
This work demonstrates the capability of Xevo TQ-S for monitoring compounds with different parent masses arising out of isotopic distribution. Oasis Ion-Exchange cartridges, UPLC chromatography, and an advanced tandem quadrupole mass spectrometer for the development of a picogram-sensitivity method with a high degree of specificity for the quantification of bromocriptine in plasma were employed. In addition to addressing one of the key challenges of achieving desired sensitivity, which is often faced by the bioanalytical scientist, this application note addresses other challenges such as reproducibility and robustness.
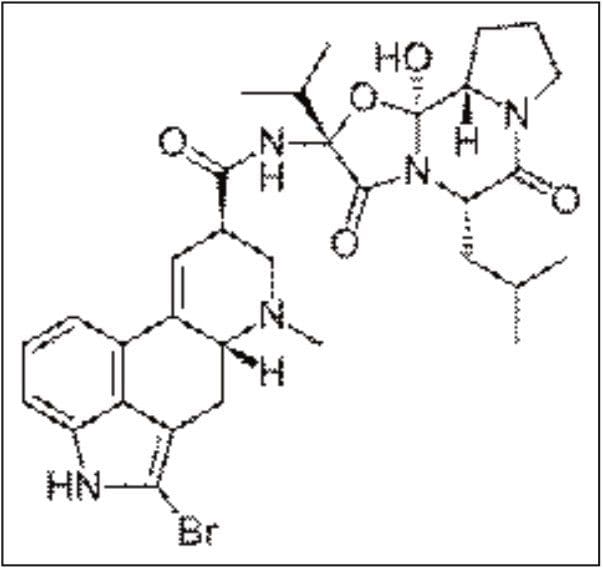
Bromocriptine, 2-bromoergocriptine, is a semi-synthetic derivative of a natural ergot alkaloid, ergocriptin (a derivative of lysergic acid), that is synthesized by bromination of ergocriptin using N-bromosuccinimide. Bromocriptine is a dopamine agonist that is used in the treatment of pituitary tumors, Parkinson’s disease (PD), hyperprolactinaemia, neuroleptic malignant syndrome, and type II diabetes. Bromocriptine is classified as an ergopeptide, comprising ergoline derivatives that contain a tripeptide structure attached to the basic ergoline ring in the same location as that of the amide group of the lysergic acid derivatives. Typically, the ergopeptides comprise two other α-amino acids linked in an unusual cyclol formation >N-C(OH)< with the carboxyl carbon of proline, at the juncture between the two lactam rings.
Bromocriptine has been available for clinical use for many years and has vasoconstrictive properties similar to ergotamine and ergonovine, due to the unsaturated bond in the D ring of the lysergide ring system. This contrasts with the vasodilating properties of ergoline-based compounds, such as pergolide and dihydroergotoxine alkaloids, which have a saturated bond in the same position in the D ring. This class of compounds, including Bromocriptine exhibits high lipophilicity, and is also extensively metabolized prior to excretion. Traditional screening methods, such as enzyme linked immunosorbent assays (ELISA) and gas chromatography-mass spectrometry (GC-MS) lack the sensitivity to screen blood or plasma at the very low concentrations that Bromocriptine is likely to be found following intake. Therefore, a sensitive and specific liquid chromatographic-mass spectrometric (LC-MS) method is needed.
