In this study, we evaluate Charged Surface Hybrid (CSH) C18 column chemistry for peptide separations, expecting similar improvements in chromatographic performance since most peptides under commonly employed acidic conditions contain a positive charge. This work with peptides demonstrates that this novel stationary phase exhibits greater peak capacity, unique selectivity, and less dependence on MS signal suppression, strong ion pairing agents in peptide analysis.
Peptide separations are of paramount importance, whether studying proteins by bottom-up proteomics1 or thoroughly characterizing biopharmaceuticals using peptide mapping.2 The mixtures encountered in these analyses are inherently complex. Reversed-phase (RP) chromatography has become the separation mode of choice, as it offers relatively high resolving power and easily interfaces with mass spectrometry (MS).
The performance metric most relevant to gradient RP chromatography is peak capacity, or the maximum number of peaks that can fit into the gradient time.3 Interestingly, chemical modifications of a stationary phase, in addition to the use of smaller particle sizes, can significantly impact peak capacity. Charged surface hybrid (CSH) C18 column chemistry, for example, is an evolution of the ethylene bridged hybrid (BEH) C18 stationary phase4 as its surface is modified to contain a low-level positive charge in addition to the C18 bonded phase.5 This modification has been shown to improve peak shapes, loading behavior, and peak capacities for small, ionized molecules.5-10
In this study, we evaluate CSH130 C18 for peptide separations, expecting similar improvements in chromatographic performance since most peptides under commonly employed acidic conditions contain a positive charge. This work with peptides demonstrates that this novel stationary phase exhibits greater peak capacity, unique selectivity, and less dependence on MS signal suppressing, strong ion pairing agents when compared to the state-of-the-art in peptide analysis.
