered into the UNIFI scientific library. It should be noted that the software did not incorporate the LockCCS functionality at the time these initial feasibility studies were performed.
The challenge of avoiding false detections is clearly illustrated in Figure 4, where the impact of the matrix upon the expected retention time is shown for boscalid – the expected retention time obtained using solvent standards was 7.33 minutes. For leek and ginger, retention time errors of 0.33 min and 0.43 min were obtained. These are outside of the SANCO/12571/2013 tolerance guidelines. If a tolerance window of +/- 0.2 min was applied in the screening parameters set, two false negative detections would have resulted. These results simply illustrate the realistic challenge of residue screening assays. Retention times are not only impacted upon by the matrix, but variations can occur from laboratory to laboratory where factors such as chromatographic system setup, organic solvent, water, and human error can impact results.

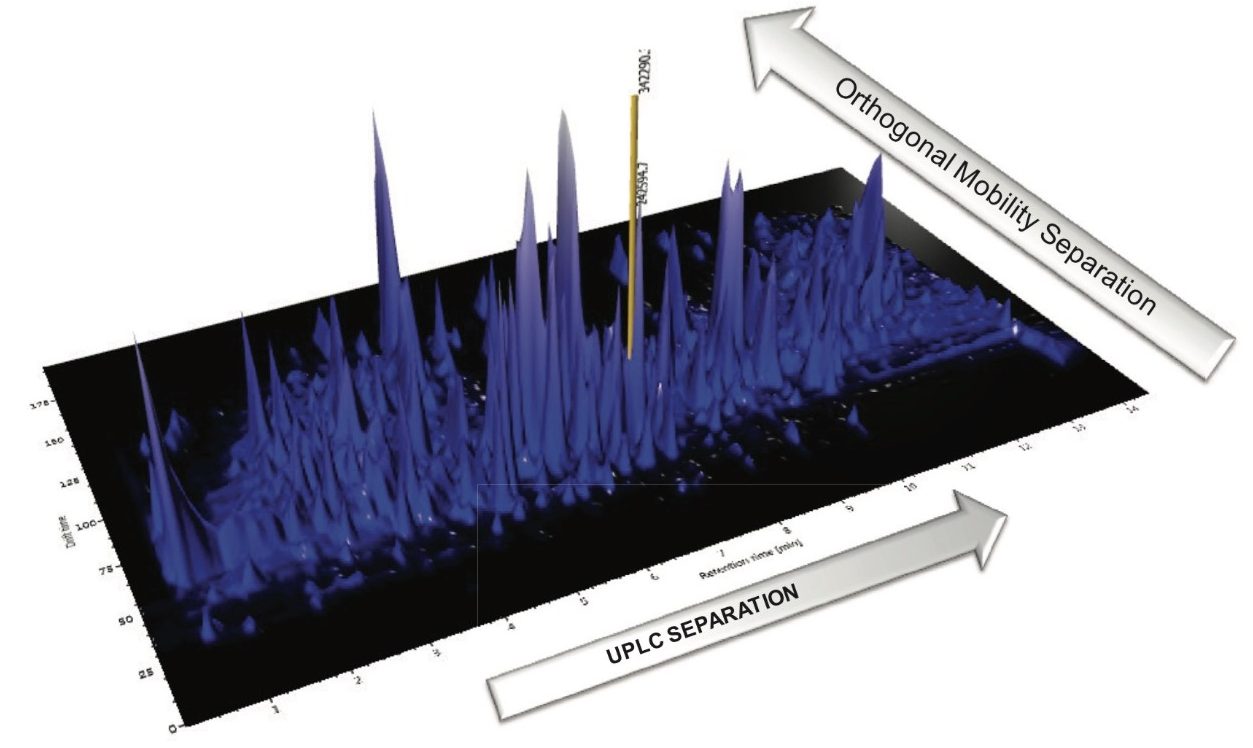
The complexity of the mandarin matrix is demonstrated in Figure 5, where the two-dimensional separation achieved using the ACQUITY UPLC I-Class System and ion mobility MS is presented, and the position of boscalid is indicated by the yellow marker.
