For research use only. Not for use in diagnostic procedures.
This Application note demonstrates the extraction and analysis of plasma catecholamines and metanephrines using Oasis WCX μElution Plates and an ACQUITY UPLC BEH Amide Column in HILIC mode.
Clinical researchers are often interested in measuring elevated concentrations of plasma catecholamines and their O-methylated metabolites (metanephrines). However, these compounds (in particular, norepinephrine, epinephrine, and dopamine) can be a challenge to analyze via reversed-phase LC-MS/MS due to their polarity. As a result, many research laboratories still analyze this panel using ion-pairing reagents and electrochemical detection (ECD). While reversed-phase LC-MS/MS has been used successfully, challenges still exist due to ion-suppression from matrix components, insufficient retention, and inadequate separation of normetanephrine and epinephrine.
Hydrophilic interaction chromatography (HILIC) is increasingly becoming a method of choice for the analysis of polar compounds.1-6 Expanding upon earlier published methods,6-7 this application note describes the extraction and analysis of monoamine neurotransmitters and metanephrines from plasma. HILIC-based chromatographic separation is achieved using a Waters ACQUITY UPLC BEH Amide Column. Waters Oasis WCX 96-well µElution Plates are used to extract these compounds from plasma. The use of mixed-mode weak cation exchange solid-phase extraction (SPE) plates, in combination with the amide column for HILIC chromatography and the Waters Xevo TQ-S mass spectrometer, result in a rapid, robust method with excellent linearity, accuracy and precision, as well as minimal matrix effects.
|
LC system: |
ACQUITY UPLC |
|
Column: |
ACQUITY UPLC BEH Amide, 1.7 μm, 2.1 x 100 mm |
|
Column temp.: |
30 °C |
|
Sample temp.: |
10 °C |
|
Mobile phase A (MPA): |
95:5 Water:ACN containing 30 mM NH4HCOO, pH 3.0 |
|
Mobile phase B (MPB): |
15:85 Water:CAN containing 30 mM NH4HCOO, pH 3.0 |
|
Needle washes: |
Strong and weak needle washes were both placed in MPB |
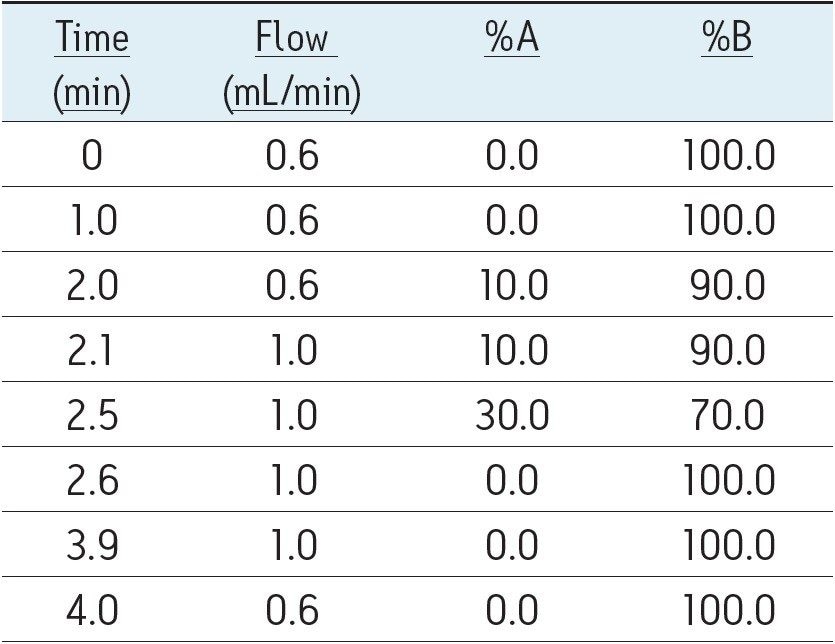
The gradient ramp is shown in Table 1 and includes an initial hold, followed by a shallow ramp and an increase in flow rate to re-equilibrate the column. The entire cycle time is 4.0 min.
