
Analysis of BAC homologues with the ACQUITY UPLC CSH C18 stationary phase offers a rapid, reproducible alternative to existing methods. The UPLC method reduces solvent consumption by 95% and analysis time by 80% relative to currently accepted methods, yielding significant cost savings while enabling high-throughput analyses. Additionally, scaling methods to utilize XSelect CSH C18 XP 2.5 μm Columns results in lower system operating pressures compatible with HPLC, maximizing the number of LC instruments that can be used for these analyses and facilitating the transfer of methods between facilities with combinations of UHPLC and HPLC instrumentation.
Benzalkonium Chloride (BAC) refers to a series of quaternary ammonium chloride homologues with the structure shown in Figure 1. The pervasive use of BAC in consumer products results from its antiseptic and antifungal properties with widespread applications ranging from cleaning products and disinfectants to sanitizing wipes and ophthalmic solutions. Because of its extensive use, BAC has been the subject of numerous studies, including the evaluation of the reactivity of BAC with ocular tissue1,2 and the study of worldwide municipal wastewater, which found BAC to be the most prevalent quaternary ammonium compound in wastewater, with concentrations ranging between 200 and 300 mg/L.3,4
The USP method for the quantitation of BAC utilizes a 10 μm particle size cyano column (L10) for the separation of the BAC homologues.5 The isocratic method uses acetonitrile and 0.1 M sodium acetate (pH 5.0) as mobile phases, resulting in a separation requiring between 15 and 30 minutes. In addition to long analysis times, these separations suffer from reproducibility issues due to the traditionally poor chemical and mechanical stability of the cyano stationary phases.6 Here we present an alternative method employing a Charged Surface Hybrid (CSH) C18 stationary phase under UPLC conditions, resulting in improved peak shapes with significant reductions in both analysis time and solvent consumption. Additionally, we include an example of this method that is transferred to conditions using the XSelect CSH C18 XP 2.5 μm stationary phase (UPLC and HPLC), demonstrating the ability to transfer methods across different instrument platforms.
Standard Solution: Prepared from a USP Reference Standard containing 10% (w/v) of BAC. Diluent was 50:50 acetonitrile/water. Standard solutions were prepared at concentrations of 800, 500, 200, 100, 75, and 50 ppm (μg/mL).
Consumer products were prepared to a final concentration of approximately 80 ppm (0.008% w/v), based on label claim, with 50:50 acetonitrile/water as diluent. Products in liquid form were diluted and analyzed without additional sample preparation or filtering.
|
System: |
ACQUITY UPLC H-Class with PDA Detector |
|
Column: |
ACQUITY UPLC CSH C18, 1.7 μm, 2.1 x 50 mm, part number 186005296 |
|
Mobile phase A: |
100 mM ammonium acetate in water at pH 5.6 (adjusted with glacial acetic acid) |
|
Mobile phase B: |
100% methanol |
|
Mobile phase C: |
200 mM tetrabutyl ammonium hydrogen sulfate [TBAHS] in water for paired-ion chromatography (PIC) |
|
Column temp.: |
35 °C |
|
Isocratic: |
17% A/78% B/5% C (78% MeOH, 17 mM AmOAc, 10 mM TBAHS) |
|
Flow rate: |
0.6 mL/min |
|
UV detection: |
262 nm (40 pts/sec) |
|
Injection volume: |
8 μL |
|
Needle wash: |
50:50 acetonitrile/water |
|
Purge: |
50:50 acetonitrile/water |
|
Seal wash: |
50:50 acetonitrile/water |
Note: After analysis, the chromatographic system and column were flushed with 50:50 acetonitrile/water followed by an additional flush with 100% acetonitrile to prevent any precipitation of the buffer or PIC reagent in the system or column.
Empower chromatography software was used to control the chromatographic instrument, acquire the data, and generate the results. Empower provides the ability to create the necessary calculations such as %BAC which will be described later in this application note.