|
LC system: |
ACQUITY UPLC System |
|
Column: |
ACQUITY UPLC HSS C18 1.8 μm, 2.1 x 150 mm |
|
LC Column elution: |
70% aqueous buffer over 2.0 minutes, followed by a 95% organic elution till 4.2 min; then change back to initial conditions. |
|
Column temp.: |
40 °C |
|
Flow rate: |
0.200 mL/min |
|
Injection volume: |
20 μL |
|
MS system: |
Xevo TQ-S |
|
MS mode: |
ESI positive MS/MS method |
|
MRM transition: |
656.27 → 348.06 |
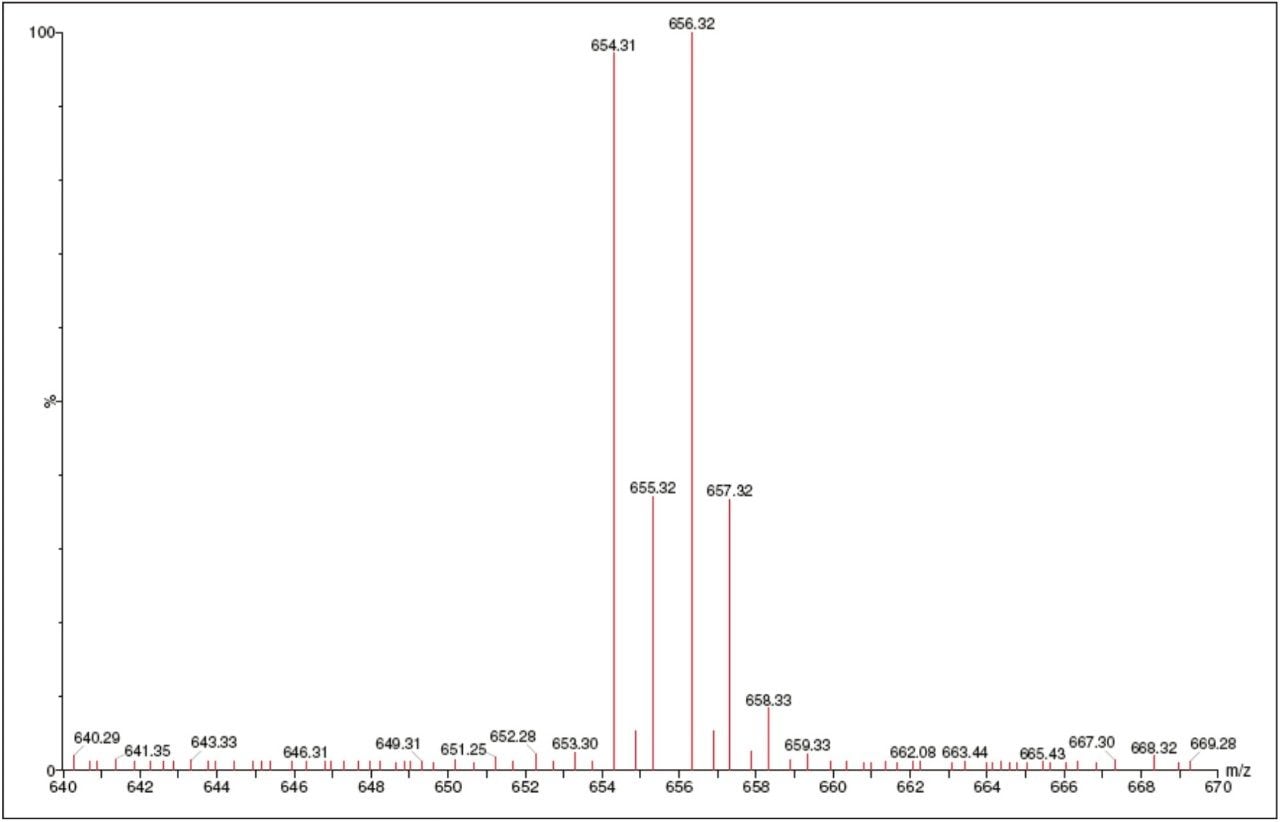
The selectivity of the isotope in Bromocriptine due to isotopic abundance is essential since the compound exhibits two different peaks of parent masses – a feature typical of molecules containing bromine. Both parent masses were monitored for method development. This molecule also exhibits strong protein binding ability, which is an essential requirement for the estimation in the pg/mL levels.
