
The aim of this study is to address some of the analytical challenges previously described when analyzing a wide range of steroid esters (boldenone, nandrolone, estradiol, and testosterone) in cattle hair.
The safety of our food supply can no longer be taken for granted. As the world changes and populations continue to grow, so will the responsibility of organizations to meet the demand of safe food supplies.
One route of human exposure to veterinary substances is through the food chain as a result of malpractice or illegal activities. The steroid esters are one group of substances causing concern, which might still be used as growth promoting agents, but are now forbidden from use in breeding animals in the European Union.1 No residues of these anabolic substances should be present in animal products.2
Many biological matrices, such as tissue or urine samples can be used for the control of steroid esters, but hair may be considered one of the most sensible matrices, due to its ease of collection and the ability to detect residues for a long time after administration of some substances.3-6 In urine, it is hard to differentiate between the metabolites of ever-present endogenous natural steroids, and the identical metabolites of natural steroids from the hydrolyzed esters.7 Furthermore, natural steroids are usually administered as synthetic steroid esters that are rapidly hydrolyzed in vivo into natural steroids. Nevertheless, the identification of the synthetic ester form in hair provides an interesting, efficient, and additional confirmatory method to overcome any inconclusive urine analyses. Indeed, the ability to detect and identify the compound itself for a long time after administration is a strong argument in delivering a confident analytical result. The main problem with this approach is that the concentrations of the steroid esters are extremely low (low ppb range); and the sample size limited (for practical reasons of sample collection, e.g. 100 mg hair).