During this study, five laboratories were given a generic protocol and asked to analyze the solvent standards and matrix-matched samples as previously described. The samples were prepared and aliquots were provided to the five laboratories taking part in order to determine inter-laboratory reproducibility. In Figure 6 shows the spread of retention times that were obtained for 40 pesticides on five independent UPLC-IMS systems. All data were acquired using the same generic UPLC and mass spectrometry system parameters; no other system parameters were stipulated.
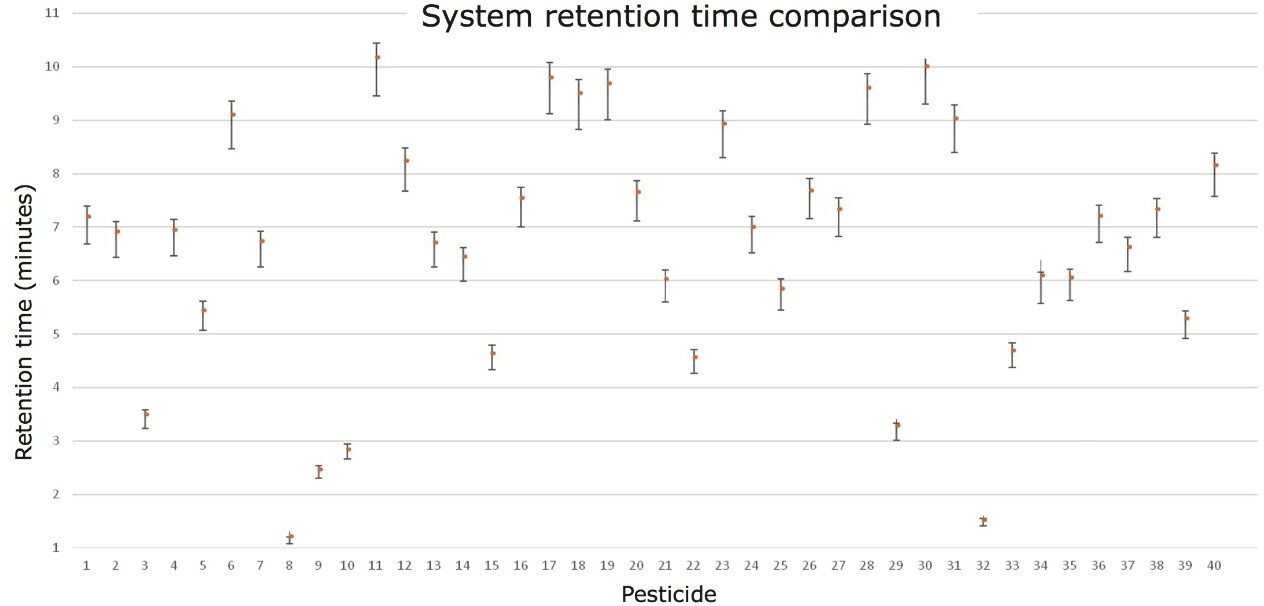
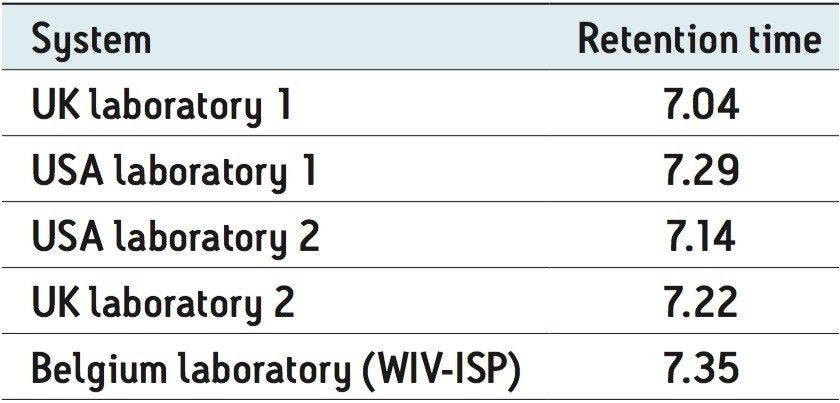
Although inter-laboratory reproducibility challenges can be overcome, the spread of retention times observed illustrates the challenge of using generic methods in inter-laboratory studies. The retention time for boscalid achieved from five different laboratories is presented in Table 1. This data demonstrates the variations that can occur due to inter/intra laboratory hardware setup and the commodities being analyzed. This further justified our goal of creating a generic “catch all” screening approach that is capable of overcoming the practical idiosyncrasies of residue analysis.