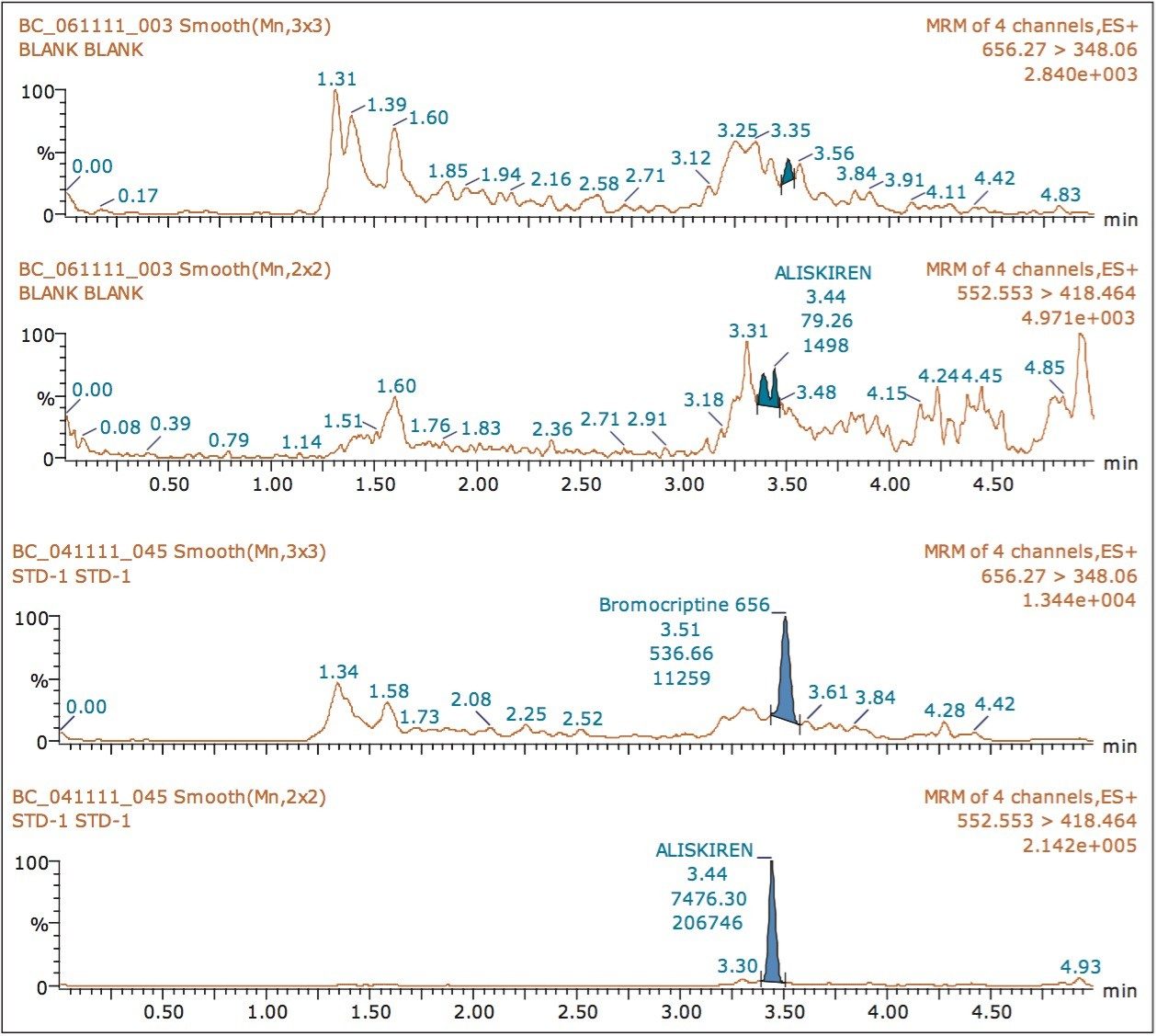
Samples were isolated using a solid phase extraction (SPE) method, employing Waters Oasis MCX (1 CC, 30 mg cartridges). A 500 μL aliquot of plasma was diluted with 5% o-Phosphoric Acid, and loaded onto the SPE cartridges that were previously conditioned with organic solvent and water. The plasma solution was then washed with acidified water followed by an organic wash, and then eluted with basic organic elution solvent. The eluted samples were evaporated and reconstituted prior to injection. Aliskiren (MW 551.76; retention time, 3.44 mins), typically employed as a renin inhibitor, was used as an internal standard (IS).
The chromatographic method using ACQUITY UPLC and the ACQUITY UPLC HSS C18 Column provided excellent resolution for the Bromocriptine analyte. Bromocriptine eluted at 3.51 minutes with a peak width of 12 s at the base. The data shown in Figure 2 illustrate the signal obtained from the lower limit of quantification (LLOQ) of Bromocriptine in human plasma, and the blank signal, shown in Figure 3. Analysis of aliskiren exhibited chromatographic and extraction conditions similar to those observed for Bromocriptine; therefore, aliskiren was selected as an internal standard (IS).