|
MS system: |
Xevo TQ-S |
|
Ionization mode: |
ESI Positive |
|
Capillary voltage: |
0.5 kV |
|
Cone voltage: |
Compound specific (see Table 2) |
|
Desolvation gas: |
900 L/hr |
|
Cone gas: |
150 L/hr |
|
Desolvation temp.: |
550 °C |
|
Source temp.: |
150 °C |
Data were acquired and analyzed using UNIFI Software
Combined stock standards containing 10-µg/mL of dopamine (DA), 3-methoxytyramine, (3-MT) metanephrine (MTN), and normetanephrine (NMT) and 50-µg/mL of norepenephrine (NE) and epinephrine (EP) were prepared in methanol containing 0.1% ascorbic acid to prevent oxidation. A combined internal standard stock solution composed of 10-µg/mL D3-metanephrine, D3-normetanephrine, D4-dopamine, D6-epinephrine, and D6-norepinephrine, was also prepared in methanol containing 0.1% ascorbic acid. Working internal standard solutions were prepared daily in 5% MeOH with 0.1% formic acid at a concentration of 2.5 ng/mL.
Human plasma (sodium heparin) was obtained from Biological Specialty Corporation (Colmar, PA). Pooled plasma (6 lots) was used to prepare calibration and quality control samples.
