For research use only. Not for use in diagnostic procedures.
Occasionally, there are instances where a hemoglobin variant peptide ion occurs in a part of the digest spectrum where an interference from another ion makes identification difficult. If variant ions with three or more charges are detected, Electron Transfer Dissociation (ETD) of these ions has the potential to aid in the identification of human Hb variants, and thus provide simple and easy to interpret tandem mass spectra.
Electron Transfer Dissociation (ETD) of these ions has the potential to aid in the identification of human Hb variants.
The global dispersion of hemoglobin variants through population migration has led to the requirement for their identification. An effective mass spectrometry-based procedure involves analysis of the intact globin chains in diluted blood to detect the variant through mass anomalies; this is followed by location of the variant amino acid residue by direct analysis of the enzymatically digested globins without prior chromatographic separation.1 The objective of this part of the procedure is to identify the variant peptide, i.e. the peptide that contains the mutant amino acid, using the mass difference of the variant from the normal peptide obtained in the first step as a guide. Generally, this is straightforward, since there are very few cases of interference between the ions in the mass spectrum. Once observed, the variant peptide can be sequenced by tandem MS in order to positively identify or confirm the mutation.
On occasion, however, the variant peptide ion occurs in a part of the mass spectrum where there is interference from another ion, which makes detection and identification difficult or impossible. Chromatographically separating the tryptic peptides using reversed phase LC-MS would potentially offer a solution. Another solution is to use ion mobility coupled with mass spectrometry (IM-MS) to separate the variant peptide ion from the interfering ion. This would allow the variant peptides to be observed and sequenced by collision induced dissociation following IM.2
In this application note, the doubly-charged tryptic peptide from a variant of low abundance occurred at the same m/z value as a singly-charged interfering ion. Here, we demonstrate fragmentation of the triply-charged tryptic peptide ions using electron transfer dissociation (ETD), (as opposed to collision induced dissociation (CID) of the triply-charged ions) for sequence information. This technique is shown for the identification of an alpha-chain variant having normal clinical presentation, namely Hb Riccarton.
|
MS system: |
SYNAPT G2 |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.5 kV (for positive) |
|
Cone voltage: |
30 V |
|
Desolvation temp.: |
200 ˚C |
|
Source temp.: |
120 ˚C |
|
Ionization mode: |
Glow discharge negative |
|
Glow discharge: |
55 μA (for negative) |
|
Reagent: |
para-nitrotoluene (m/z 137). |
A blood sample was submitted for investigation by mass spectrometry because an abnormality had been detected during a routine screen in a hematology laboratory. For mass spectral analysis, the sample was prepared and analyzed as described previously.1 The sample was diluted 50-fold with water, denatured, and digested with trypsin for 30 minutes. The resulting mixture of peptides was diluted a further 10-fold (final solution in 50.0% aqueous acetonitrile containing 0.2% formic acid) for direct infusion into the ESI source of the mass spectrometer.
ESI-MS was performed on a SYNAPT G2 hybrid quadrupole / ion mobility /oa-ToF mass spectrometer fitted with ETD functionality. In brief, the instrument comprises three consecutive, gas filled, travelling wave (T-Wave) RF stacked ring ion guides prior to the ToF mass analyzer. For ETD type fragmentation, a sub-ambient pressure (~2 mbar) glow discharge anion source was used to fill the Trap T-Wave cell with quadrupole mass selected ETD reagent anions formed from para-nitrotoluene (m/z 137). During acquisition, the source polarity and quadrupole set mass were switched to allow triply-charged peptides formed from ESI to interact with stored reagent anions in the Trap T-Wave. For efficient ETD within the Trap T-Wave region, the helium bath gas was set to a pressure of 5 x 10-2 mbar. The Transfer T-Wave cell was pressurized to 5 x 10-3 mbar with argon. The T-Wave speed and amplitude were set to 300 m/sec and 0.2 V respectively. Solutions were infused at a flow rate of 5 µL/min.
A low abundance hemoglobin variant was detected during a routine hospital screening by cation exchange-HPLC and it was submitted for identification by MS. Analysis of the intact chains showed that the variant was 30 Da heavier than the normal α-chain. There are five mutations that, on genetic grounds, are likely to increase the mass by 30 Da; either Ala→Thr, Arg→Trp, Gly→Ser, Thr→Met, or Val→Glu.