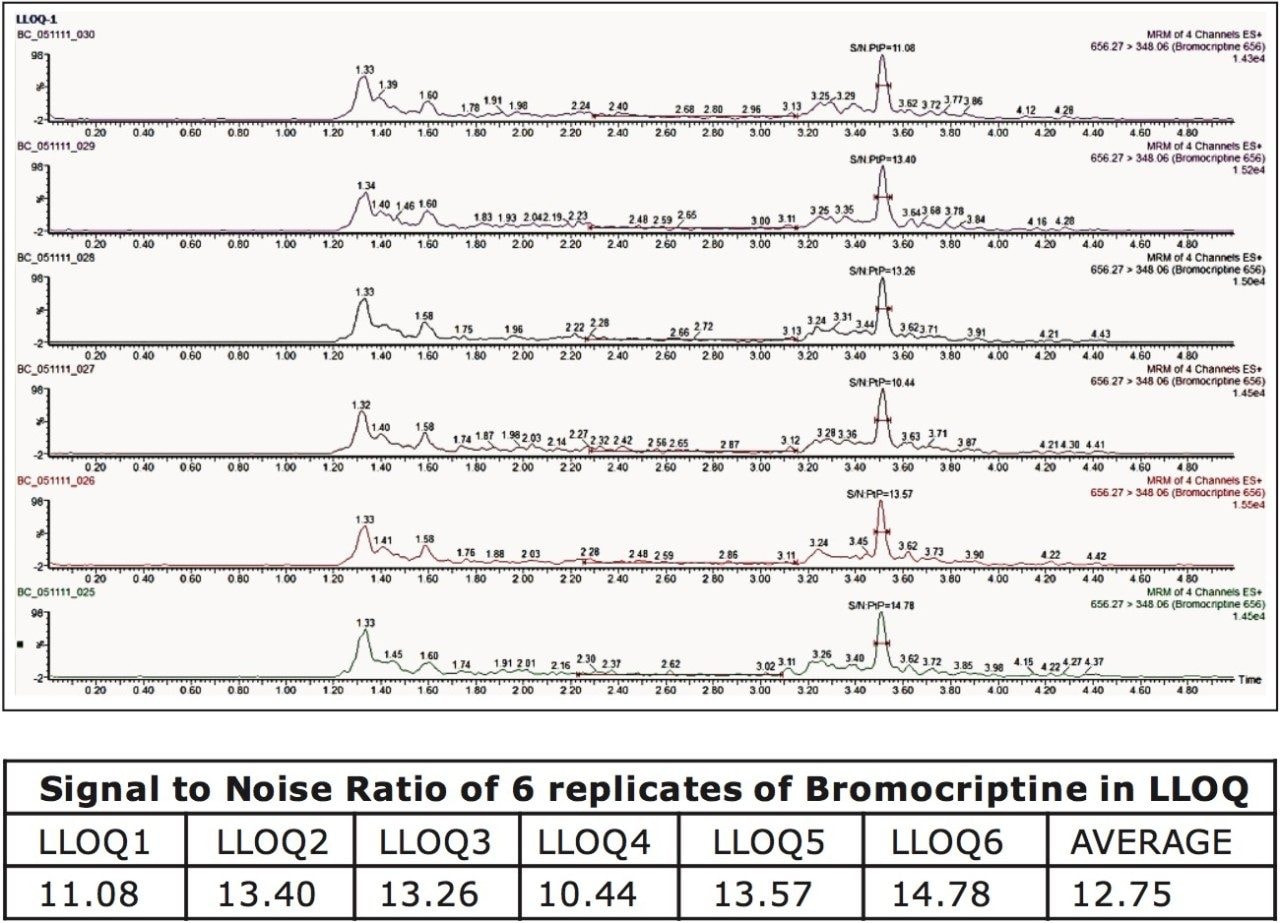
As can be observed in Figure 2, the retention time of Bromocriptine does not interfere with any endogenous signals, and the signal corresponding to the analyte of interest can be easily observed at the LLOQ level. The Xevo TQ-S MS is equipped with a novel StepWave ion guide, which when combined with the high-resolution chromatography obtained from the ACQUITY UPLC System, results in the successful completion of extremely sensitive applications that can be performed with high reproducibility. Bromocriptine was detected at a concentration of 2.0 pg/mL, shown in Figure 2, with a signal-to-noise ratio of 12.75:1.00 for an average of six replicate injections.