Pooled plasma samples (250 µL) were pre-treated with 250-µL of 50-mM NH4CH3COO and 50-µL of an internal standard working solution (2.5 ng/mL). Pre-treated samples were loaded in individual wells of an Oasis WCX 96-well µElution Plate that had been conditioned with 200-µL of MeOH and 200-µL of H2O. After loading the samples, wells were washed with 200-µL of 20-mM NH4CH3COO followed by 200-µL of 50:50 ACN:IPA. The 96-well plate was then dried under vacuum for 30 s to remove as much solvent as possible from the sorbent bed. The target compounds were eluted from the plate with 2 x 25-µL aliquots of 85:15 ACN:H2O containing 2% formic acid into an 700-µL 96-well sample collection plate (p/n 186005837). 15-µL of the eluate was injected onto the UPLC-MS/MS System.
The structures of all compounds are shown in Figure 1 along with their individual logP values, demonstrating the highly polar nature of many of these compounds. Table 2 shows the retention times and individualized MS parameters of each compound, including MRM transitions, cone voltage, and collision energy.
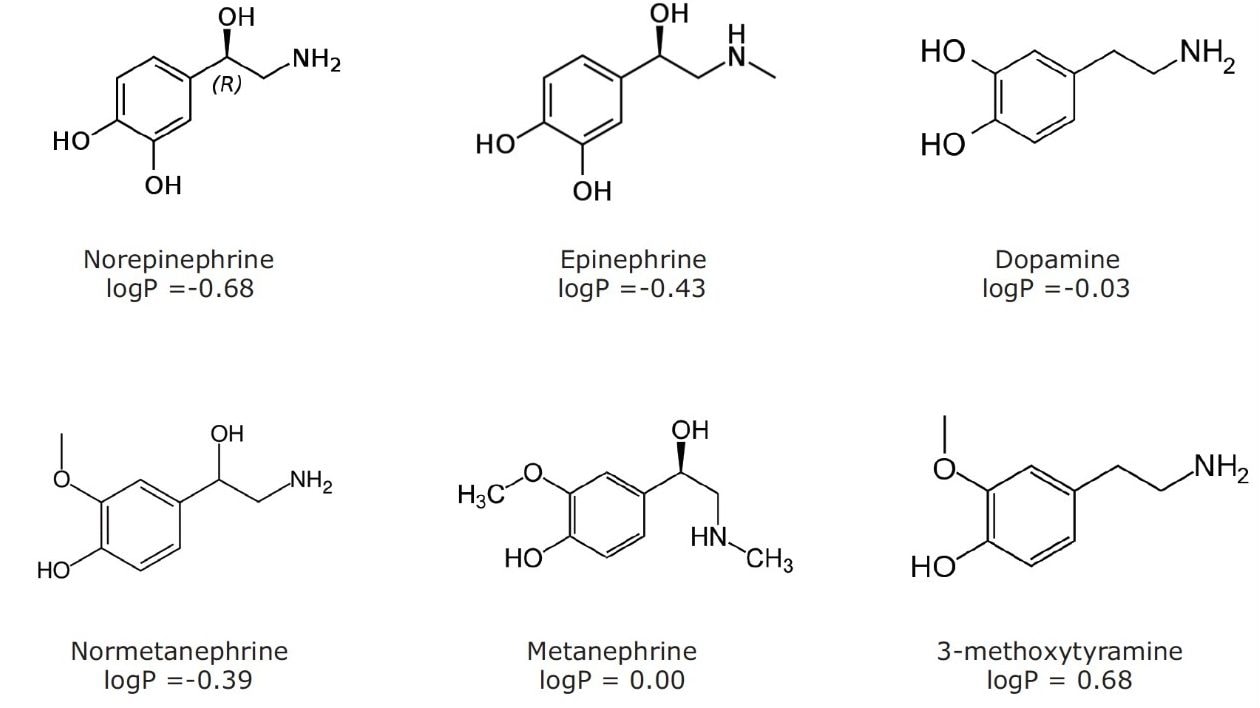
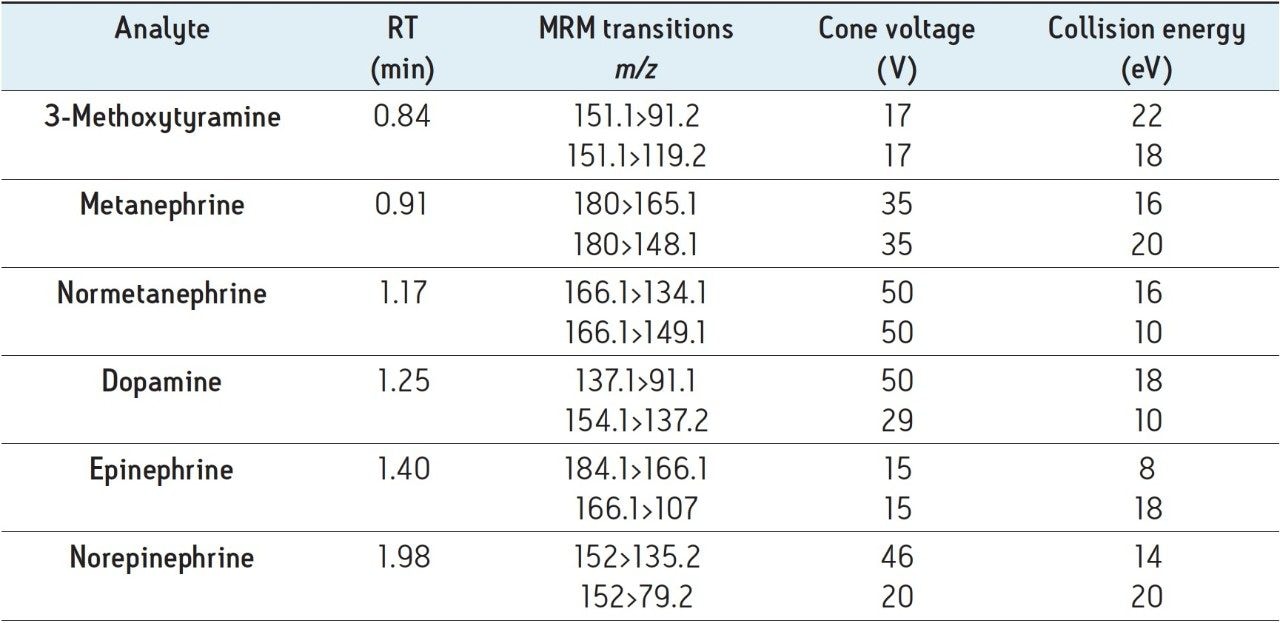
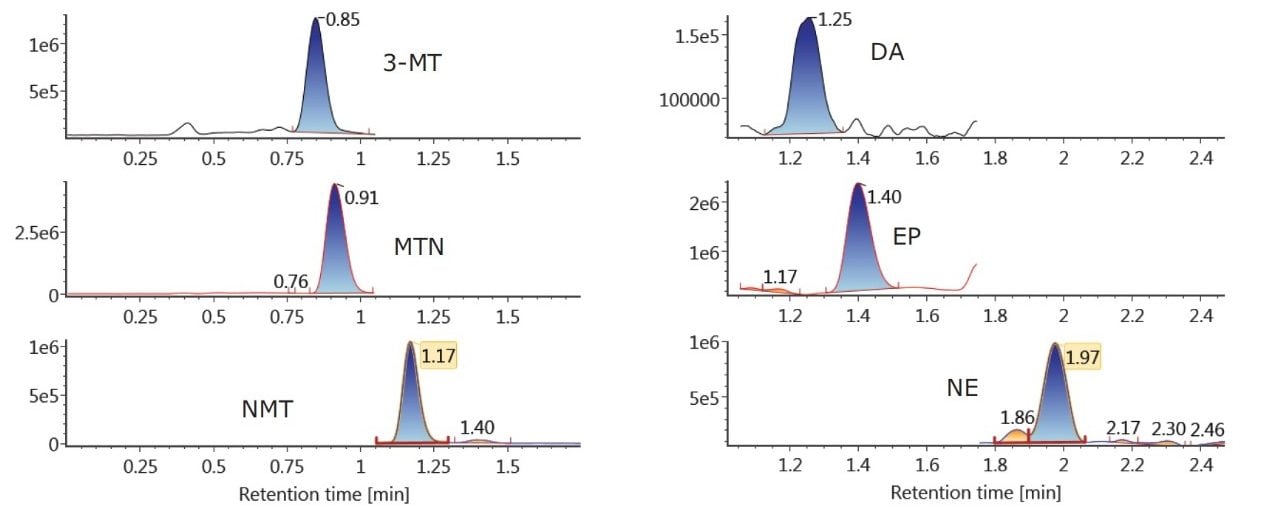
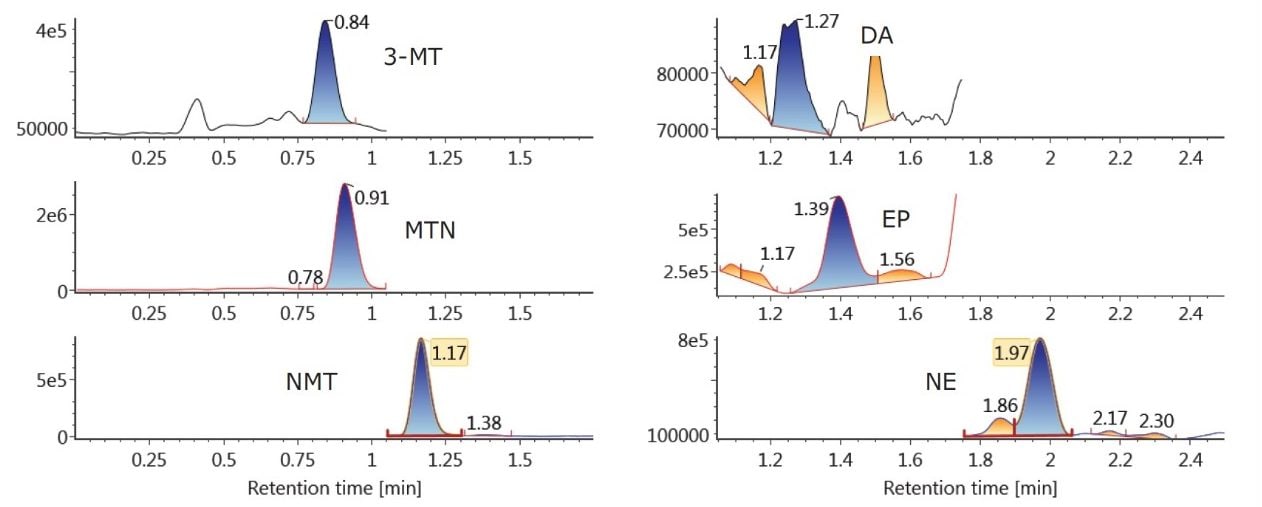
Figure 2A shows the chromatography of all compounds from a 20 pg/mL calibration standard using the ACQUITY UPLC BEH Amide Column. Previous work6 had shown that 30 mM NH4HCOO and 15% water in MPB resulted in an ideal balance of ionic strength and solubility, enabling the resolution and peak shape seen in Figure 2A. One important feature of this separation is the resolution between NMT and EP. These two compounds have the same molecular formula and can interfere with each other if not adequately separated. Figure 2A demonstrates the baseline separation of these compounds in HILIC mode, enabling their unambiguous identification and quantification. Figure 2B shows the HILIC chromatography of an unspiked plasma sample, demonstrating the ability to determine endogenous concentrations of 3-MT, MTN, NMT, DA, EP, and NE (7.0, 31.7, 70.6, 0.0, 29.4, and 360.9 pg/mL, respectively).
