This is an Application Brief and does not contain a detailed Experimental section.
This application brief details successful convertion of the compendial normal phase HPLC method for the assay of the drug substance tolbutamide to a supercritical fluid chromatography method using the Waters ACQUITY UPC2 System.
This new UltraPerformance Convergence Chromatography (UPC2) method produced data of equal or better quality than the current HPLC method, was 10 times faster, and consumed less solvent.
Supercritical fluid chromatography (SFC) is a normal phase separation technique that uses carbon dioxide as the main mobile phase and often employs the use of polar modifiers such as methanol. Since the principles of SFC are similar to those of HPLC, methods should be able to be converted to SFC reducing solvent usage and disposal which will lower cost per analysis while enhancing green initiatives. Methods converted to an SFC solution must maintain data quality and must produce results that are equivalent to the current normal phase method. Currently, the United States Pharmacopeia (USP) specifies a normal phase HPLC method for the assay of the drug substance tolbutamide (benzenesulfonamide, CAS# 64-77-7). This isocratic separation is done at 1.5 mL/min using a 4.0 x 300 mm, silica column (L3) with a mobile phase that consists of 475:475:20:15:9 hexane, water-saturated-hexane, tetrahydrofuran, alcohol, and glacial acetic acid with a run time of approximately 20 minutes. Like most compendial methods, this method is proven and reliable. It does, however, use a complicated mobile phase mixture that contains hexane and THF, solvents that many labs would like to eliminate in favor of green initiatives and reduced costs.
Samples were prepared and analyzed using the current USP method and contained tolbutamide along with the internal standard tolazamide. The results of this analysis were used to compare the results obtained with the method developed on an ACQUITY UPC2 System. The UPC2 method conditions were as follows:
|
Column: |
ACQUITY UPC2 BEH, 3.0 x 100 mm, 1.7 μm |
|
Temperature: |
50 °C |
|
Mobile phase: |
95% Carbon dioxide: 5% methanol/IPA (1:1) containing 0.2% TFA |
|
Flow rate: |
2.5 mL/min |
|
Back pressure: |
120 Bar/1740 psi |
|
Detection: |
UV /PDA at 254 nm |
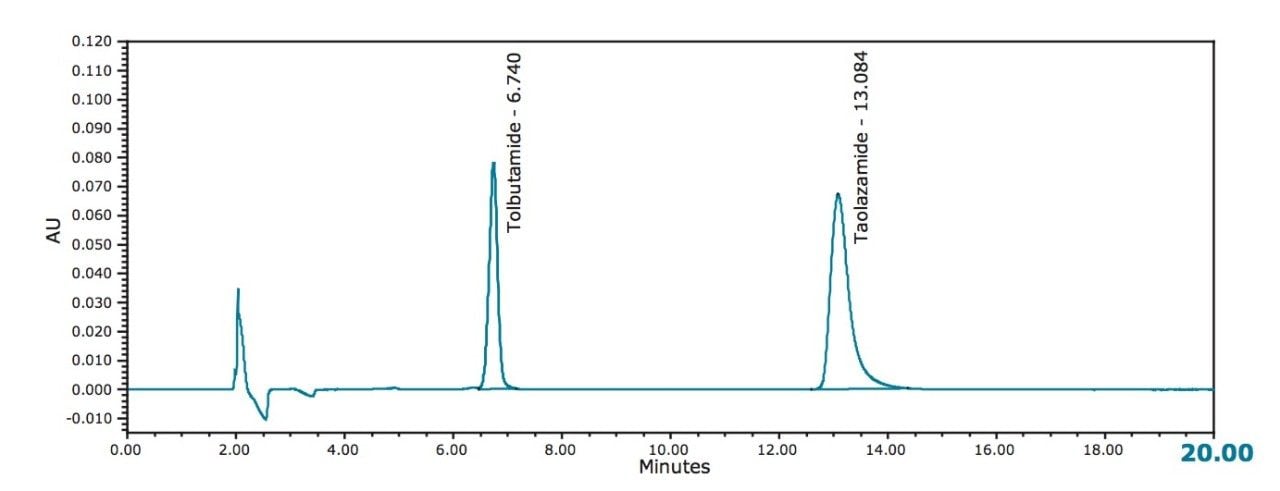
The current normal phase HPLC method shown in Figure 1 generated acceptable chromatography, although it was noted that there was significant peak tailing of the internal standard peak (tailing factor of 1.65). No effort was made to improve the method as it passed the listed suitability criteria (the relative standard deviation for replicate injections is not more than 2.0%, and the resolution, R, between tolbutamide and tolazamide is not less than 2.0).