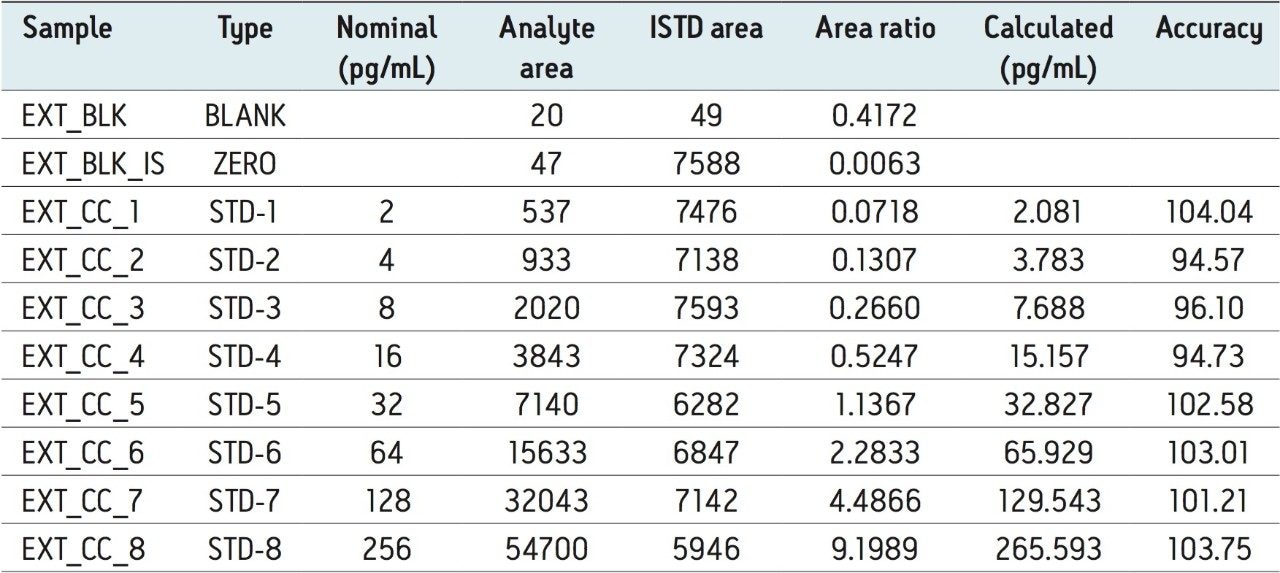
The assay in this report showed linear calibration over the range of 2 to 256 pg/mL with an excellent r2 value of 0.997, as shown in Table 1. The back-calculated concentration of the standard was found to be within 6% of the nominal concentration, as shown in Table 1. As observed from the data in Table 1, an excellent degree of accuracy was achieved for each sample. Although both parent masses, showed acceptable results owing to higher specificity and the chromatographic separation, shown in Figure 4, the parent mass of 656 was chosen for the estimation of Bromocriptine in matrix. This assay was performed with a 5-minute injection-to-injection time scale, highlighting the capability of Waters Regulated Bioanalysis System solution to deliver fast, accurate, and highly sensitive results while maintaining desired precision, and high throughput value.