In the literature, either GC-MS/MS5 or LC-MS/MS7-9 could provide the detection of steroids esters to relevant levels. The less volatile compounds, such as benzoate or decanoate steroids, do not give expected results in GC-MS/MS in routine analysis, because the instrument has to be under very clean and optimized conditions to reach the validation performance.5 Furthermore, the analysis resulted in a 40 minute experiment.5 Given these reasons and the wish for an additional analytical technique for confirmation, we investigated the analysis of steroid esters in cattle hair by LC-MS/MS.
All steroid esters reference compounds were provided by Steraloids Inc. Ltd. (London, England).
Sample extraction and purification procedures applied for steroid esters in hair have been described elsewhere.5,9
|
System: |
ACQUITY UPLC |
|
Run time: |
12.00 min |
|
Column: |
ACQUITY UPLC BEH C18, 2.1 x 100 mm x 1.7 μm |
|
Column temp.: |
50 °C |
|
Mobile phase: |
A: 0.1% formic acid B: CH3CN + 0.1% formic acid |
|
Flow rate: |
0.6 mL/min |
|
Injection volume: |
3 μL |
|
System: |
Xevo TQ MS |
|
Polarity: |
ESI+ |
|
Capillary voltage: |
3.50 kV |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
450 °C |
|
Desolvation gas flow: |
1000 L/Hr |
|
Collision gas flow: |
0.15 mL/min |
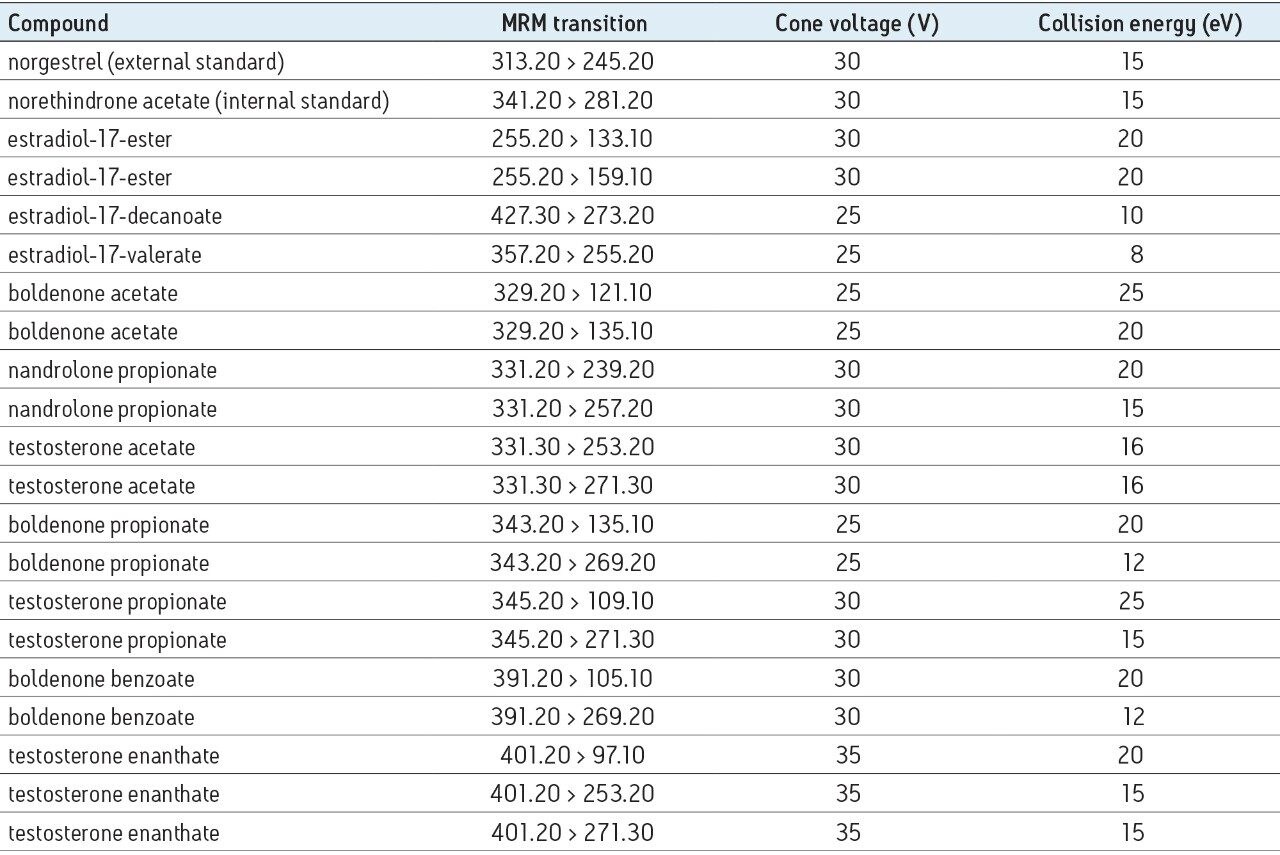
Appendix 1 contains a list of the MRM transitions used along with the precursor ion experimental conditions.
The works of Nielen et al.7 and Duffy et al.9 in LC-MS/MS helped us to start this work in UPLC-MS/MS analysis.
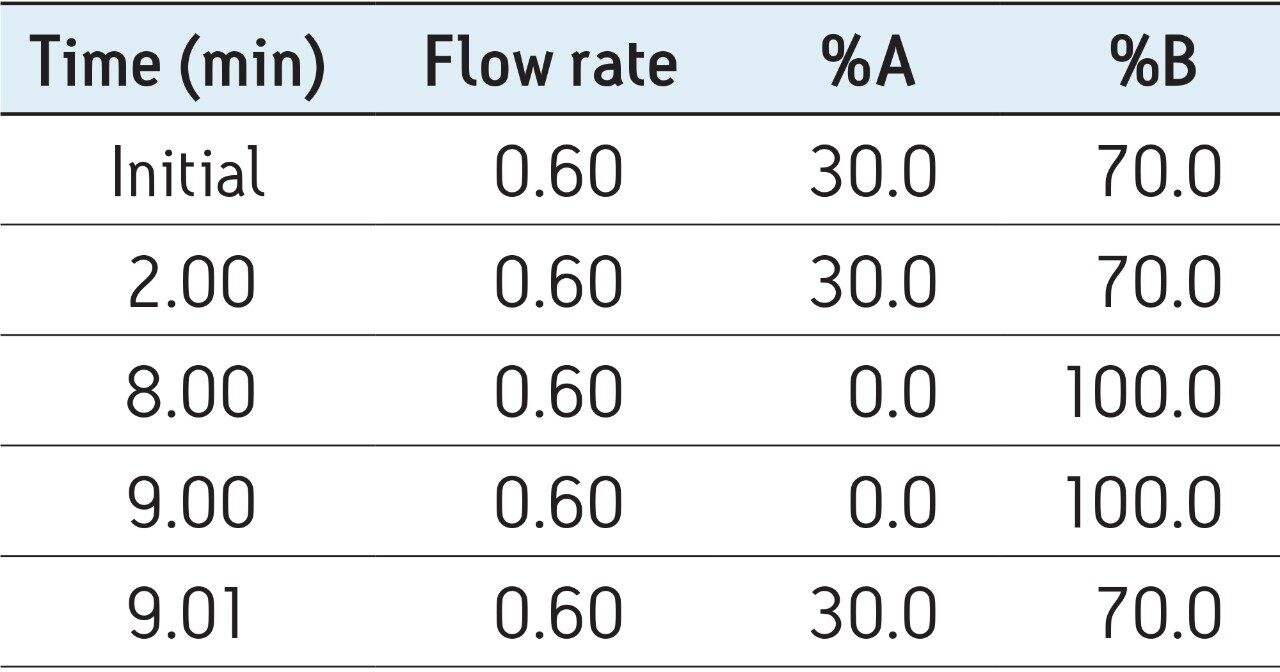
The first assays were carried out according to the gradient described by Nielen et al.7 with initial conditions of 80% acetonitrile. Unfortunately, the most polar compounds, such as the estradiol esters containing short alkyl groups (acetate, propionate), eluted too close to the void volume. The solution was to decrease the initial percentage of acetonitrile to 70% and to stay in isocratic mode for the first 2 min to allow their efficient separation.