|
System: |
Waters ACQUITY UPLC H-Class Bio System with a 20 or 30 cm Column Heater (20 cm Column Heater for 150 mm columns and 30 cm Column Heater for 250 mm columns) |
|
Detection: |
ACQUITY UPLC TUV Detector with 500 nL Analytical Flow Cell; Xevo G2 QTof Mass Spectrometer |
|
Wavelength: |
214 nm |
|
Scan rate: |
10 Hz |
|
Columns: |
ACQUITY UPLC BEH130 C18, 2.1 x 150 mm, 1.7 μm, Porous, 130Å (p/n 186003556) ACQUITY UPLC CSH130 C18, 2.1 x 150 mm, 1.7 μm, Porous, 130Å (p/n 186006938) XSelect CSH130 C18 XP, 2.1 x 150 mm, 2.5 μm porous, 130Å (p/n 186006943) C18, 2.1 x 250 mm, 5 μm, porous, 300Å (competitor product) C18, 2.1 x 150 mm, 1.7 μm, superficially porous (1.25 μm core, 0.22 μm shell) 100Å (competitor product) |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
10 μL |
|
Flow rate: |
0.3 mL/min |
|
Mobile phases: |
A: 0.1% FA (v/v) in water B: 0.1% FA (v/v) in acetonitrile (ACN) C: 0.1% trifluoroacetic acid (TFA) (v/v) in water D: 0.1% TFA (v/v) in ACN |
|
Vials: |
LCGC Certified Clear Glass 12 x 32 mm Screw Neck Qsert Vial (p/n 186001126C) |
|
Gradient: |
2% ACN for 1 min, then to 50% ACN for 60 min (Acid composition was controlled by proportioning mobile phases A/B and C/D.) |
|
Mass spectrometer: |
Xevo G2 QTof |
|
Ionization mode: |
ESI+ |
|
Analyzer mode: |
Resolution |
|
Capillary voltage: |
3.00 kV |
|
Cone voltage: |
25 V |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
350 °C |
|
Cone gas flow |
0.0 L/h |
|
Desolvation gas flow: |
800 L/h |
|
Calibration: |
NaI 2 μg/μL from 50 to 2000 m/z |
|
Acquisition: |
50 to 1990 m/z, 10 Hz scan rate |
|
Data management: |
MassLynx Software |
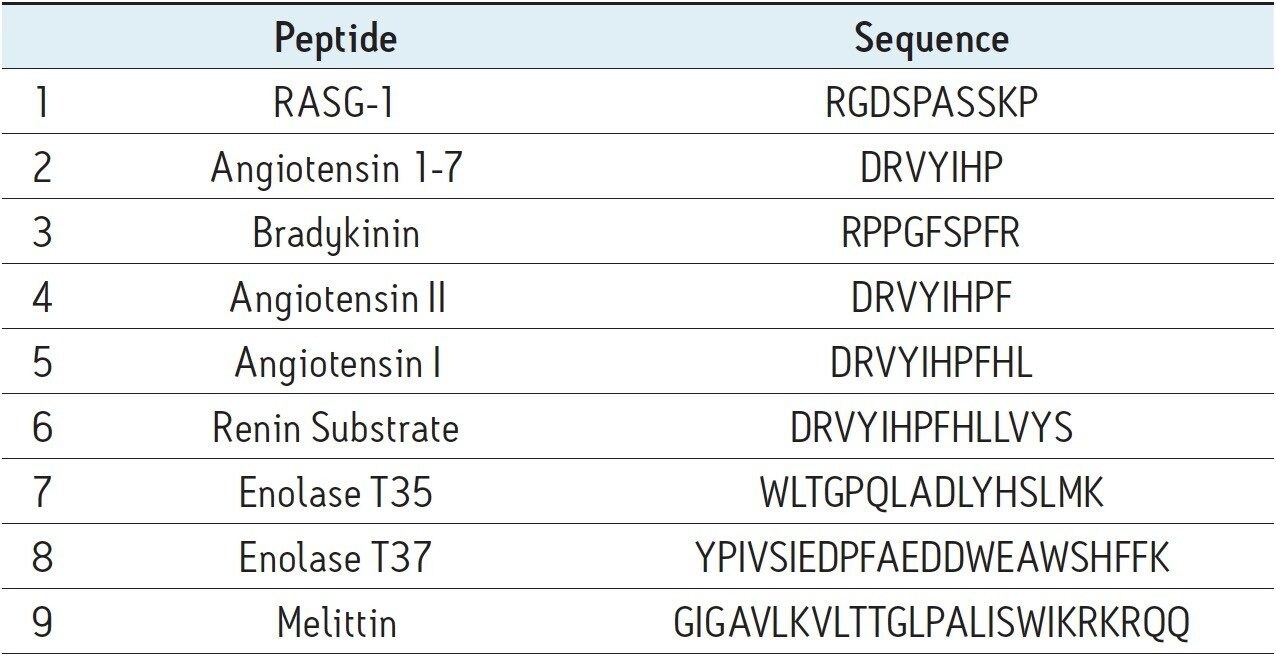