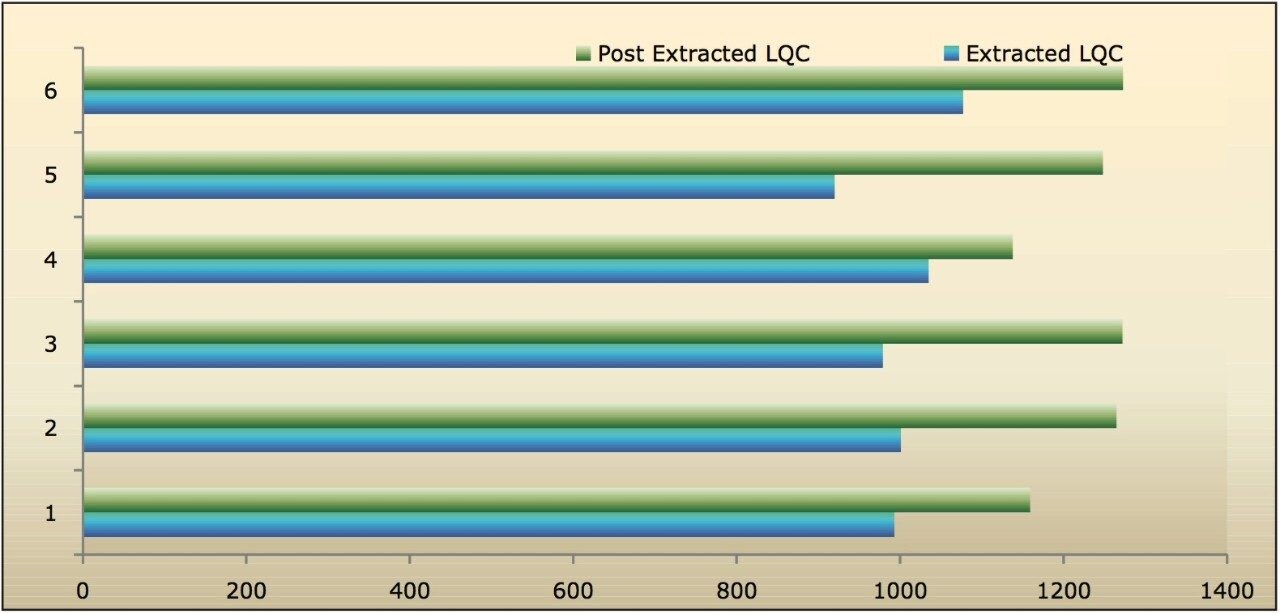
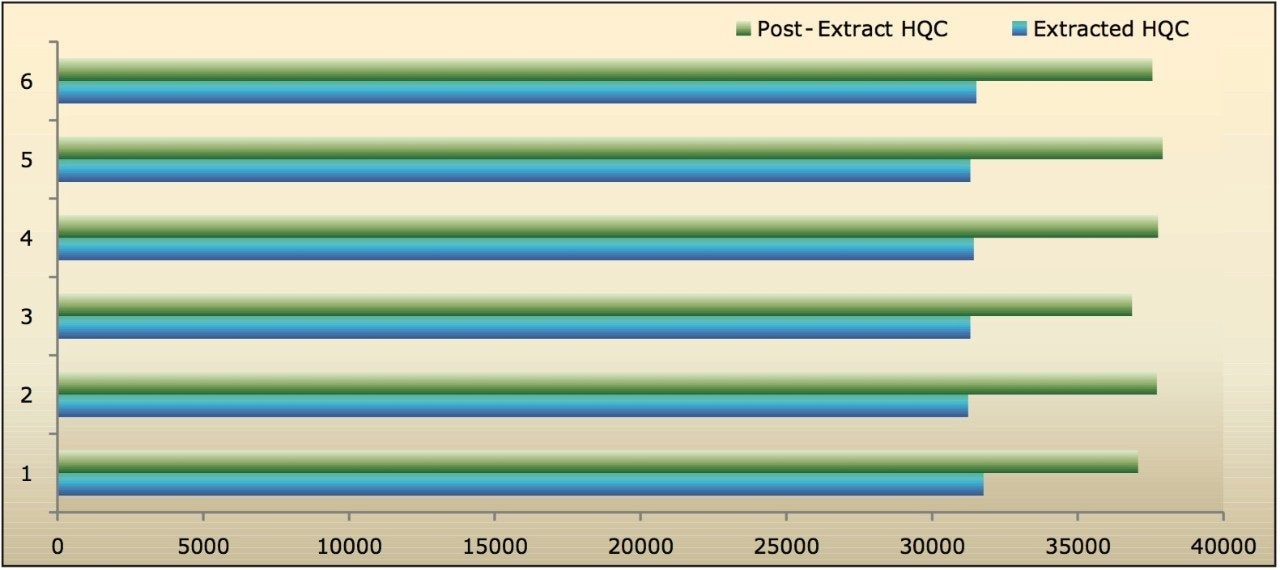
Recovery of the analyte and internal standard (IS) were performed by comparing the extracted QC samples against six post-extracted samples which were found to be approximately 84% at LQC, MQC, and HQC levels for both analyte and IS, as shown in Figure 4 and Table 2. The %CV for repeat batches were found to be within 10% of LLOQQC and varied between 1% to 3% for all QC levels.
