
This study demonstrates the options available to collect MS data with UPC2, using the analysis of liquid crystal intermediate compounds as an example.
Considering three ionization modes ESI, APCI, and APPI, optimizing the choice of modifier and tune parameters to illustrate how the high speed and unique selectivity of UPC2 can be combined with the greater selectivity and specificity that can be achieved using MS detection.
Convergence Chromatography (CC) is a separation technique that uses carbon dioxide as the primary mobile phase, with a co-solvent such as acetonitrile to give similar selectivity as normal phase LC. Waters UltraPerformance Convergence Chromatography (UPC2) builds upon the potential of CC, utilizing the benefits of sub-2 μm particle size stationary phases, while using proven and robust Waters UPLC technology.
Various detection methods can be used with UPC2 including UV and Evaporative Light Scattering Detection (ELSD). But there is also the option of interfacing UPC2 with Mass Spectrometry (MS) detection as illustrated in Figure 1. UPC2 can easily and quickly be connected to a wide-range of MS systems, with the addition of a MS splitter to the system, which introduces a controlled leak to the system and enables the maintenance of the CO2 pressure, while maintaining peak shape and width.
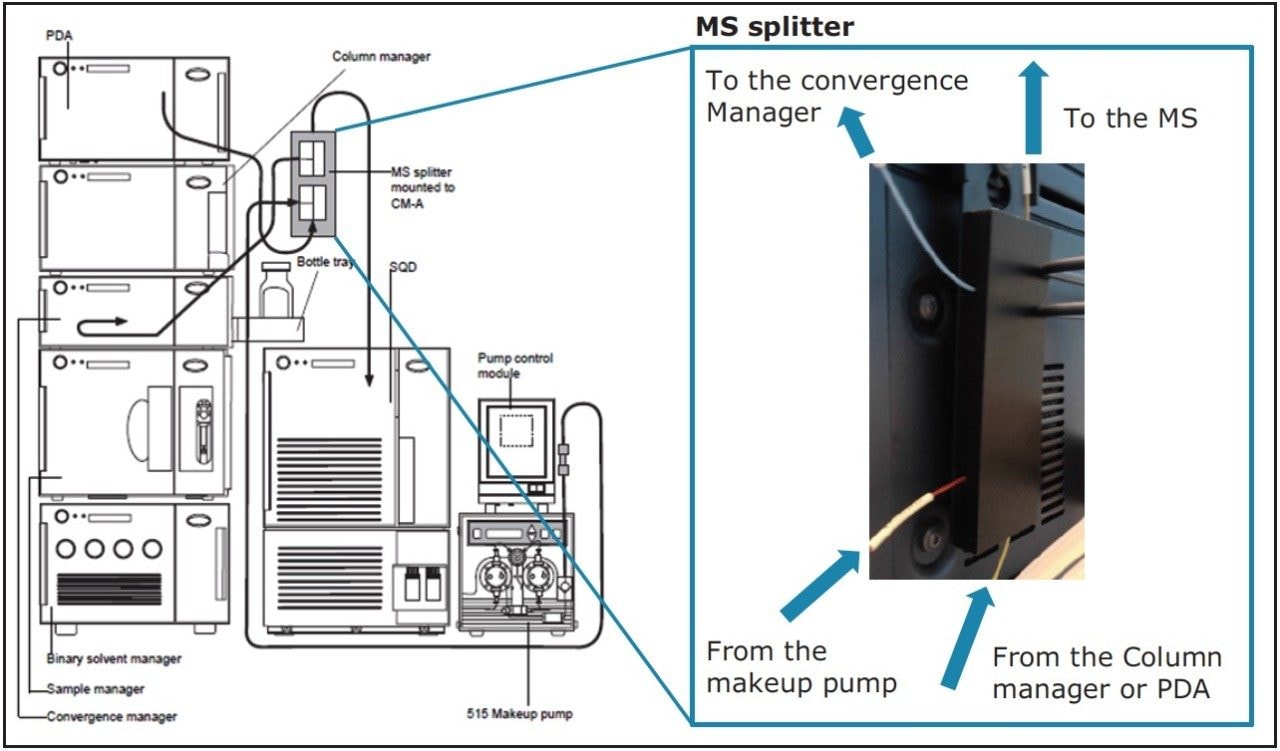
The MS splitter provides sufficient back pressure to maintain the CO2 in the liquid state and maintain its supercritical properties such as density and solvating power which could alter factors such as compound solubility and the selectivity of the stationary phase. The additional option to add a makeup solvent via a makeup pump to the flow prior to MS detection can be used to provide greater solvating powers, to enhance the selectivity and sensitivity of MS detection, and also to influence ionization.