MassPREP Peptide Mixture (p/n 186002337), as shown in Table 1, was reconstituted with 0.1% FA in water to a total peptide concentration of ca. 0.6 mg/mL.
Calculations:
Peak capacities for each separation were calculated by measuring peak widths for peptides 2 through 7. Peptides 8 and 9 co-eluted in several of the separations, precluding them from the calculations. Similarly, peptide 1 (RASG-1) was excluded from the calculation because it eluted from the CSH130 C18 column near the void volume using 0.1% FA in the mobile phase. Peak widths were measured from the UV chromatograms at peak half-height (wh), averaged, then subsequently converted to 4σ peak capacities according to the following equation:
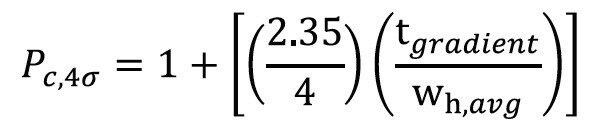
The MassPREP Peptide Mixture contains nine different peptides, varying in amino acid composition, mass, length, and polarity. Peptide sequences are shown in Table 1. Since this mixture is composed of such a diverse set of peptides, it is useful for evaluating mass spectrometric as well as chromatographic performance. To this end, the peptide mixture was employed to benchmark the separation performance of CSH130 C18 against other commonly used stationary phases for peptide analysis. Included in this study were phases with pore sizes large enough in diameter (100 to 300Å) to efficiently separate the peptides in the mixture, which are all less than 3 kDa in mass. In a separate application note, the use of CSH130 C18, with its 130Å pores for separating even larger polypeptides is discussed.11 For the sake of relevance, mass loads for the individual peptides in the mixture were made approximate to conditions common for peptide mapping of antibodies, wherein 20 to 50 μg of Lys-C or tryptic digests are typically analyzed on 2.0 or 2.1 mm I.D. columns.2,12-13 