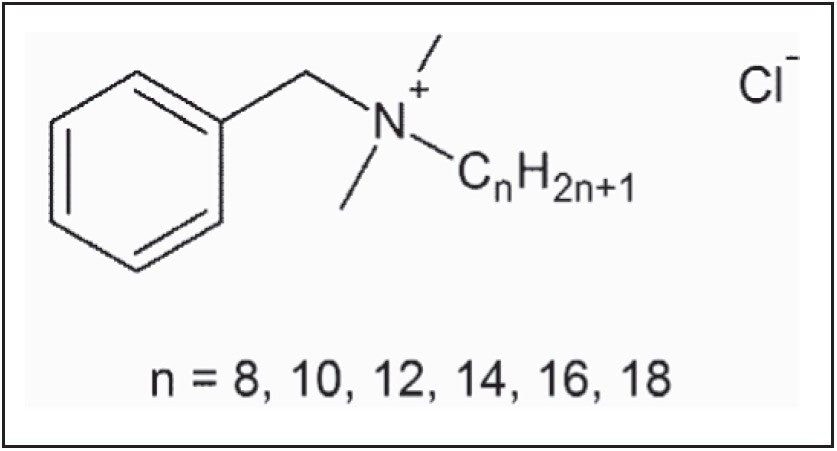
Methods for the analysis of BAC, based on the USP method, rely on cyano ligand bondings on silica base particles. Employing such methods using modern stationary phases based on high purity silica can result in a significant degradation of peak shape, with increased peak tailing and propensity for overloading. This is attributed to the lack of charged impurities in the highly pure silica base particles, resulting in an increase in the undesirable interactions between the charged analyte and the stationary phase. Paired Ion Chromatography [PIC] is a technique for separating charged analytes on reversed-phase columns by exploiting electrostatic interactions between the analyte and the charged PIC reagent. Paired-ion reagents added to the mobile phase are adsorbed onto the stationary phase, where they alter the interaction between the analyte and the stationary phase surface. For oppositely charged analytes, increased stationary phase interactions can result in analyte retention, whereas similarly charged analytes can exhibit decreased interactions and result in faster elution.7 In this application, tetrabutyl