When using electrospray ionization (ESI) where an electrically charged field is used to generate charged droplets, then analyte ions are formed by evaporation prior to MS analysis. The addition of a protonation source such as formic acid to the makeup solvent can be used to enhance ionization and increase sensitivity. In atmospheric pressure photo ionization (APPI), ultraviolet light produced from a krypton lamp ionizes gas phase analytes and dopants leading to gas-phase reactions. Therefore, the addition of a dopant such as toluene to the makeup solvent can enable and enhance ionization. Whereas when using atmospheric pressure chemical ionization (APCI), the solvent present, from both the co-solvent and the makeup solvents, acts as chemical ionization reagent gas in order to ionize the sample.
Waters’ Xevo family of MS systems share a universal source platform which enables easy, quick, and tool-free source exchange, without venting the system.1 This facilitates the quick optimization and screening of the compounds of interest using different ionization modes when using UPC2.
This document demonstrates interfacing UPC2 with Xevo TQD, considering three atmospheric pressure ionization modes ESI, APCI, and APPI for the analysis of liquid crystal intermediate compounds as a model. This illustrates how the high speed and unique selectivity of UPC2 can be combined with the greater selectivity and specificity that can be achieved using MS detection.
|
UPC2 conditions |
|
|---|---|
|
System: |
ACQUITY UPC2 |
|
Run time: |
5.00 min |
|
Column: |
