This is an Application Brief and does not contain a detailed Experimental section.
This application brief describes the process of probing mAb conformation change caused by amino acid mutation using both Hydrogen Deuterium Exchange Mass Spectrometry (HDX MS) and nano Differential Scanning Calorimetry (DSC) technologies.
HDX MS provides insights in days that may be challenging to achieve in months with other techniques.
Conformational changes in biotherapeutics may impact their safety and efficacy. Therefore it is important to regulators and manufacturers to understand what affects conformational changes. Guidelines also stipulate that manufacturers must produce a well-characterized biotherapeutic product (WCBP) including knowledge of higher order structure (HOS). The analytical tools described here help organizations more effectively discover and develop biotherapeutics in order to maintain their competitiveness.
A human immunoglobulin G (IgG) was developed with a site-specific mutation. When the higher order structures of non-mutated and mutated IgGs were compared using alternative methods, the localized conformational changes at the substitution sites were not easily detected. Two techniques were used in this study: nano Differential Scanning Calorimetry (DSC), and hydrogen deuterium exchange with mass spectrometry. Both techniques contribute to the understanding of the higher order structure of biomolecules and biotherapeutics.
The Nano DSC instrument from TA Instruments can detect changes in heat content down to the nanowatt range from 2 micrograms of protein. The heat signal on a DSC is used as a measure of the folding or unfolding process and can therefore provide insights into the overall structure. DSC is useful in providing rapid and robust measurements with little sample preparation.
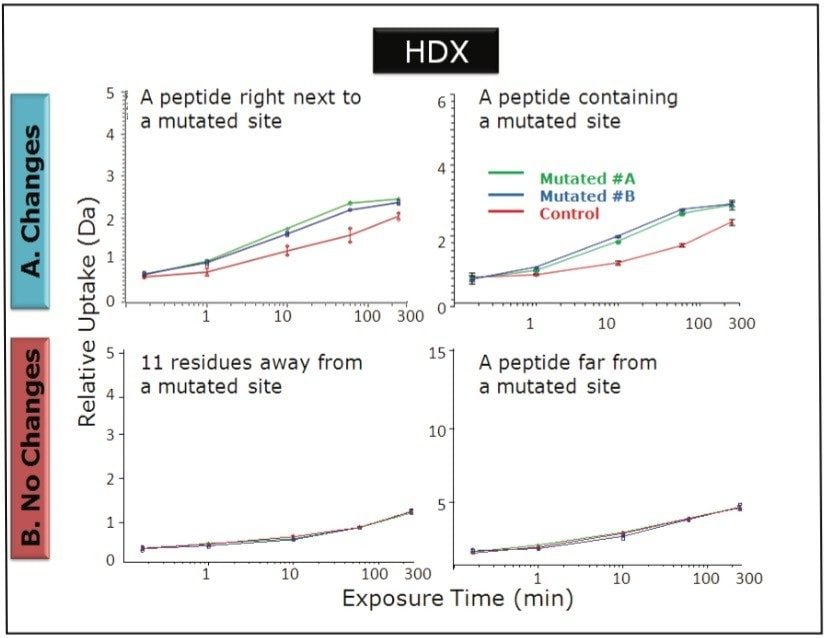
HDX MS can be used to locate the region where the conformational change has taken place. HDX MS experiments provide extremely high detail and leverage the power of UPLC/QTof-MSE to maximize coverage of a protein. Here, a nanoACQUITY UPLC System with HDX Technology was used along with a Xevo G2 QTof MS. The local HDX workflow demonstrates the ability to detect differences in deuterium uptake at the peptide level. The differences in deuterium uptake between different conformations allowed us to locate precisely where the mutated and non-mutated forms differed.
HDX data processing used to be tedious and time-consuming when performed manually. DynamX software was developed by Waters to automate data processing and provide efficiency that had not previously existed. DynamX automatically calculates deuterium uptake. Results are displayed in convenient comparative views: uptake curves, butterfly charts, and difference plots.1 The processing time is thereby reduced from months of specialist interpretation to a day of reporting and curation. We report efficient HDX studies of a control and of two batches of mutated IgG1.


Waters’ tools for high order structure of protein allow the determination of conformational changes for all sectors of a biopharmaceutical company. Benchtop scanning calorimetry provides a robust and rapid measure of the change in folding across different temperatures. Rapid comparisons can be made for different states, to be used in discovery or development. HDX MS provides highly detailed information to locate where conformational changes occur.
HDX MS provides insights in days that may be challenging to achieve in months with other techniques. Both tools contribute to efficiency in an industry that requires robust, rigorous, and rapid information. Using these tools will help a pharmaceutical organization manage their pipeline effectively, satisfy regulators, and protect better against competition.
720004224, February 2012