ammonium hydrogen sulfate [TBAHS], a low UV absorbing PIC reagent, is used to reduce the unwanted interaction of the charged quaternary ammonium salt with the stationary phase, producing sharper peaks with reduced tailing. Figure 2 demonstrates the separation, based on the USP method, of BAC on a traditional Spherisorb Cyano column and on the high purity silica HSS Cyano Column, with and without the PIC reagent. Increased interactions between the BAC homologues and the high purity silica result in severe peak tailing. The addition of a similarly charged PIC reagent to the mobile phase reduces this interaction resulting in a decrease in retention and a significant improvement in peak shape.
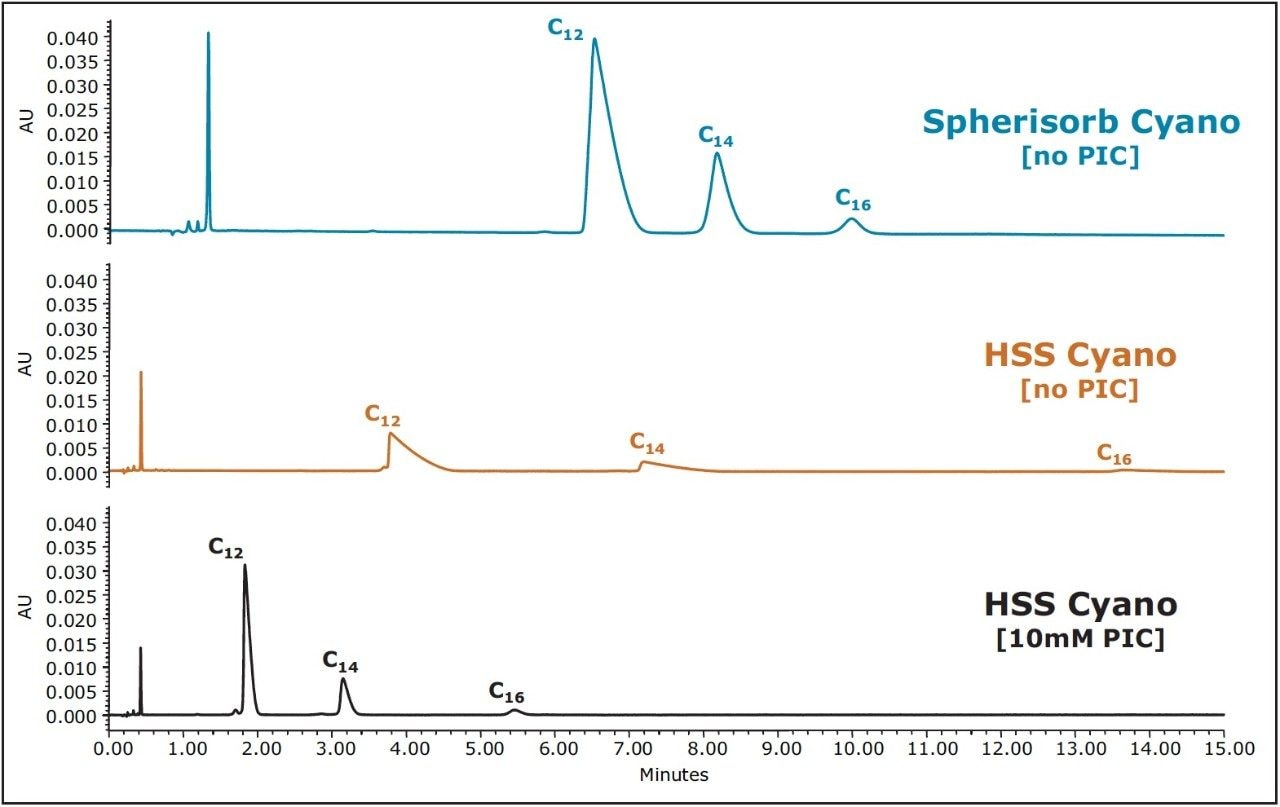
Additional improvements in peak shape and analyte loadability are realized with the use of the Charged Surface Hybrid (CSH) C18 stationary phase, and the replacement of acetonitrile with methanol. Figure 3 demonstrates the improvement in peak shape and loading for the C12 BAC homologue on the CSH C18 Column as a function of analyte concentration and PIC reagent concentration.
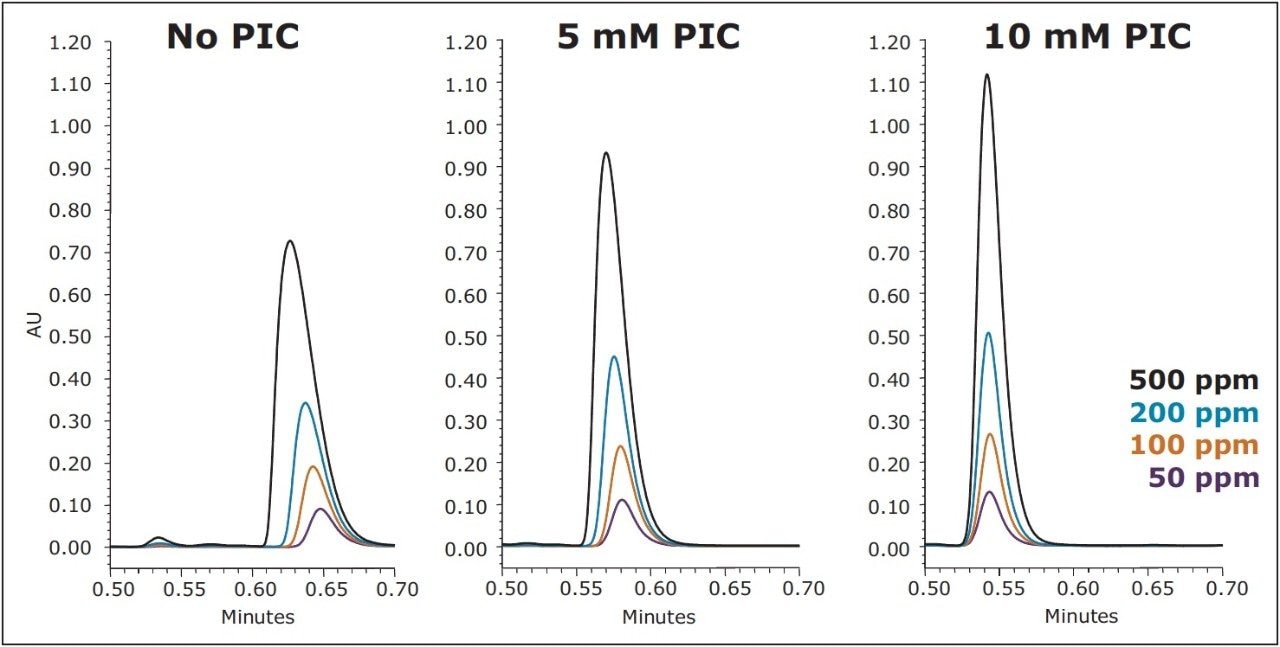