ACQUITY UPC2 BEH 2-EP, 3.0 mm x 100 mm, 1.7 μm |
|
Column temp.: |
65 °C |
|
CCM back pressure: |
2000 psi |
|
Sample temp.: |
20 °C |
|
Mobile phase A: |
CO2 |
|
Mobile phase B: |
Methanol |
|
Flow rate: |
2.0 mL/min |
|
Injection volume: |
1 μL |
|
Vials: |
Waters Amber Glass 12 x 32 mm Screw Neck Vial, 2 mL |
|
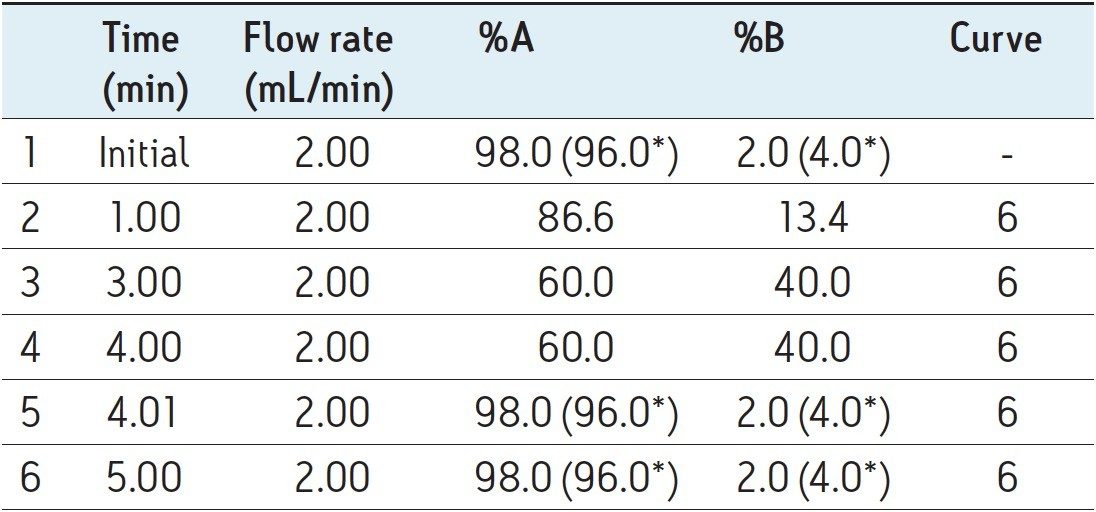
Mobile phase gradient is detailed in Table 1. |
|
PDA conditions |
|
|---|---|
|
UV system: |
ACQUITY UPC2 PDA Detector |
|
Range: |
210 to 450 nm |
|
Resolution: |
1.2 nm |
|
Sampling rate: |
20 pts/sec |
|
Filter time constant: |
Slow (0.2 sec) |
The liquid crystal intermediate compounds were purchased from Sigma-Aldrich. Individual stock solutions were prepared to a concentration of 5 mg/mL, dissolved in either heptane/ethanol (9:1) or methanol. Serial dilutions of the stock solutions were preformed in heptane/isopropanol (9:1) in order to prepare mixed standards, or in methanol to prepare infusion MS tuning standards.
MassLynx Software, v4.1, SCN 882, was used to control the ACQUITY UPC2 and the Xevo TQD and also for data acquisition.
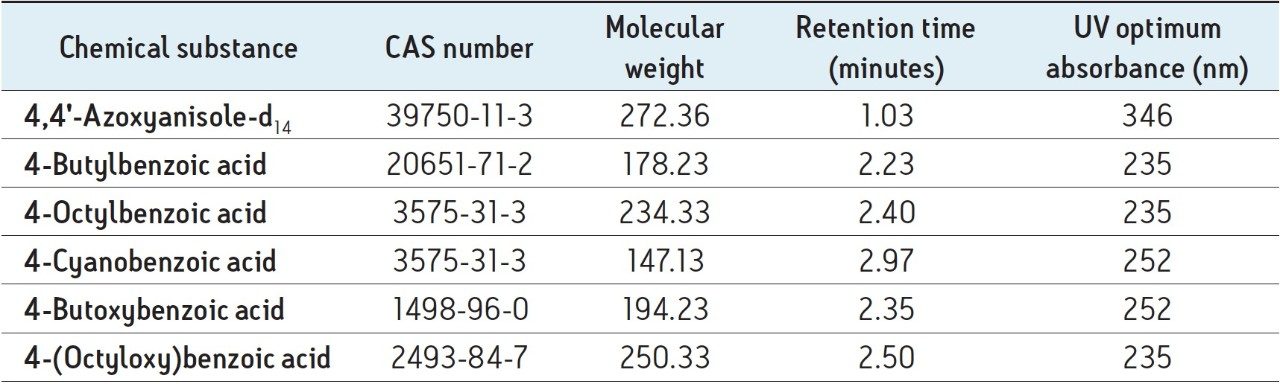
The UPC2 conditions were optimized for the analysis of five liquid crystal intermediate compounds along with one internal standard using the Waters ACQUITY UPC2 System equipped with an ACQUITY UPC2 PDA Detector. Different UPC2 columns, co-solvents, column temperatures, mobile phase temperatures, and system CO2 back pressures were considered. Retention times were established by analyzing single component standards, as described in Table 3.