Figure 1 shows the analysis of the 30-minute digests of a normal control (top spectrum) and the sample containing the variant (bottom spectrum). The analysis revealed that the αT62+ ion at m/z 932.5 in the variant spectrum had an interfering ion (normal βT2+ ion) at the expected m/z of the variant αT62+ ion. This peak, m/z 15 higher than the αT62+ ion, suggested two possible mutations, either α51Gly→Ser or α53Ala→Thr, as shown in Figure 1.
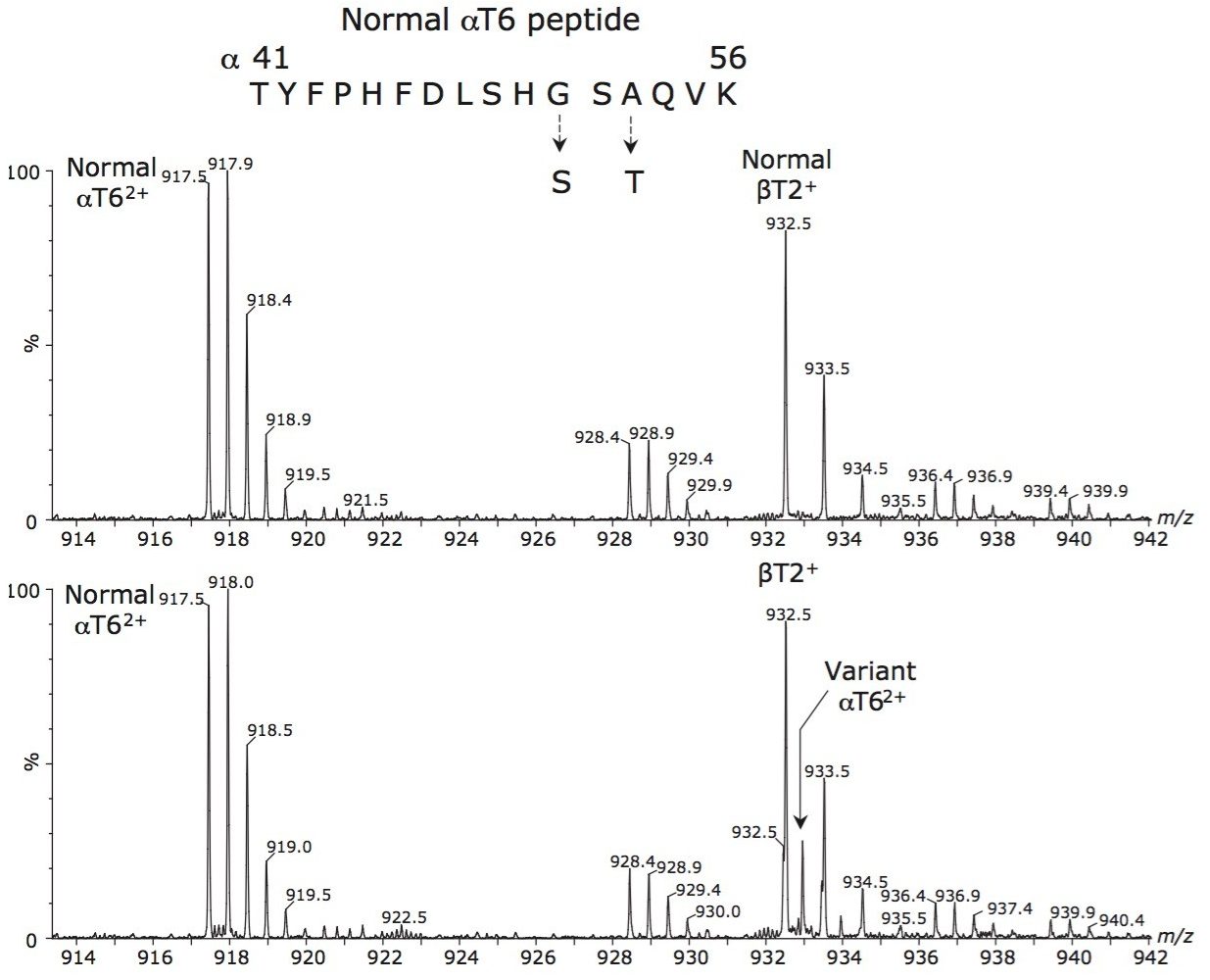
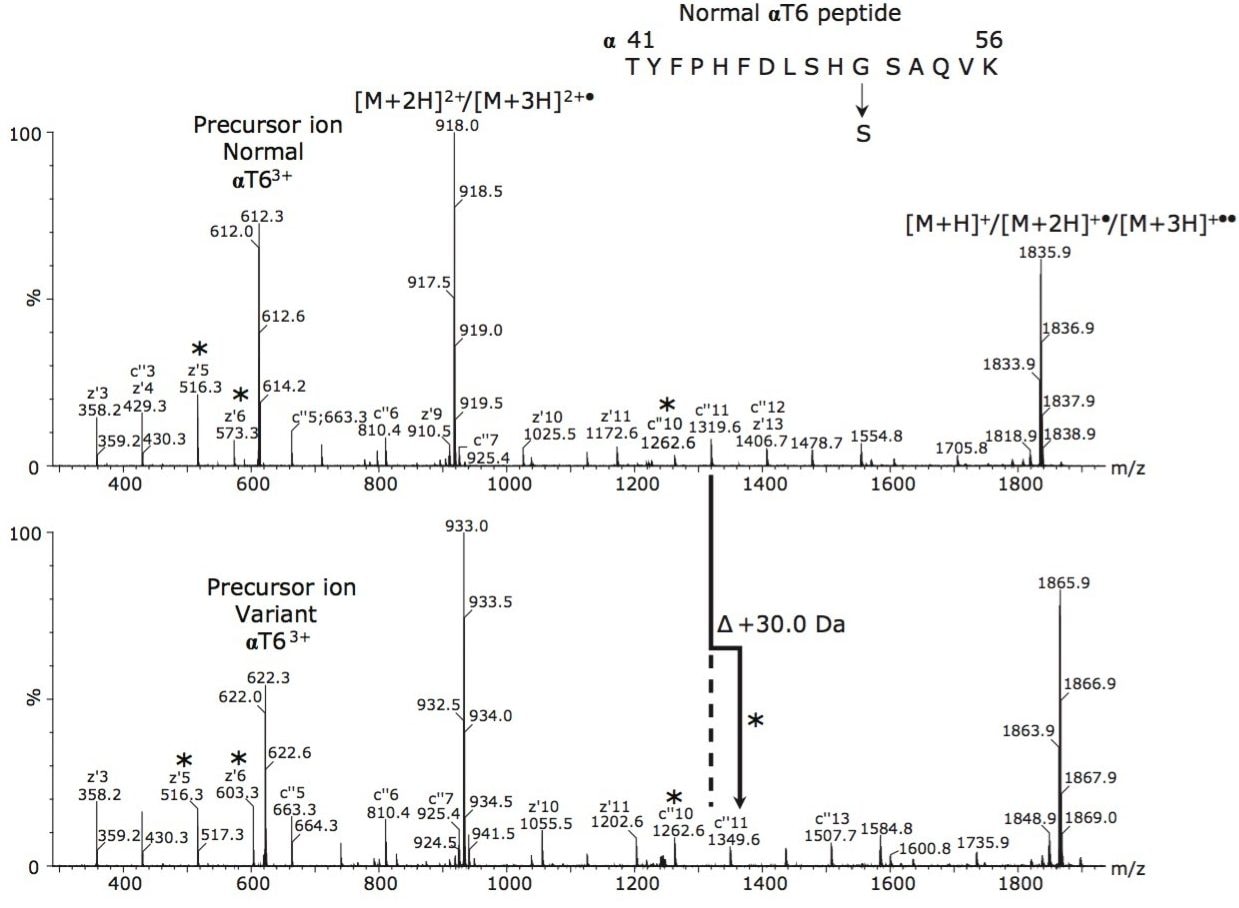
As a consequence of the interference from the intense singly-charged ion, detection and subsequent identification of the variant through MS/MS of the doubly-charged ion would have been difficult. In order to distinguish these two possibilities, the normal and variant αT63+ ions were sequenced by ETD since there were no interferences observed for these ions. Accurate mass alone would not be sufficient to determine if the mutation was Gly→Ser or Ala→Thr from the digest mass spectrum since the accurate mass difference between these two possibilities is 30.0106 Da in both instances; hence, the need for tandem mass spectrometry.
Figure 2 (top) shows the ETD mass spectrum of the peak at m/z 612, which is consistent with the sequence for the normal αT63+ ion. Figure 2 (bottom) shows the ETD mass spectrum for the peak at m/z 622 for the putative variant αT63+ ion. The 30 Da mass-increase between the normal and variant at z'6 and c"11 and all subsequent z' and c" ions identifies the mutation as α51Gly→Ser, Hb Riccarton.
720004291, April 2012