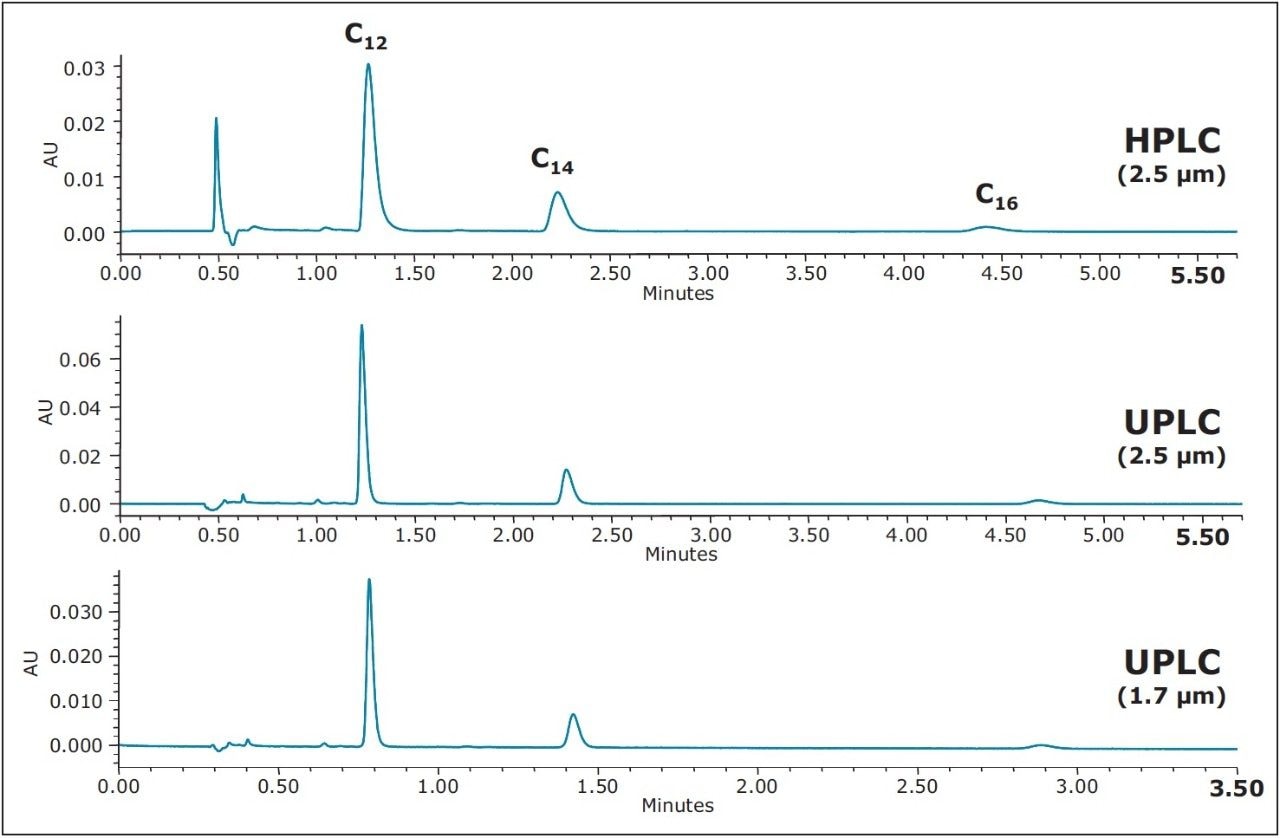
Although there is an improvement in peak shape with the use of a PIC reagent, a decrease in analyte retention is also observed, as demonstrated in Figure 3. A simple adjustment in the organic concentration of the mobile phase, from 80% to 78% methanol, is all that is required to increase the retention factor, while still maintaining the improvement in peak shape. The resulting chromatography, shown in Figure 4 (bottom), under UPLC conditions, gives excellent peak shape and sensitivity for the USP reference standard for BAC, facilitating integration and quantitation. With the aid of the ACQUITY UPLC Columns Calculator, the method was also easily scaled to utilize the XSelect CSH C18 XP, 2.5 μm Column (3.0 x 75 mm) under UPLC (middle) and HPLC (top) conditions. The column dimension was chosen in order to maintain the same length to particle size ratio (L/dp) as for the separation on the 1.7 μm particle size. When scaling methods between different column configurations, maintaining the L/dp ratio, while scaling flow rates and injection volumes accordingly, results in similar chromatography, with different time scales.