To avoid high false detect rates from an automatic screening system, different parameters and related criteria that can impact peak designation should be carefully tuned and finalized. The same protocol was applied to a previous proficiency test sample, FV-13. The first step of tolerance criteria was applied and 29 pesticide residues matching the criteria were observed. The resulting data was subsequently filtered using 10 ppm mass accuracy on the precursor ions, thereby removing seven potential false detects and reducing the list down to 22. Further filtering with fragmentation criteria resulted in ten matches in total. Ultimately, applying a CCS tolerance of 2% reduced the number of compounds to eight, removing two more false detect identifications.
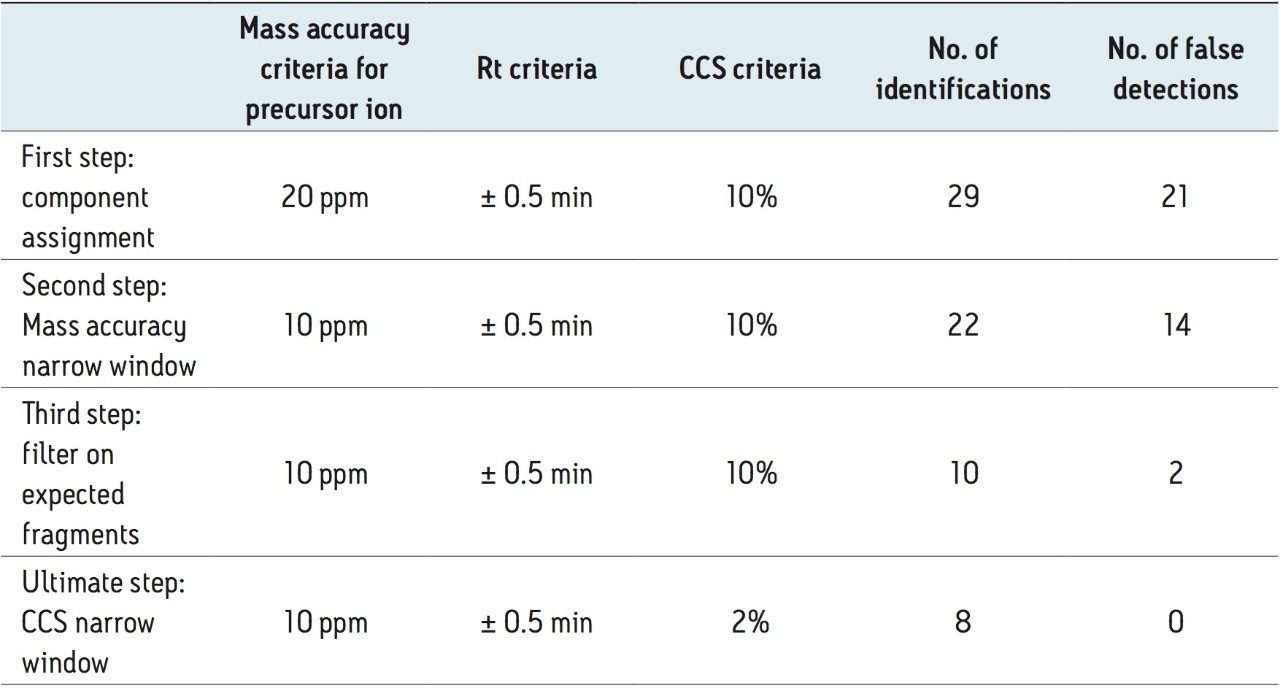
See Table 2 for a complete summary. In this study, under the conditions used, eight compounds were detected and identified. Importantly it should be noted that 100% of the detected compounds were identified correctly. Also this approach used a standard CCS screening workflow to interrogate the data. After the initial data processing has been performed and the component summary reviewed, by simply selecting the workflow step allows the impact upon the results obtained to be reviewed. Once the data have been processed, workflow steps, including tolerances and filters, can be employed and incorporated into a workflow design. There is no requirement to reprocess the data.