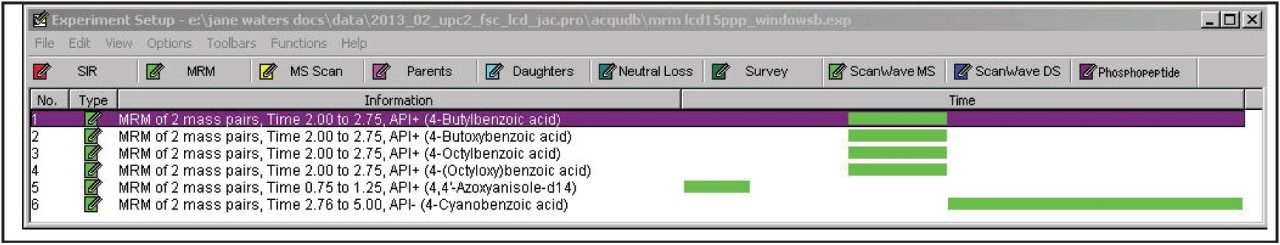
Using the on-board fluidics system on the Xevo TQD, 2-ppm individual standards were infused into the source using APCI ionization in order to optimize the APCI MS conditions (Table 2) and the MRM conditions (Table 4). The established MRM conditions were also used in APPI and ESI ionization modes. The established MRM method is shown in Figure 2.
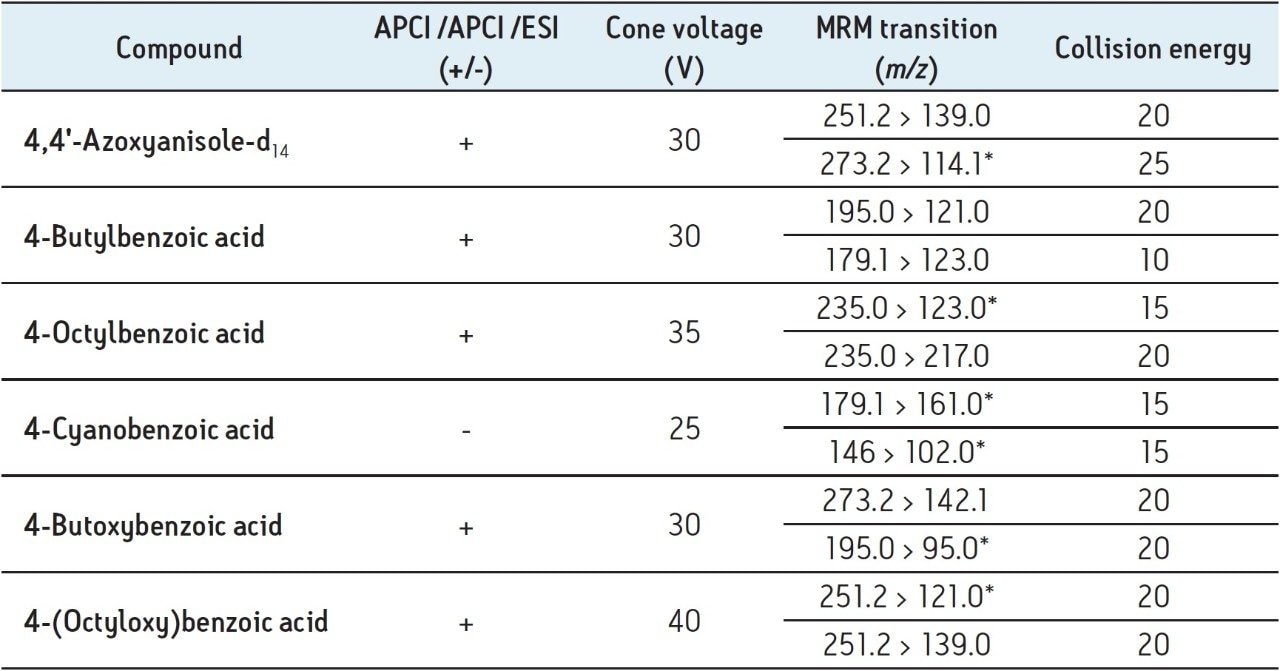
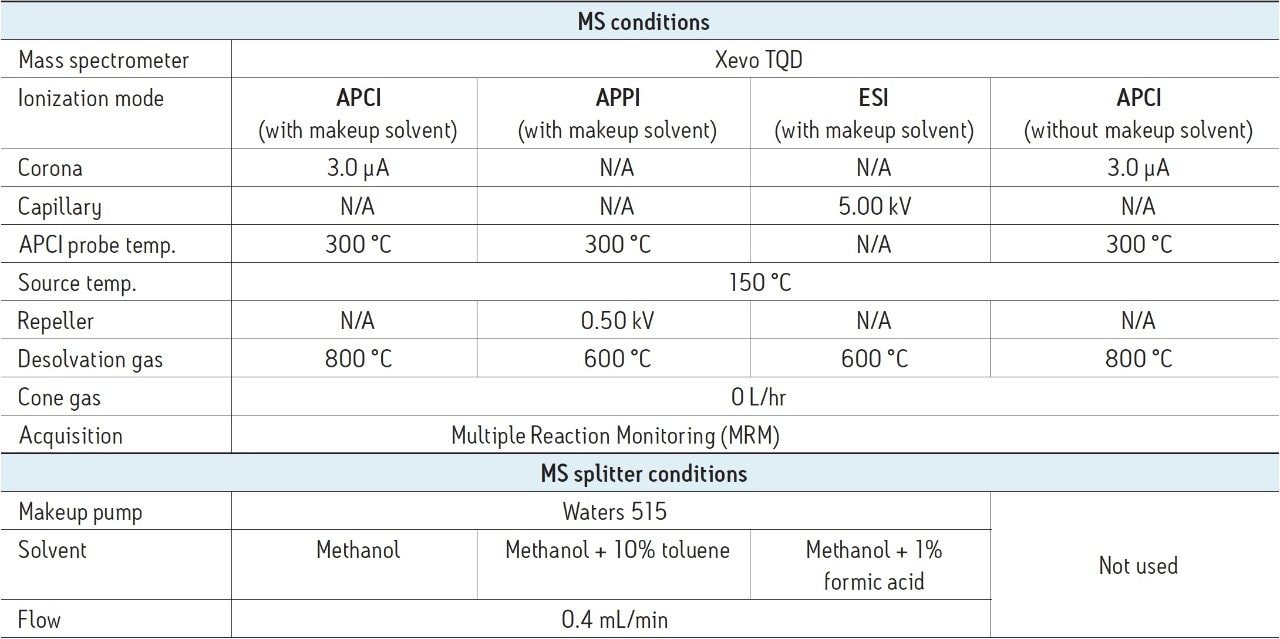
The APPI, ESI MS conditions, and the MS splitter conditions were optimized (Table 2) by analyzing mixed 0.1 mg/mL calibration standards using the optimized UPC2 conditions, and MRM method using either APCI, APPI, or ESI ionization modes as required. MS conditions considered included desolvation temperature and flow, cone gas flow, repeller voltage (APPI), and capillary voltage (ESI). When considering the MS splitter conditions, the makeup solvent and flow required for each ionization mode were optimized. Using ESI formic acid was added as a protonation source to the makeup solvent to enhance ionization and increase sensitivity. Whereas, when using APPI a dopant (toluene) was added to the makeup flow to enable and enhance ionization. When considering APCI the solvent present, from both the co-solvent and the makeup solvents, were considered in order to optimize the ionization of the sample.
Mixed 0.1 mg/mL calibration standards were analyzed using the optimized UPC2, MS and MS splitter conditions as detailed ( Table 2) with the addition of a makeup solvent via the MS splitter, using APCI, APPI, and ESI ionization modes. The resulting MRM chromatograms are shown in Figure 3.