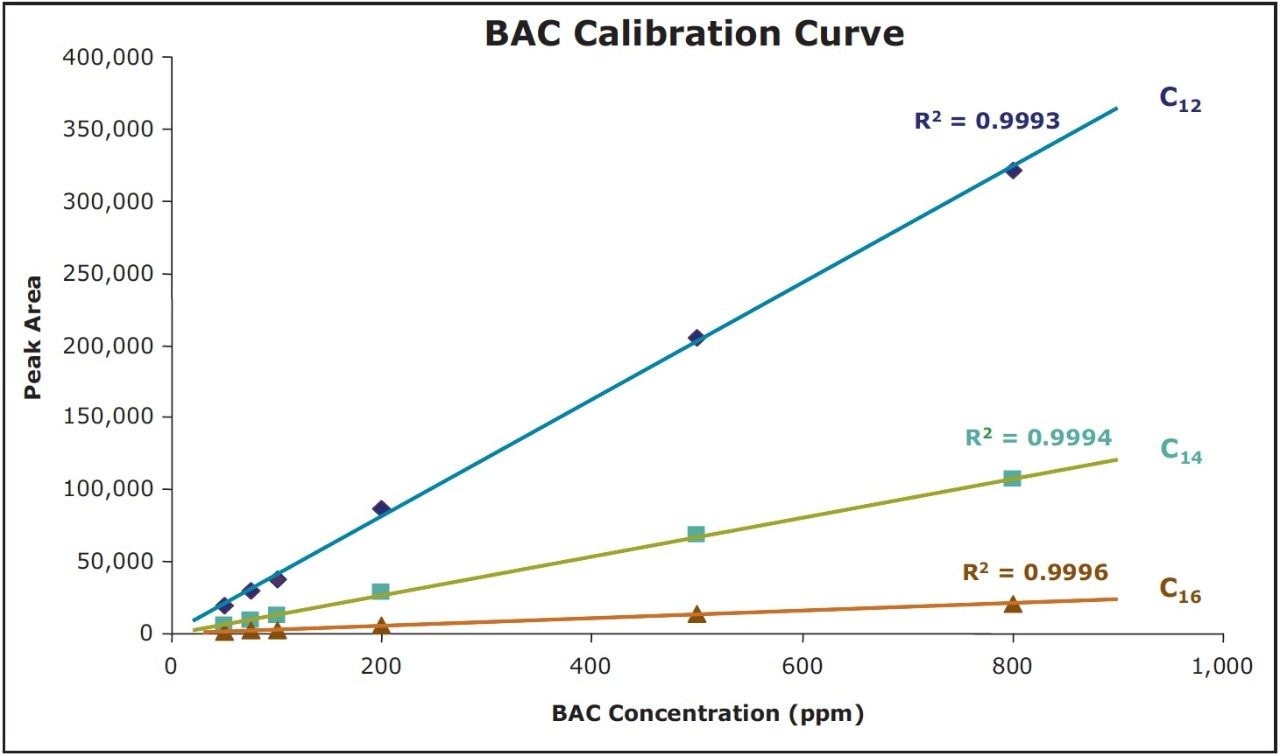
Integration of UPLC chromatograms for the C12, C14, and C16 homologues in the BAC reference standard, prepared at various concentrations from 50 to 800 ppm (μg/mL), shows excellent linearity of detector response versus concentration with R2 values greater than 0.999 (Figure 5).