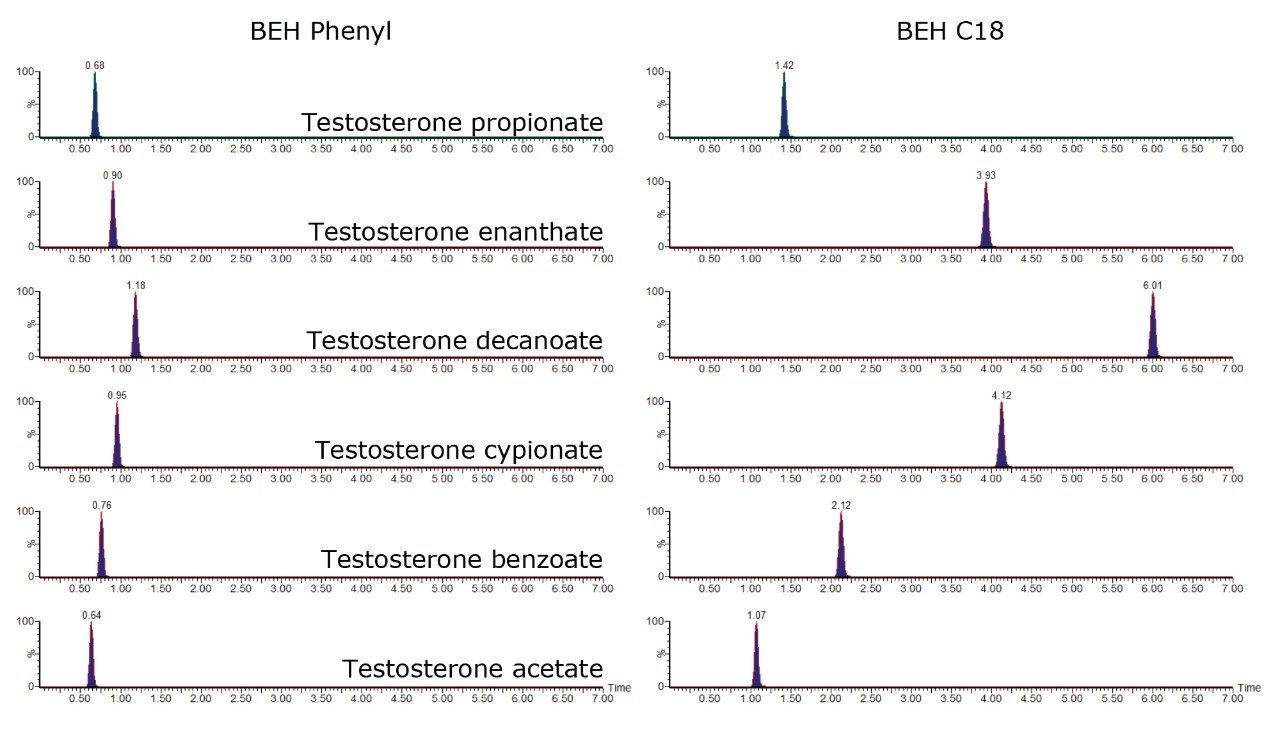
After these considerations, two different Waters column chemistries with 1.7 μm particles were tested: octadecyl (ACQUITY UPLC BEH C18) and phenyl (ACQUITY UPLC BEH Phenyl). The specific retention of estrogens on the BEH phenyl was expected due to π-π interactions. The best separation was achieved on Waters ACQUITY UPLC BEH C18 Columns, even for estrogen esters, as illustrated in Figure 1. However, all the compounds can be analyzed in 2 min with the phenyl column if sample throughput is the main challenge to overcome. For the hair samples, ACQUITY UPLC BEH C18 Columns, which provided the best separation, were chosen to minimize potential matrix effects.