Similarly, separations were completed with mobile phases containing a strong ion pairing agent, TFA, a weaker ion pairing agent, FA, or a combination thereof. Performance under these various conditions was of interest, since RP peptide separations are routinely coupled with mass spectrometry. In such applications, formic acid is often preferred over TFA as a mobile phase additive because it permits more sensitive detection.14-15
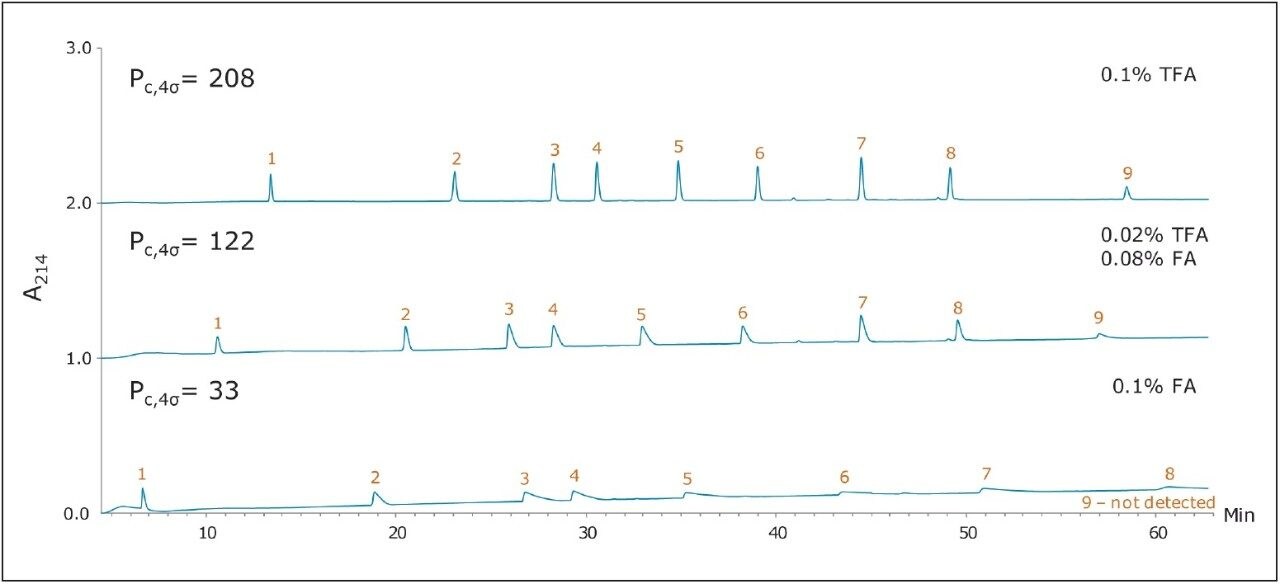
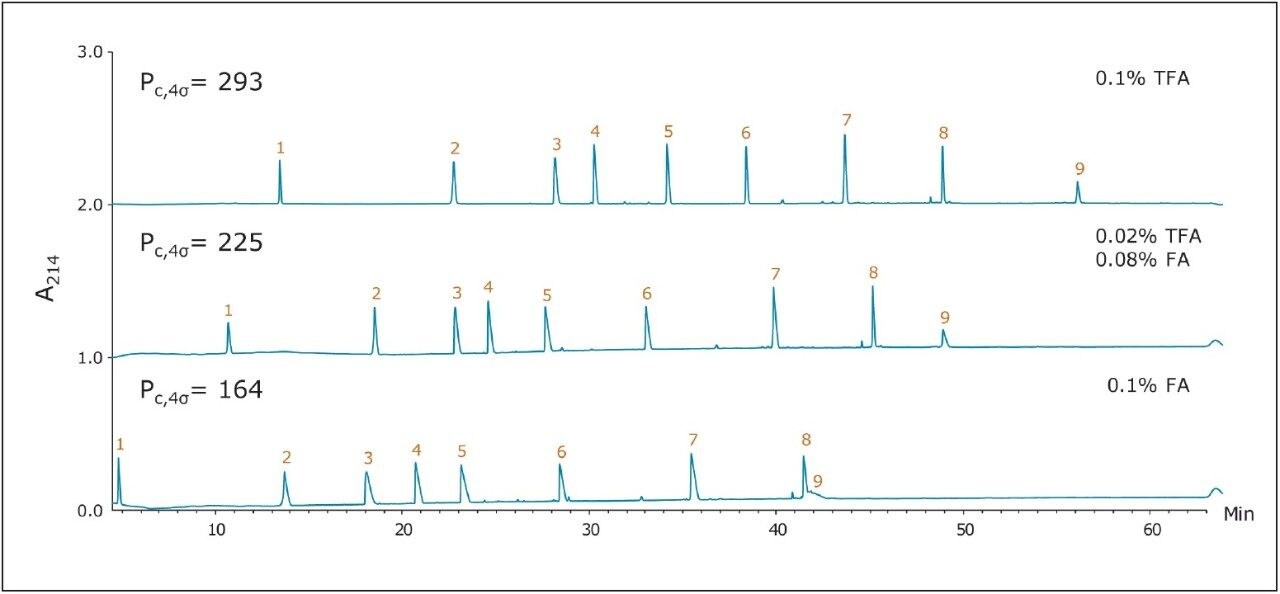
Figure 1 shows three chromatograms of the MassPREP Peptide Mixture obtained using a 5 μm silica C18 column under different mobile phase conditions. Separations with TFA provided peaks that were generally symmetrical. Most peaks did not exhibit excessive broadening or tailing. However, peak shape was found to be extremely poor with the FA containing mobile phases. In addition, the largest peptide in the mixture, melittin, was not detected, either because it eluted as too broad a peak or simply failed to elute. Results such as these are typical for HPLC-based peptide separations.
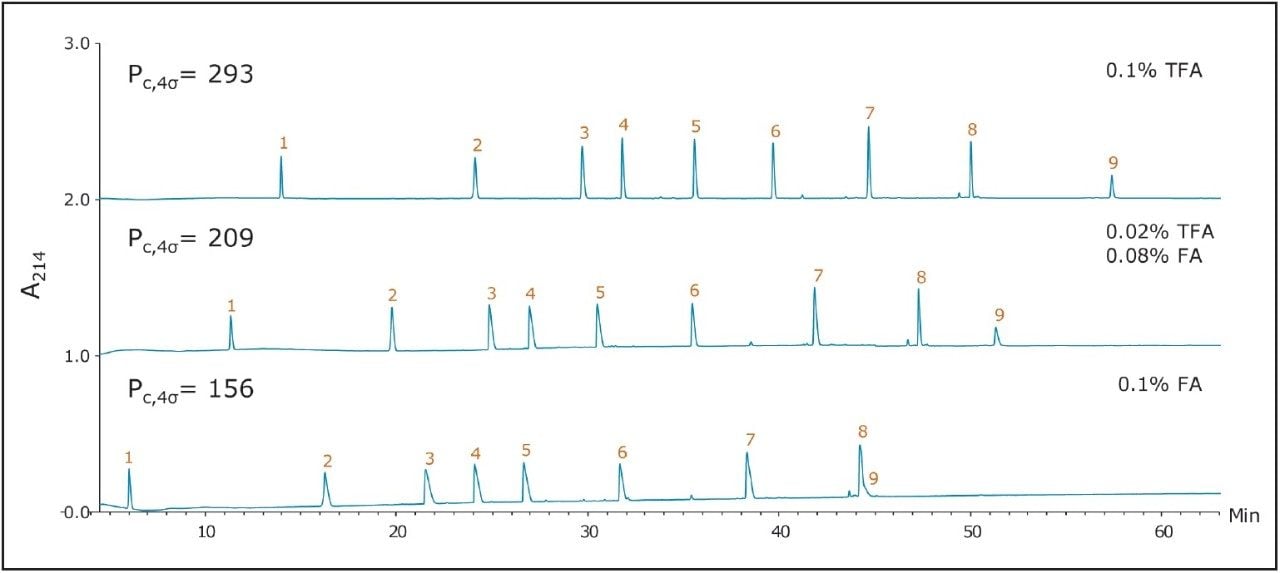
The use of sub-2-μm particles provides performance benefits to peptide separations. Chromatograms obtained for two different columns both packed with 1.7 μm particles are displayed in Figures 2 and 3. The chromatograms in Figure 2, in particular, were obtained using a superficially porous C18 column. With TFA in the mobile phase, the peaks were symmetrical and generally narrower than peaks obtained with the 5 μm, porous C18 column, shown in Figure 1. With little or no TFA in the mobile phase, peaks became broader and exhibited significant tailing. The fully porous BEH130 C18 column produced comparable separations to the superficially porous C18 column, shown in Figure 3, in all conditions tested.