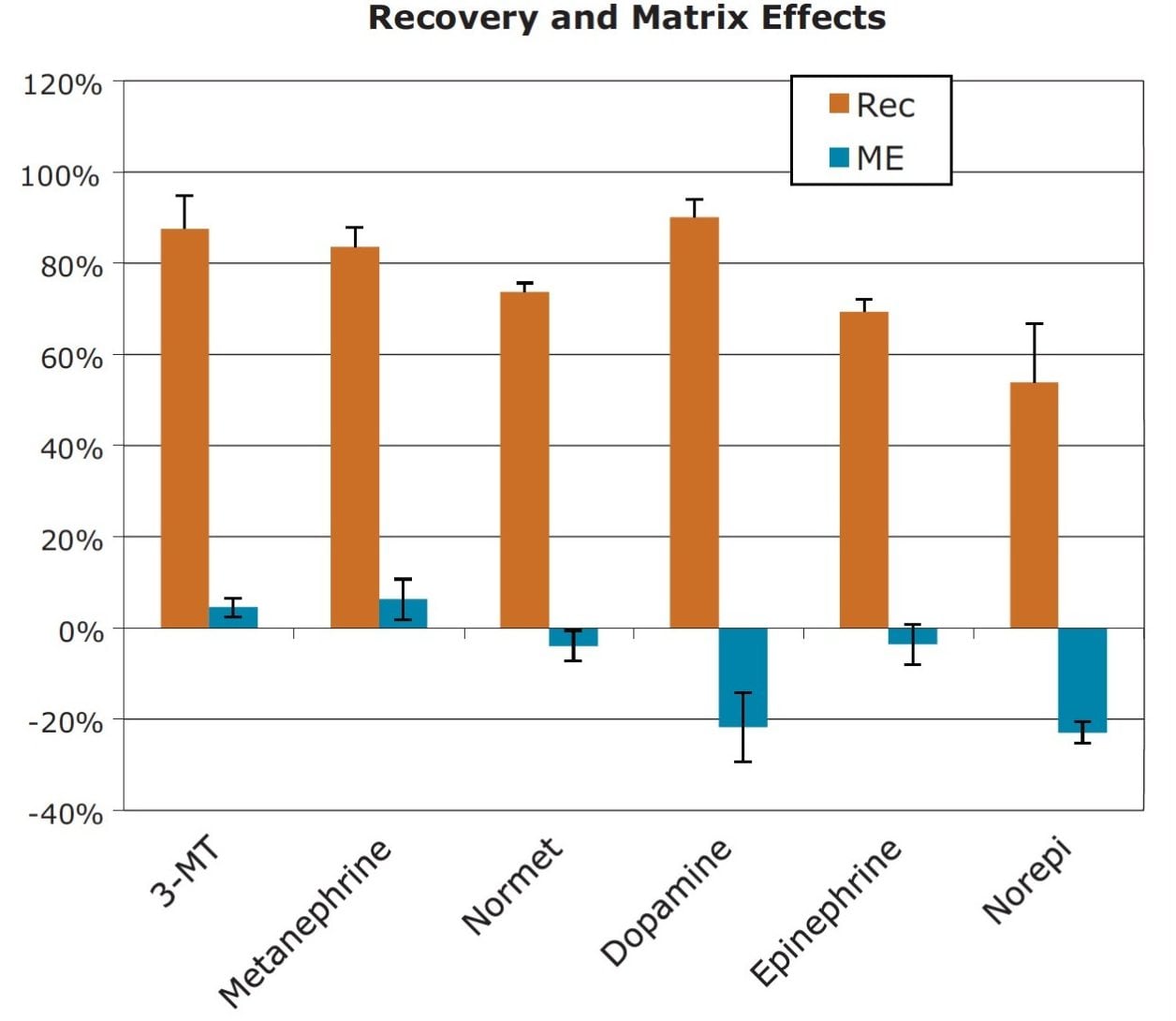
Extraction recoveries and matrix effects are shown in Figure 3. Recoveries ranged from 54% for NE to 90% for DA, with an average recovery of 76.4%. Matrix effects averaged -6.9%. The largest matrix effects were -23% and -22% for NE and DA, respectively, but were negligible for all other compounds. These results highlight another advantage of HIILIC chromatography, the ability to minimize matrix effects when analyzing polar compounds.