After a brief study in negative ion mode, which was theoretically dedicated to the estrogen esters, positive ion mode was adopted. The pseudo-molecular ion was always detected under these conditions for all the steroids considered.
An acquisition in scanning mode was completed after optimizing the capillary and cone voltages to achieve the best intensity for the [M+H]+ pseudo-molecular ion. A product ion scan of each pseudo-molecular ion was performed to reveal the major fragment ions associated to a specific collision energy applied in the collision cell. After a manual ramp of collision energy, two energies per compound were selected, allowing a specific precursor to product transition to be recorded. Example full scan and product ion spectra for boldenone benzoate are illustrated in Figure 2.
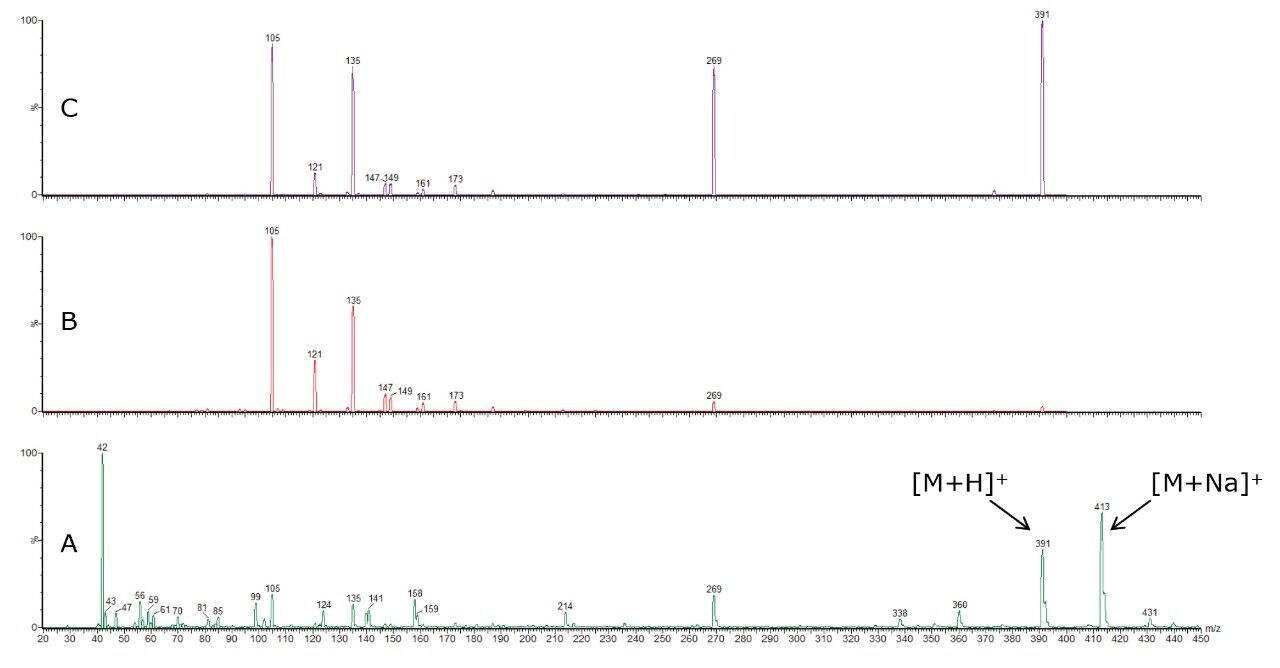
The specific fragmentation of these esters also allows selectivity to be gained in MRM analysis. Indeed, with the same [M+H]+ ion of m/z 331, the analysis of testosterone acetate andnandrolone propionate would not be specific in full scan or selected ion recording (SIR) modes. Fortunately, the fragment ions are characteristic to the steroid structure, so the transitions of testosterone acetate and nandrolone propionate are different: 331 > 271 and 331 > 257 respectively, and lead to specific detection.