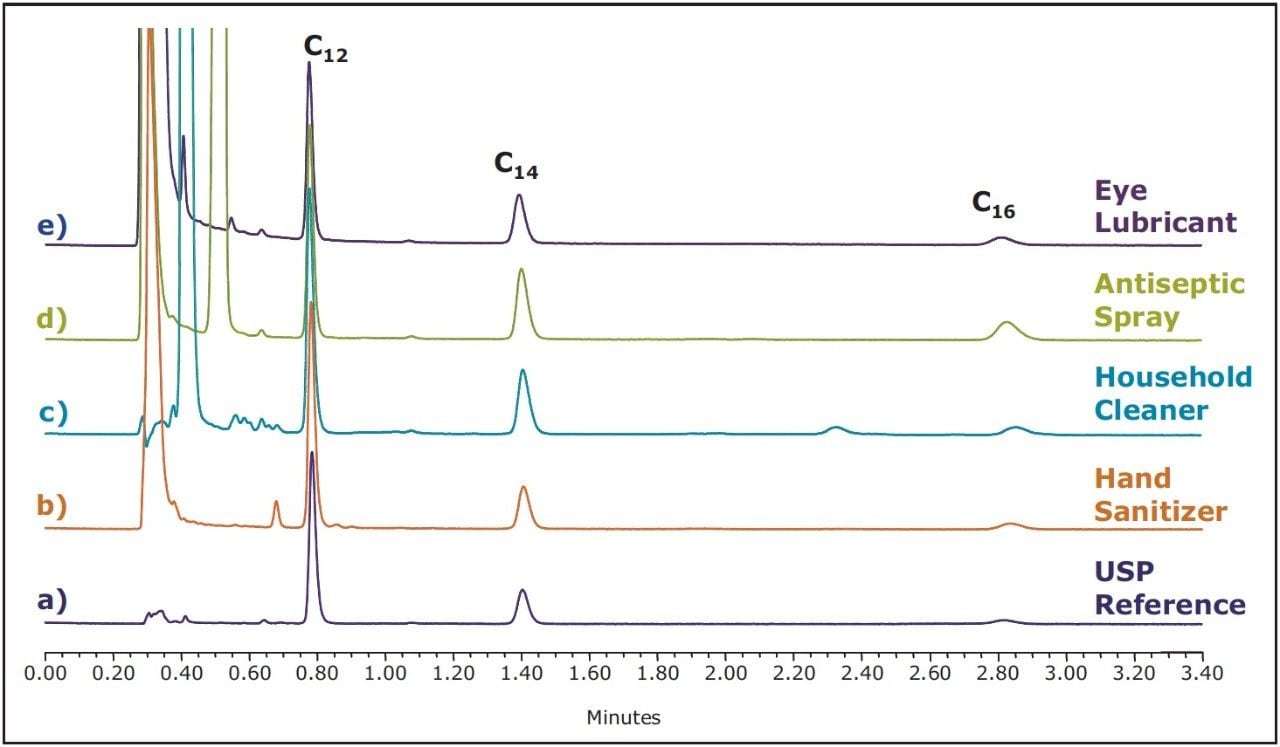
The UPLC method developed using the USP reference standard was applied to a variety of consumer products. Figure 6 shows a small sampling of products tested, each confirming the applicability of this method.
The BAC concentrations in each sample can be calculated by integration of the individual peak areas for the C12, C14, and C16 homologues, and comparing those values with the peak areas from the BAC reference standard using the following equations (results shown in Table 1).