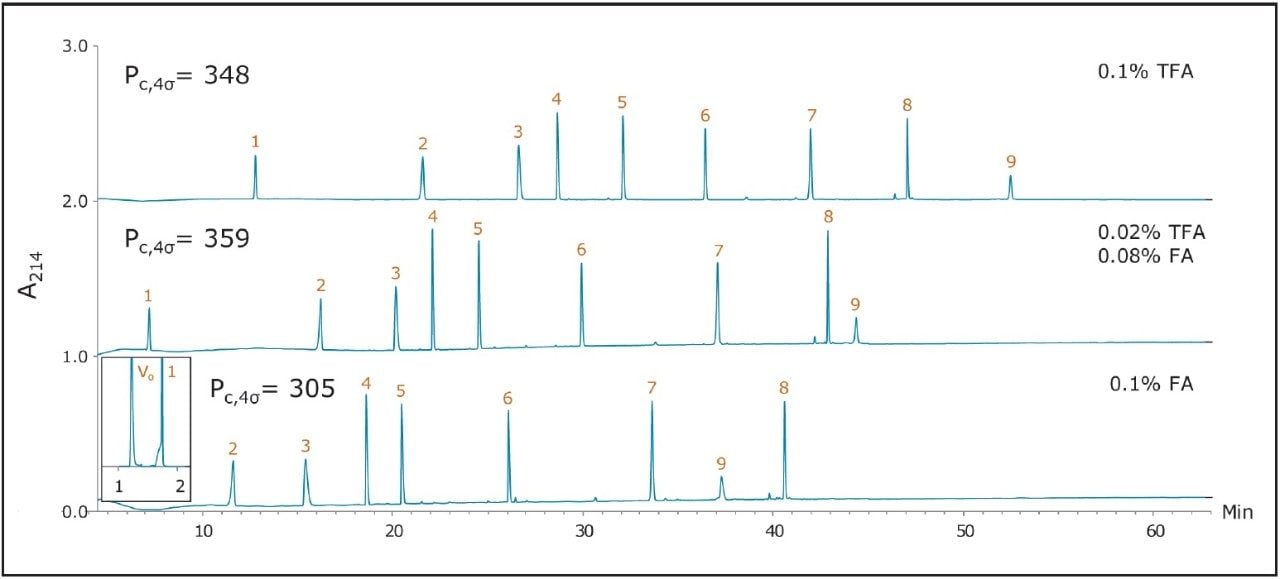
The separations shown in Figures 1 through 3 served to benchmark the performance of a CSH130 C18, 1.7 μm column for peptide analyses. Chromatograms corresponding to separations with this column are shown in Figure 4. Comparison of Figures 1 through 4 clearly indicates that, unlike the other columns, CSH130 C18 provides the best peak shape with both TFA and FA mobile phases. Additionally, it is evident that CSH130 C18 exhibits unique selectivity, particularly when TFA is excluded from the mobile phase. For instance, with only 0.1% FA in the mobile phase, peptides 8 and 9 were not well resolved on any column used in this study other than the CSH130 C18, when the two peptides eluted as symmetrical peaks and were separated by more than 3 min. Retention differences were also observed among these columns. Most notably, the CSH130 C18 column was found to be slightly less retentive than each of the other columns included in this study. This is consistent with CSH130 C18 being the only sorbent with a purposely incorporated low level positive surface charge. Coulombic repulsion between the positively charged peptides and the CSH130 C18 surface is likely to be the cause of reduced retention. The insert in Figure 4 displays the earliest portion of the 0.1% FA gradient on CSH130 C18. Under those conditions, the most hydrophilic peptide RASG-1 (peptide 1) eluted near the void marker (Vo). On average, peptides eluted from the CSH130 C18 column with 2% to 4% less ACN when compared to the BEH130 C18 column. Differences in retention were least pronounced with 0.1% TFA and most pronounced with 0.1% FA. When analyzing very polar peptides, initial gradient conditions for the CSH130 C18 may require adjustment accordingly.