Finally, an MRM method was created, allowing the monitoring of at least two transitions per compound. Functions were created after the UPLC optimization to increase the dwell time to allow the longest time for each transition.
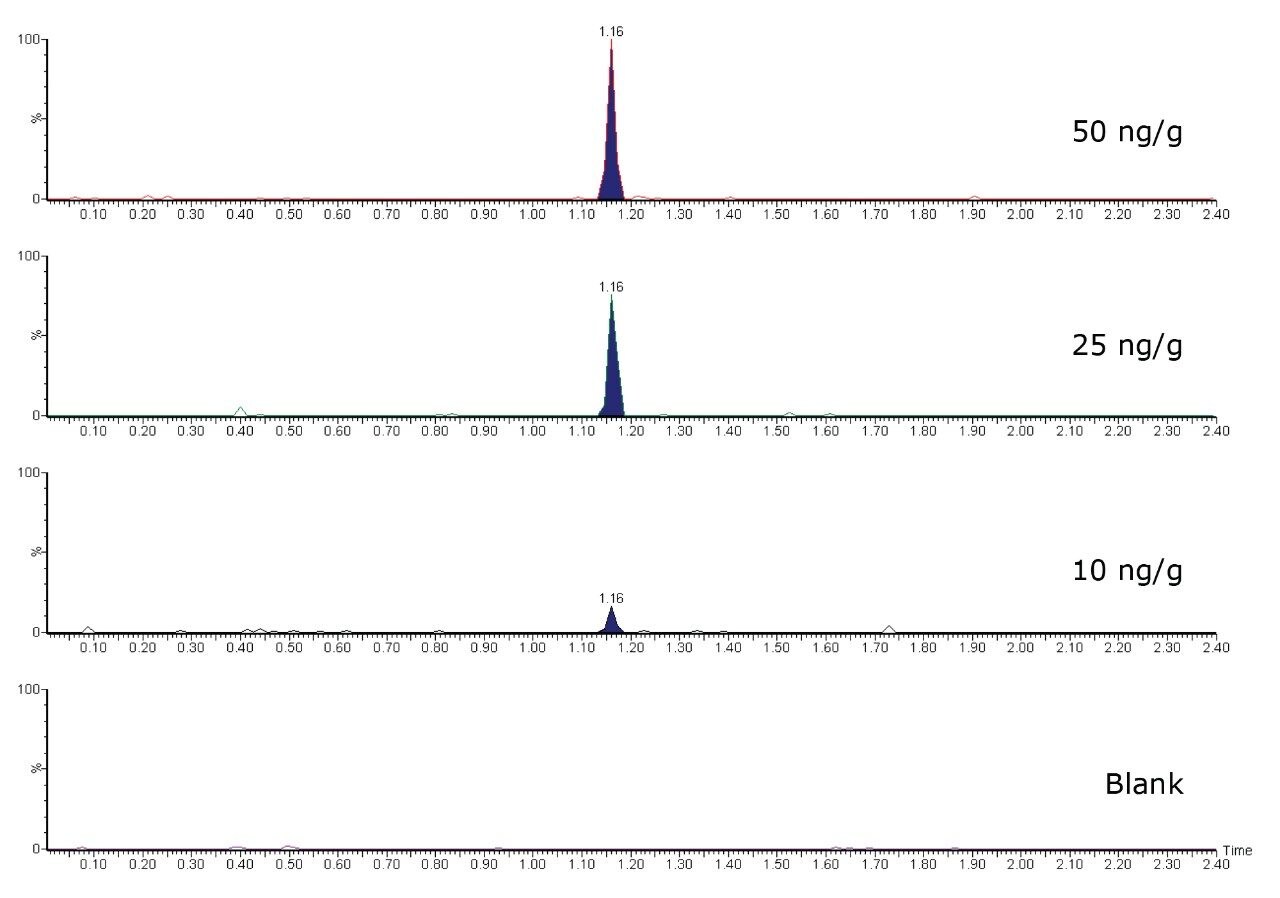
Spiked samples (between 10 and 250 ng/g) and blank samples were injected using the UPLC-MS/MS method. Figure 3 illustrates the analytical results acquired for androgen esters.