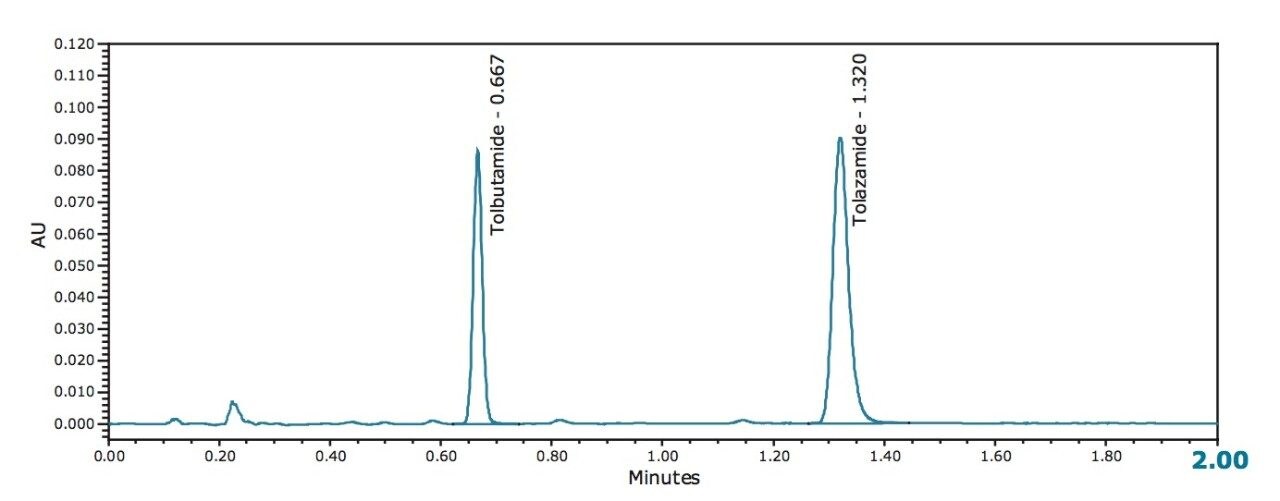
Results from the newly developed UPC2 method also met required USP suitability values (1.2% and 0.9% retention time RSD for tolbutamide and tolazamide respectively and <0.90% RSD for area for both compounds, n=6) and had a significantly faster run time (2.0 min) while maintaining a similar resolution between the 2 compounds of interest (R = ~15). Tailing of the internal standard, tolazamide, was significantly improved (tailing factor of 1.2). It is also interesting to note that a number of small peaks were resolved and detected from the mixture using UPC2, demonstrating the high separation efficiency of this technique. In this example, each normal phase HPLC run used approximately 29 mL of hexane and less than one mL each of THF and ethanol. In contrast, each injection with the UPC2 method used roughly 0.25 mL each of methanol and IPA. This shows the significant reduction in organic solvent use that can be achieved by moving normal phase methods to UPC2. Based on current solvent prices, each normal phase HPLC run costs roughly $1.40 per run compared to $0.01 for each UPC2 run indicating the significant cost savings that can be achieved by moving traditional normal phase HPLC methods to UPC2.
A USP compendial HPLC method was successfully converted to a UPC2 method, using the ACQUITY UPC2 System. This new UPC2 method produced data of equal or better quality than the current HPLC method, was 10 times faster, and consumed less solvent. When high quality results are produced faster, laboratory productivity increases and cost per sample decreases. The ACQUITY UPC2 System is an ideal solution for laboratories wanting to convert their current normal phase HPLC methods to more efficient and cost effective methods while enhancing health, safety, and environmental concerns.
720004247, February 2012