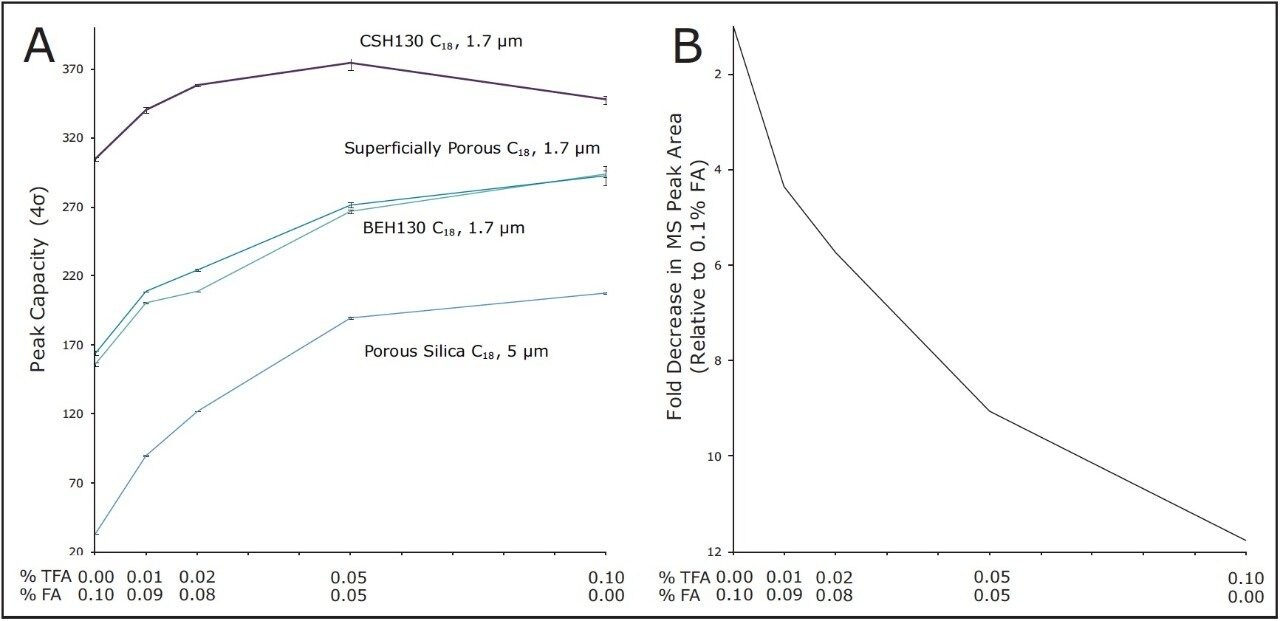
A qualitative comparison of these separations indicates that the CSH130 C18 column offers a performance advantage over the other columns. However, a quantitative comparison is even more compelling. Peak capacities observed for each separation were calculated as described in the experimental section. Figure 5A shows the relationship between acid modifier composition and the peak capacities obtained with each column. The left side of Figure 5A shows the peak capacity values for separations achieved with only FA and no TFA ion pairing agent. Moving right along the x-axis presents peak capacity values corresponding to decreasing concentrations of FA and increasing concentrations of TFA.