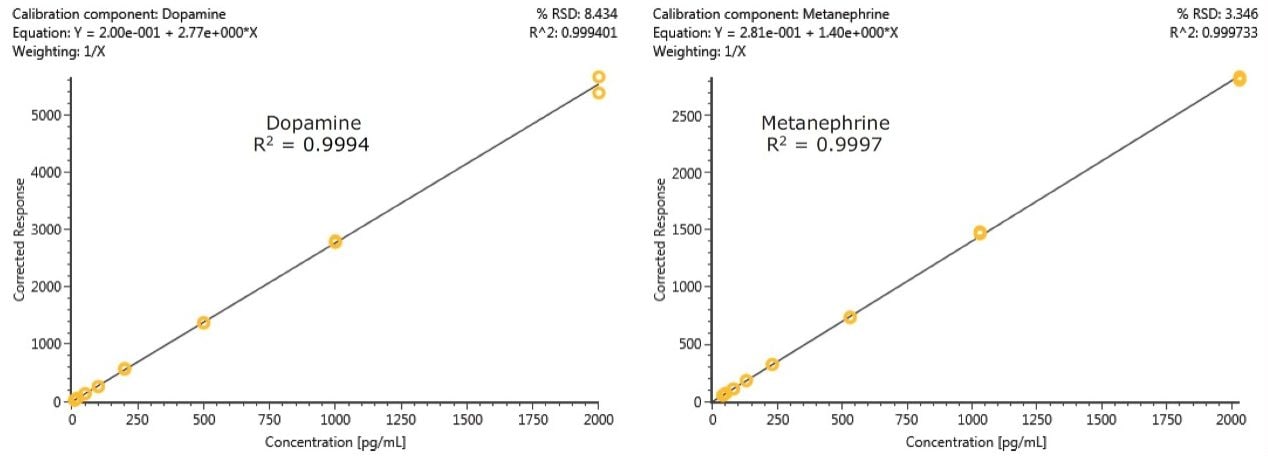
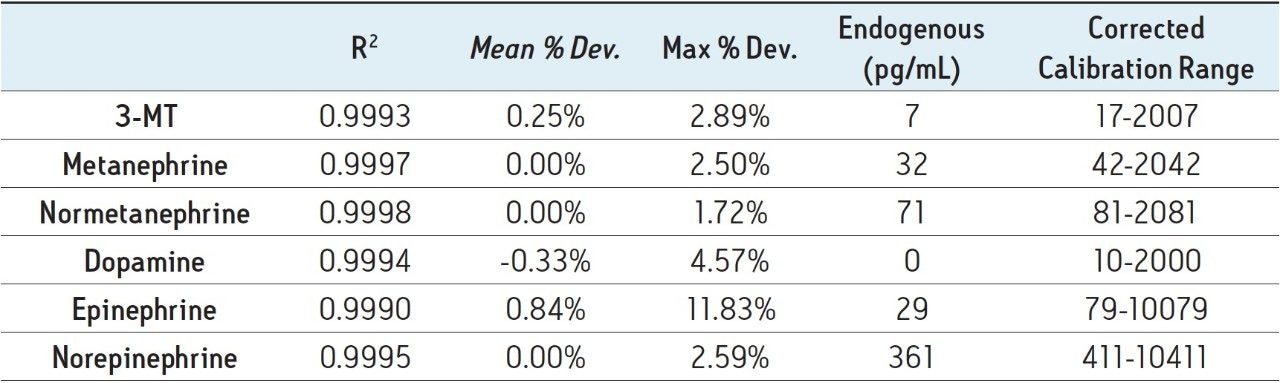
Calibration curves and quality control samples were prepared via the standard addition method by spiking pooled plasma samples with known concentrations of analytes. Two ranges of calibration curves were used, reflecting different expected concentrations of various compounds in plasma. Calibration levels for 3-MT, metanephrine, normetanephrine, and dopamine ranged from 10–2,000 pg/mL. Calibration levels for epinephrine and norepinephrine ranged from 50–10,000 pg/mL. After data processing, the endogenous concentrations were extrapolated from the resulting calibration curves. These data were used to correct the actual calibration concentrations. For example, the plasma sample used for calibration was determined to contain 31.7 pg/mL of metanephrine, so the calibration concentrations were adjusted to 41.7–2031.7 pg/mL. The resulting calibration curves showed excellent linearity, with R2 values of 0.999 or greater for all compounds. Figure 4 shows representative calibration curves for DA and MTN, both of which have R2 values greater than 0.999. Table 3 summarizes the calibration data for all compounds. Mean % deviations from expected calibration values were less than 1% for all analytes. In addition, the maximum % deviations from calibration values are listed and show that with the exception of epinephrine, the maximum % deviation for all calibrators was less than 10%. The calculated endogenous concentration of compounds in the pooled plasma used for calibration is also listed, along with the corrected calibration ranges for each compound.