The data in Figures 4.1, 4.2, and 4.3 exhibit analyte recovery values for the six samples in all three concentration levels (LQC, MQC, and HQC). As observed from Figures 4.1, 4.2, and 4.3, the recovery values for Bromocriptine did not vary significantly between the three concentration levels. In addition, the mean analyte recovery for all three concentration ranges was well within acceptable limits, as provided in Table 2.


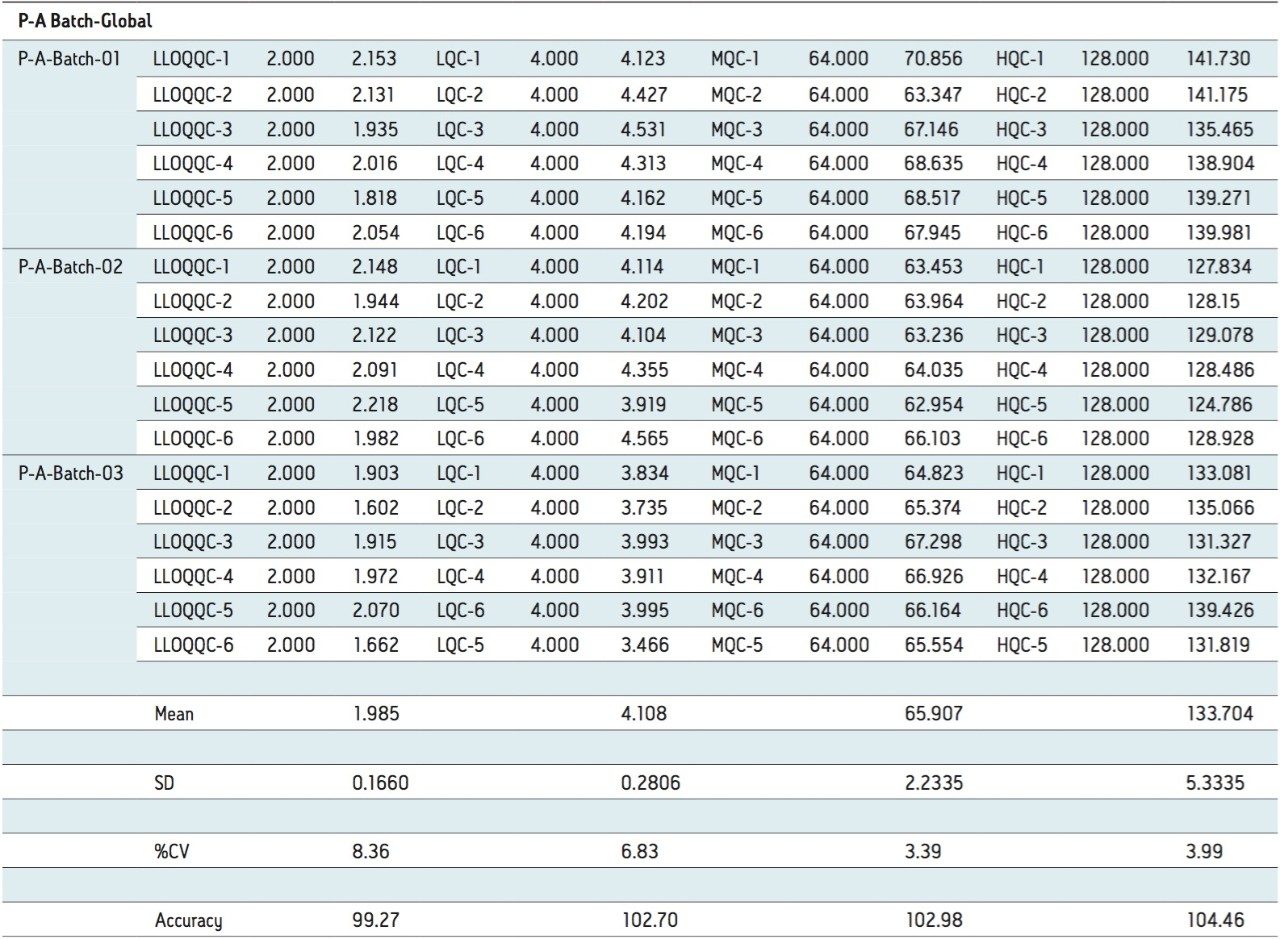
For a comparison of samples within the global batches, three separate batches were prepared in three different concentration levels with six samples in each batch. These batches were categorized as LLOQQC (Lower Limit of Quantification Quality Control), LQC, MQC, and HQC. The data showed excellent agreement between the six samples for all three batches, as shown in Table 3. The mean accuracy obtained for all the samples was found to be >94% for every concentration, shown in Table 3.