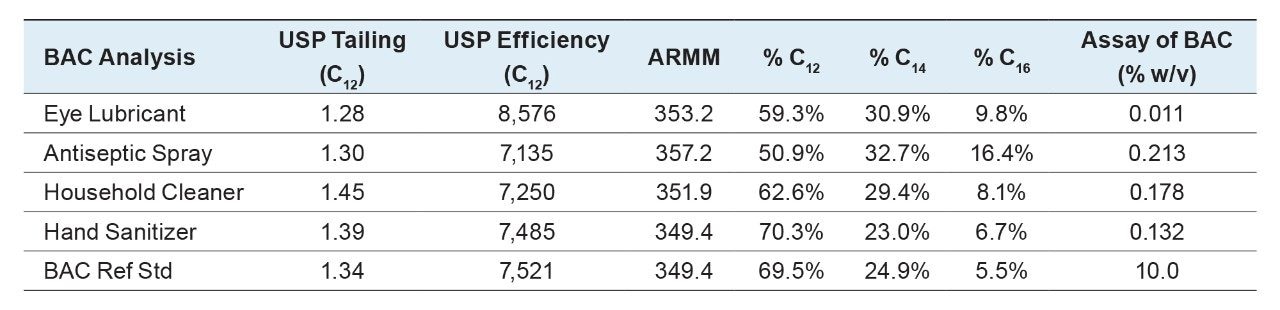
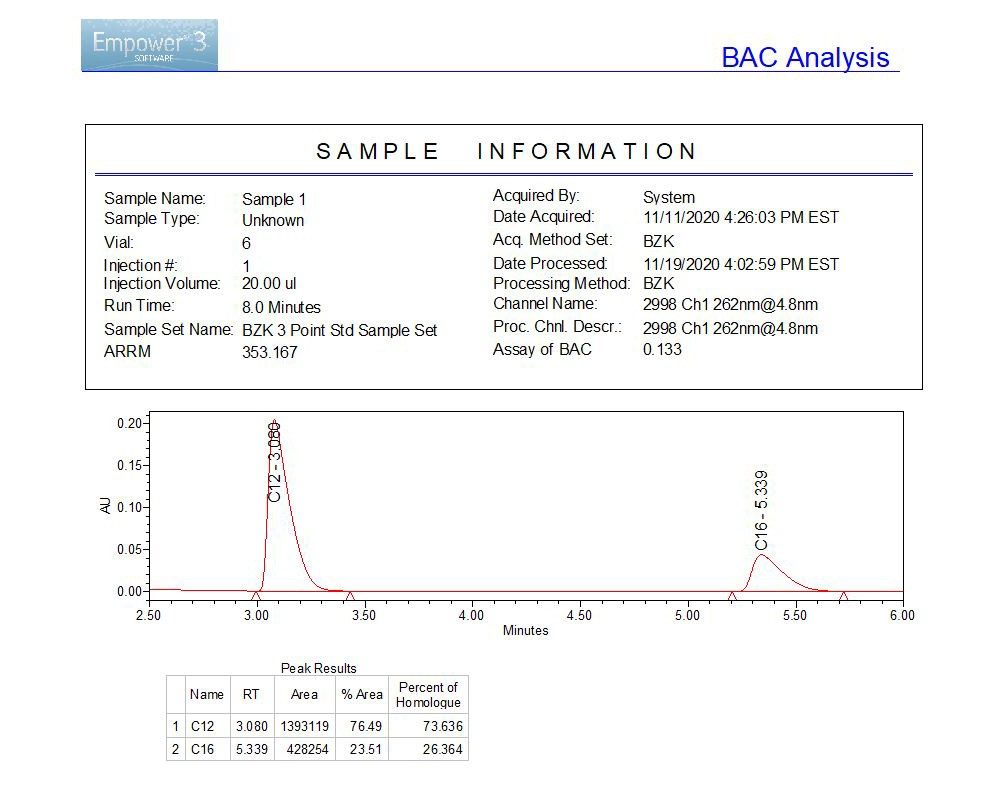
Figure 7 shows an Empower report for a sample which was tested.
Analysis of BAC homologues with the ACQUITY UPLC CSH C18 stationary phase offers a rapid, reproducible alternative to existing methods. The use of a PIC reagent reduces the undesirable interactions between the charged quaternary ammonium analyte and the stationary phase, resulting in significant improvements in peak shape. The improvement in peak shape, in combination with the excellent linearity of response, facilitates quantitation. The UPLC method reduces solvent consumption by 95% and analysis time by 80% relative to currently accepted methods, yielding significant cost savings while enabling high-throughput analyses. Additionally, scaling methods to utilize XSelect CSH C18 XP 2.5 μm Columns results in lower system operating pressures compatible with HPLC, maximizing the number of LC instruments that can be used for these analyses and facilitating the transfer of methods between facilities with combinations of UHPLC and HPLC instrumentation.
720004647, Revised December 2020