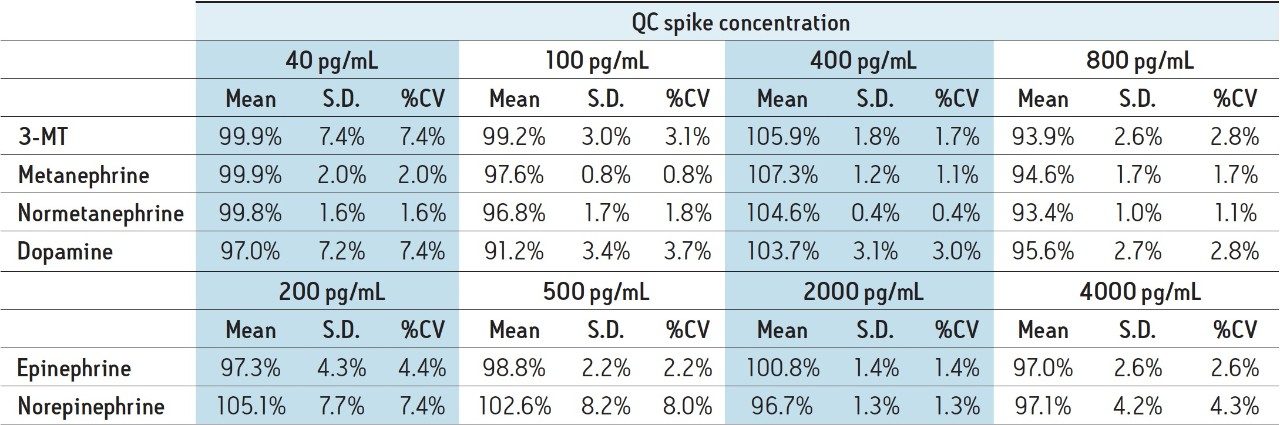
Quality control samples (N=6) were overspiked at 200, 500, 2000, and 4000 pg/mL for EP and NE and at 40, 100, 400, and 800 pg/mL for the remaining compounds. QC results were accurate and precise (see Table 4). All QC values were within 10% of their target values, and most were within 5%. In addition, all coefficients of variation (%CV) were less than 10%. This demonstrates that the method is linear, accurate, and precise over a calibration range that includes the entire scope of expected values for normal and pathologically elevated samples.
The extraction and analysis of plasma catecholamines and metanephrines using Oasis WCX µElution Plates and an ACQUITY UPLC BEH Amide Column in HILIC mode is detailed. Extraction using the Oasis WCX μElution Plate resulted in low matrix effects and consistent recoveries for all compounds that translated into excellent analytical accuracy and precision. In addition, the ability to elute the samples in an extremely low volume (50 µL) enabled 5x sample enrichment without the extra time or risk associated with evaporation and reconstitution. The ACQUITY UPLC BEH Amide Column used for HILIC separation resulted in rapid and efficient separation of all compounds, with baseline resolution between normetanephrine (NMT) and epinephrine (EP). It also enabled the analysis of the monoamines, dopamine, norepinephrine and epinephrine. Quantitative results were excellent, with highly linear responses across the entire calibration range and excellent accuracy and analytical precision.
720005094, July 2014