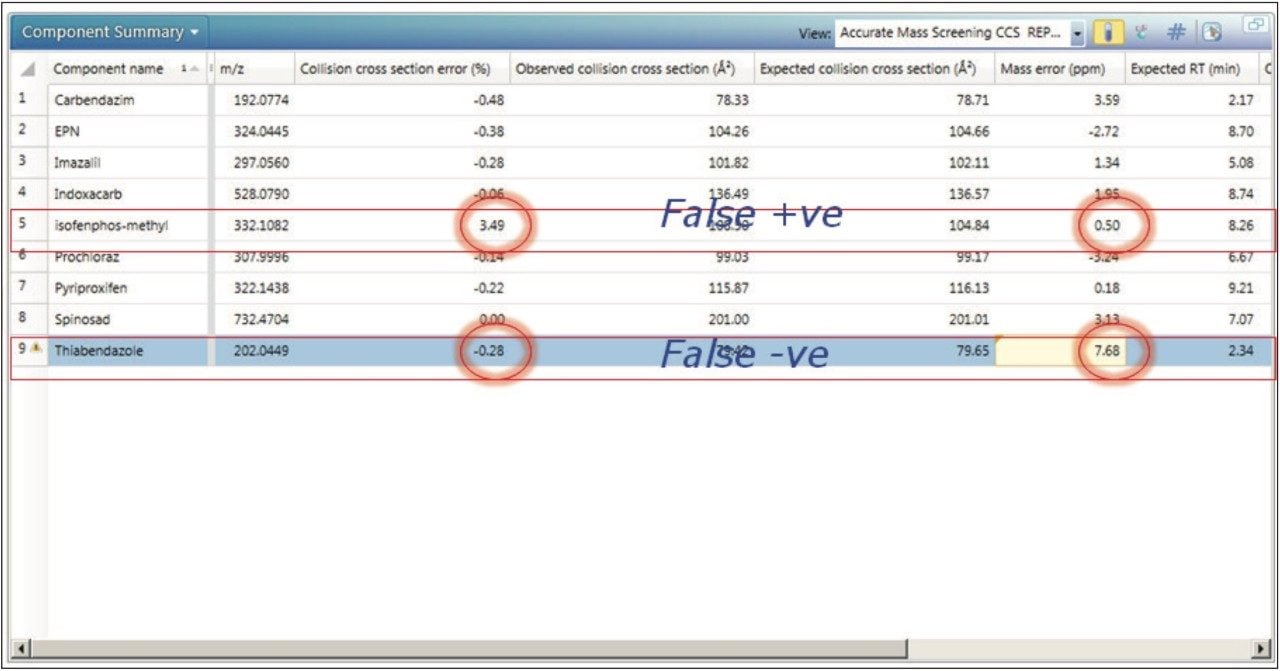
Figure 7 shows an example component summary where the avoidance of false positive and negative detections is illustrated in the results from one of the laboratories. For this particular compound/laboratory combination, if the mass tolerance parameters had been restricted to 5 ppm, thiabendazole (mass accuracy 7.68 ppm) would have been discarded and a false negative would have resulted. The CCS error for thiabendazole, however, was just -0.28%. An analyst would likely postulate that isofenphos-methyl was correctly identified because a mass accuracy of 0.5 ppm was obtained. The CCS error measured for isofenphos-methyl was, however, 3.49%. With the use of IMS-MS and the application of a 2% CCS tolerance, a false positive and a false negative was avoided. The time-consuming process of ruling out a false positive can be avoided and more importantly, the consequences of a false negative can be avoided as well.
The application of less stringent parameters in terms of mass accuracy and retention time tolerance in combination with CCS measurements can provide greater confidence when dealing with the analytical challenges of residue screening. This approach has the potential to create inter/intra generic processing methods and also to overcome the requirement of repeatedly performing the expensive process of retention confirmation using analytical standards.
720005055, June 2014