The performance advantage of the CSH130 C18 column is easily visualized. With 0.1% TFA, the CSH130 C18 column has 20% higher peak capacity than the other 1.7 μm C18 columns. However, when the mobile phase contains 0.1% formic acid, the CSH130 C18 column provides 90% greater peak capacity compared to the other 1.7 μm columns. Thus, not only does the CSH130 C18 column exhibit greater peak capacity than the other columns, its performance is far less dependent on TFA. A profound consequence of this is that CSH130 C18 is highly compatible with mass spectrometry. The effect of TFA on the MS detection of the peptides in these analyses is shown in Figure 5B. It is well known that TFA causes significant ion suppression during electrospray ionization.16-17 Here, the use of TFA instead of FA caused a 12-fold decrease in MS signal. CSH130 C18 does not require much, if any, TFA for optimal peak capacity, making it ideal for LC-MS applications when high sensitivity is desired.
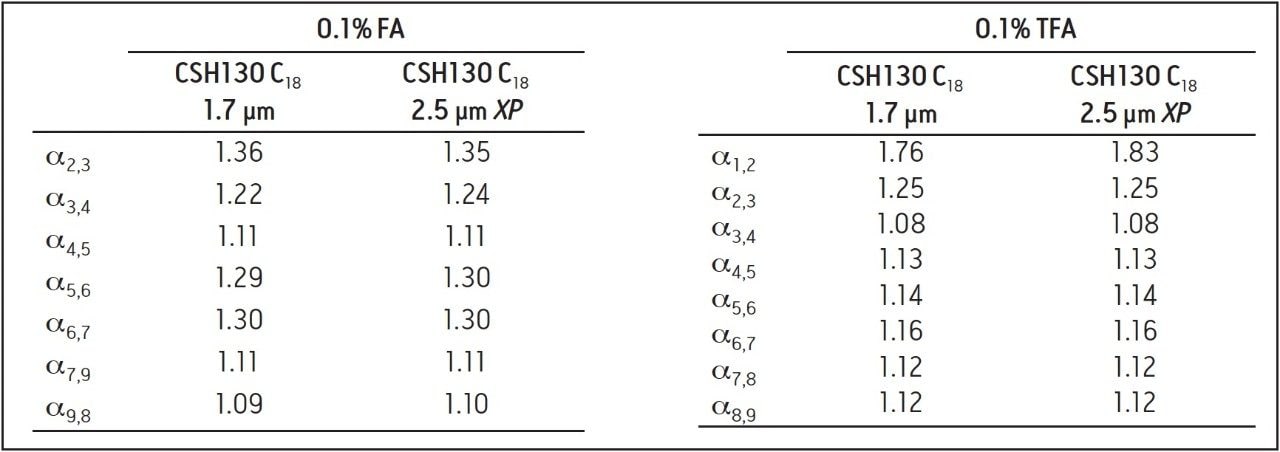
Many peptide separations are still performed on traditional HPLC instruments. Due to their prohibitively high back pressures, columns packed with sub-2-μm particles are generally not well suited for use with HPLC systems. Conversely, columns packed with 2.5 μm particles can be used on any LC instrument due to their lower back pressure. To determine if the high peak capacity peptide separations obtained using a CSH130 C18, 1.7 μm column could be successfully transferred to a CSH130 C18 XP, 2.5 μm column, an HPLC compatible method for a 2.5 μm XP column was programmed from the method for a 1.7 μm column. This was done by decreasing flow rate and increasing the gradient time by a factor of 1.5, as suggested by the ACQUITY UPLC Column Calculator.18 Figure 6 shows that separations with the 1.7 μm column were successfully transferred to the 2.5 μm XP column. The consistency of the selectivity factors underscores this observation, as shown in Table 2. Separations with the 2.5 μm XP particle column resulted in considerably lower back pressures compared to the 1.7 μm particle column (~3000 versus ~8000 psi), thereby demonstrating that CSH technology can be readily employed for high peak capacity peptide separations using either UPLC or HPLC instrumentation.