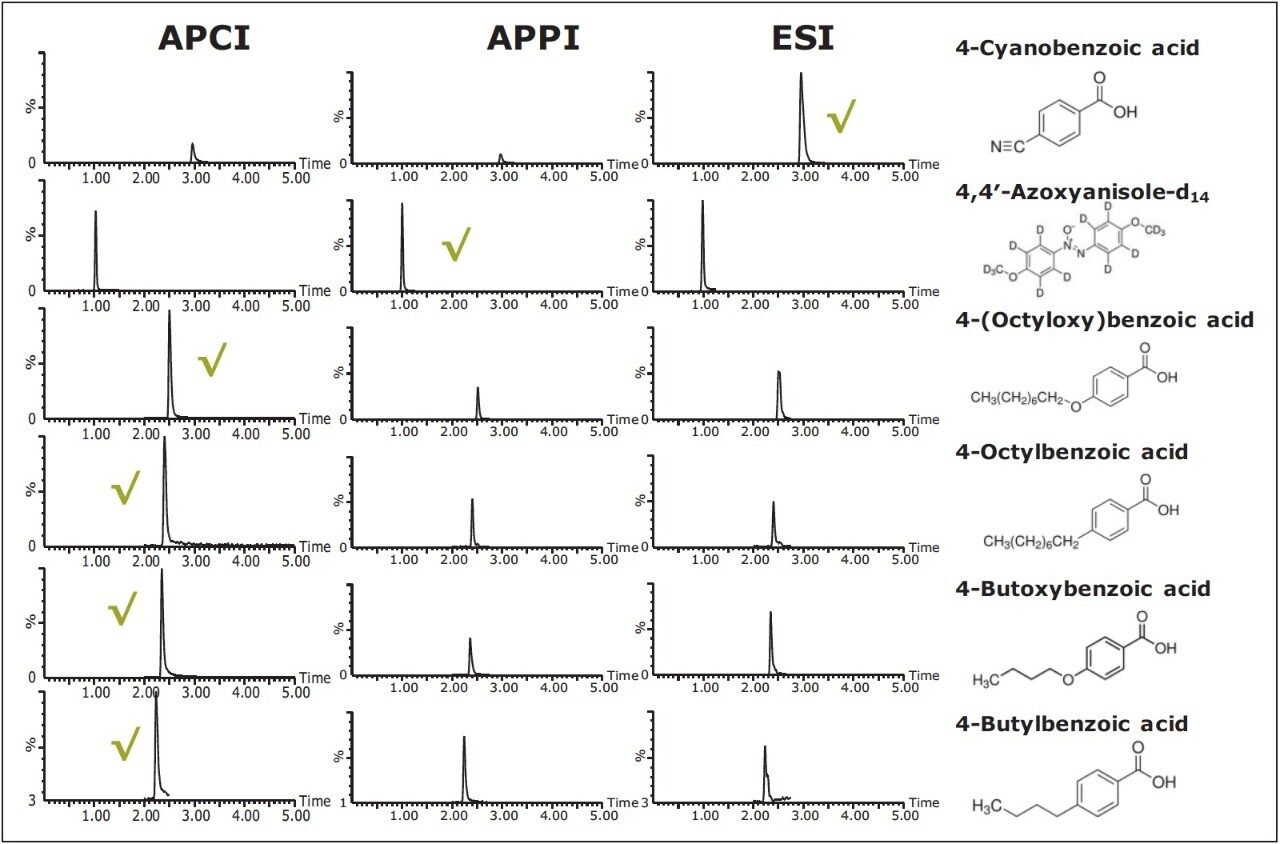
There is also the option when optimizing conditions of not adding an additional makeup solvent via the MS splitter, and purely using the co-solvent to aid ionization. This was considered using APCI ionization mode, where no additional buffers or dopants are required to aid ionization.