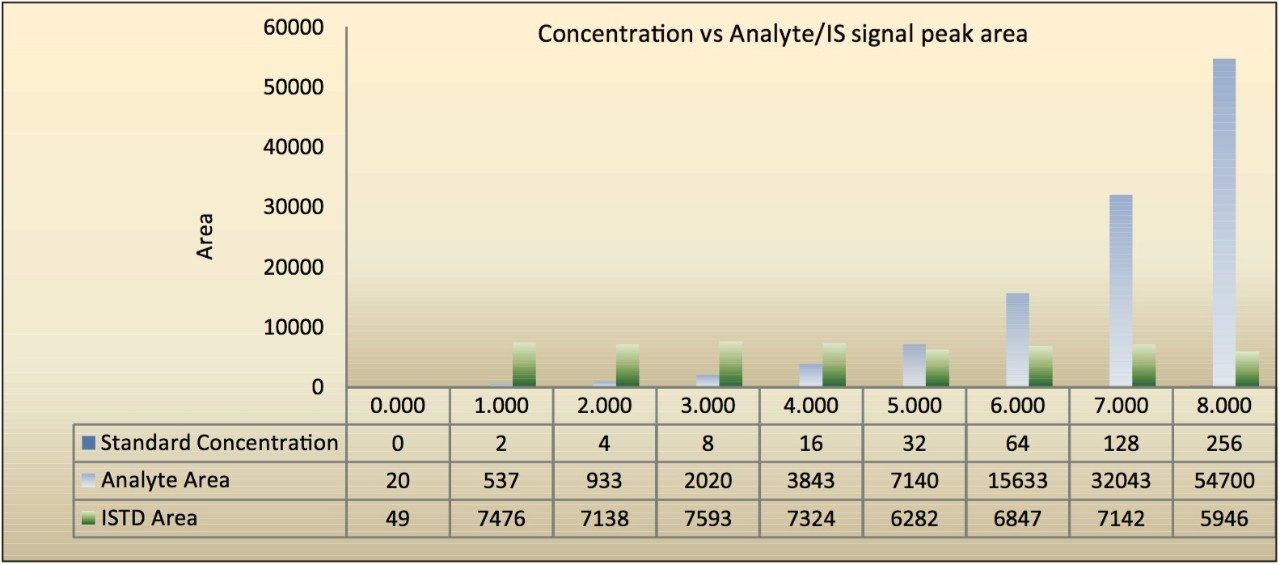
LC-MS analysis of Bromocriptine was highly challenging due to the isotopic distribution for parent masses. It was a challenge to identify and quantify Bromocriptine with such distribution pattern for isotopic abundance and specificity issues. The combination of all the components of Waters Regulated Bioanalysis System solution (best-in-class chemistry solutions for sample preparation and column chemistries, UPLC Technology, and Xevo TQ-S) was able to achieve a highly sensitive, specific, and robust LC-MS method for Bromocriptine.
720004402, June 2012