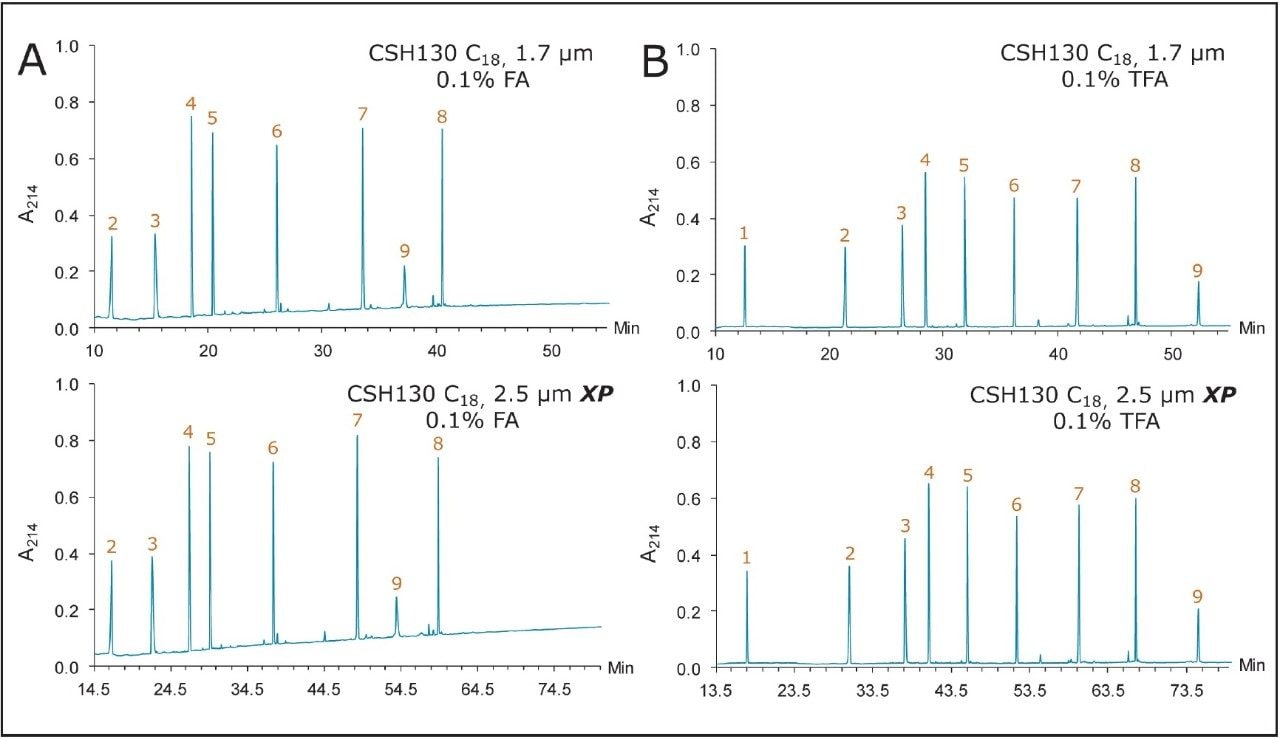
The CSH130 C18 column, with its novel incorporation of a low level positive charge, has proven to be an enabling technology for peptide separations. Based on analyses of a nine-peptide mixture, it was found that CSH130 C18 columns exhibit greater peak capacity as well as unique selectivity when compared to columns containing C18 stationary phases without a purposely incorporated positive surface charge. The performance of CSH130 C18 columns was also observed to be significantly less dependent on strong ion pairing agents, such as TFA, which suppress electrospray ionization. Furthermore, separations achieved with a CSH130 C18, 1.7 μm column were transferrable to a CSH130 C18, 2.5 μm XP column under lower pressure HPLC conditions, albeit at the cost of analysis time. The availability of different particle size CSH130 C18 columns should facilitate the adaptation of UPLC separations for routine use in laboratories restricted to HPLC instrumentation as well as expand the capabilities of UHPLC instrumentation with 9000 to 12,000 psi pressure limits.
720004568, January 2013