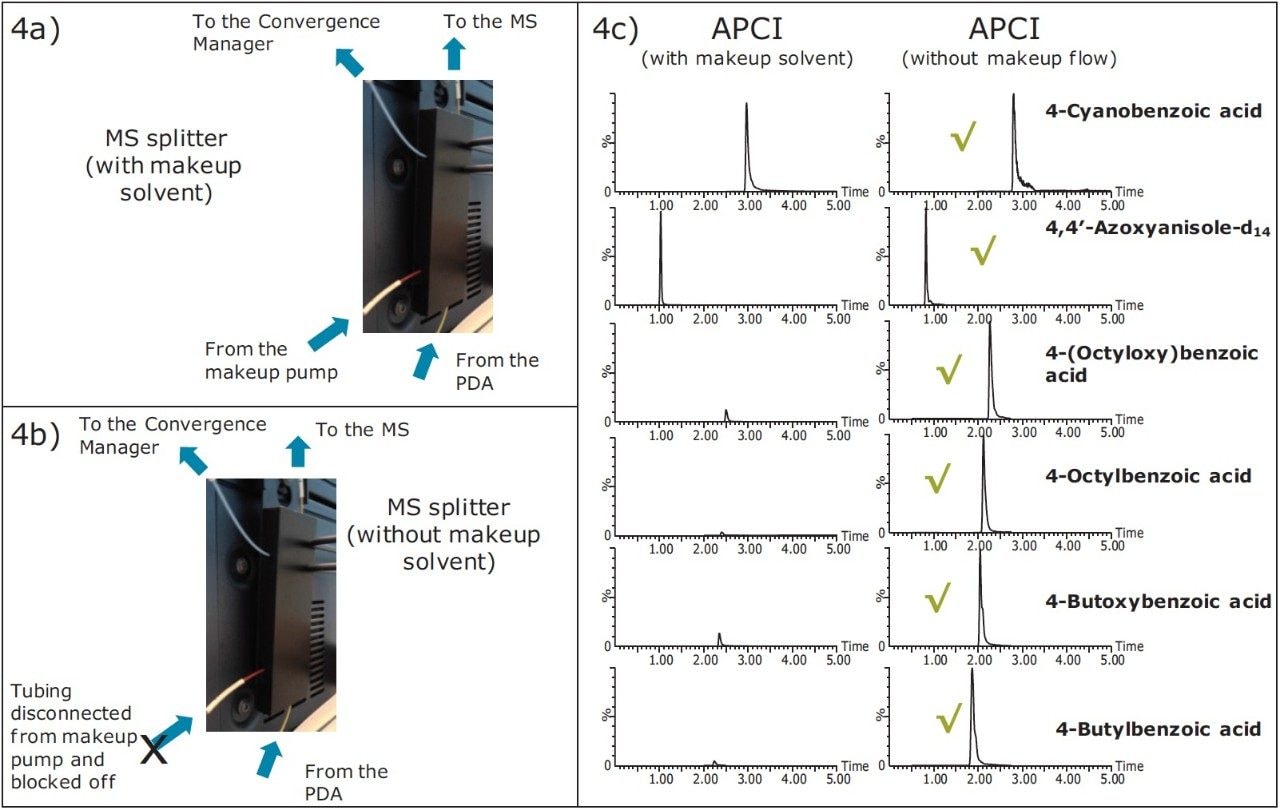
Using the UPC2, MS and MS splitter conditions as detailed in Table 2, APCI (without makeup solvent) mixed 0.1 mg/mL calibration standard were analyzed. No additional solvent was added via the MS splitter, the same system configuration was used, with the exception that the tubing from the makeup pump to the MS splitter was isolated off (Figures 4a and 4b).
When using the initial 2% co-solvent the response of the earlier eluting peak was markedly reduced. In this example by increasing the initial co-solvent percentage from 2% to 4% and using no additional makeup solvent overall higher response values were observed for all the compounds considered. The MRM chromatograms using APCI with and without additional makeup solvent are shown in Figure 4c.
This document demonstrates the options available to collect MS data with UPC2, using the analysis of liquid crystal intermediate compounds as an example.
UPC2 can easily and quickly be connected to a Xevo TQD with the addition of an MS splitter, which introduces a controlled leak to the system and enables the maintenance of the CO2 pressure. That makes available the option to add a modifier to the flow prior to MS detection, which can provide greater solvating powers, enhance the selectivity and sensitivity of MS detection, and also influence ionization.
Waters' Xevo family of MS systems share a universal source platform which enables easy, quick and tool free source exchange, allowing different ionization modes to be quickly optimized and screened for the compounds of interest when using UPC2. Here we considered three ionization modes ESI, APCI, and APPI, optimizing the choice of modifier and tune parameters to illustrate how the high speed and unique selectivity of UPC2 can be combined with the greater selectivity and specificity that can be achieved using MS detection.
720004781, September 2013