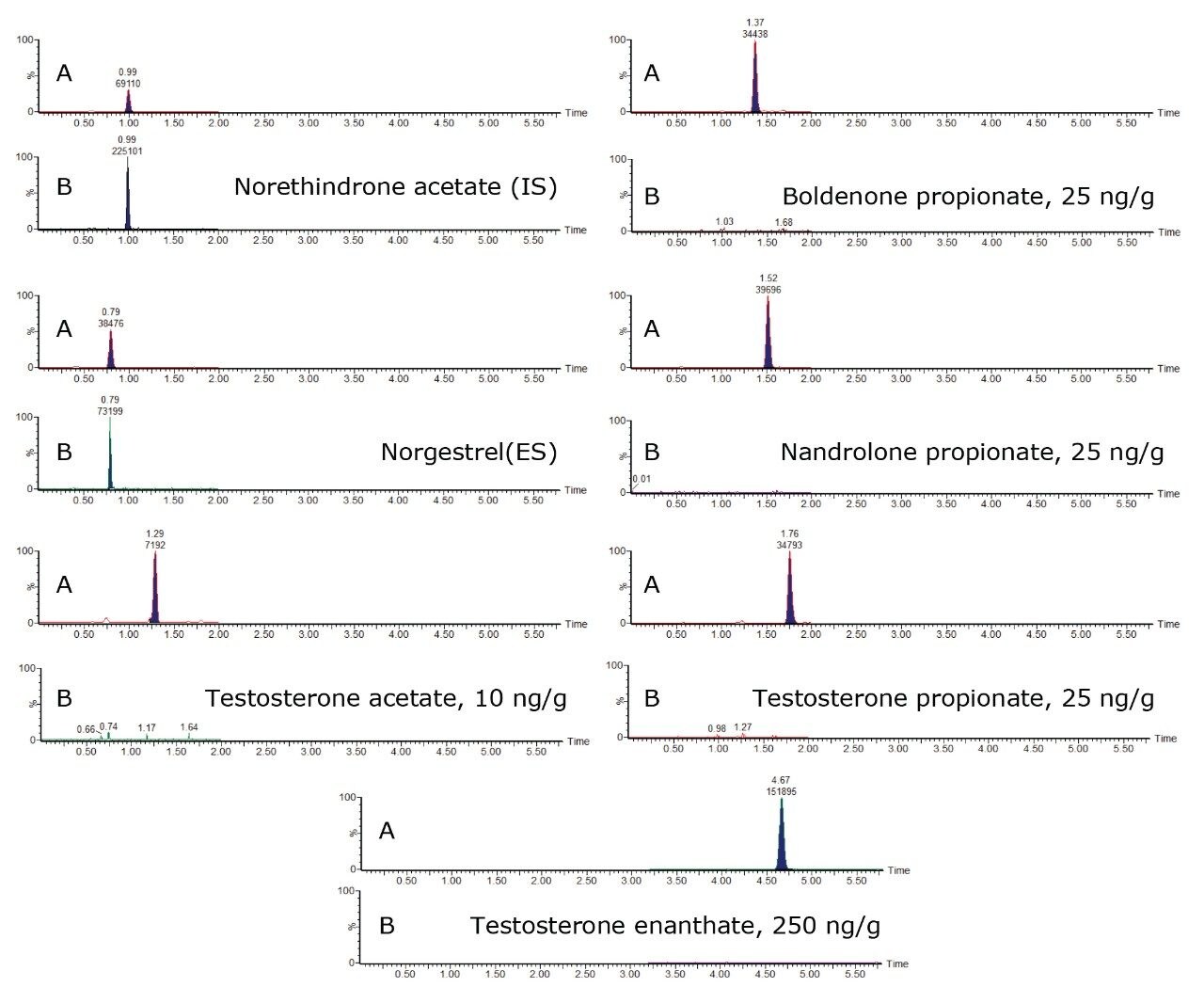
Compared to the established GC-MS/MS method,5,9 the UPLC-MS/MS method is five times faster, with all esters included in a single run. The chromatograms are clear of interference at relevant levels, particularly for the androgen esters. However, at low concentration levels, around 10 ng/g, some interference was still observed for the estrogen esters. The UPLC-MS/MS method was compared to the widely accepted GC-MS/MS method5,9 and demonstrated reasonable agreement for testing estradiol benzoate and nortestosterone decanoate.
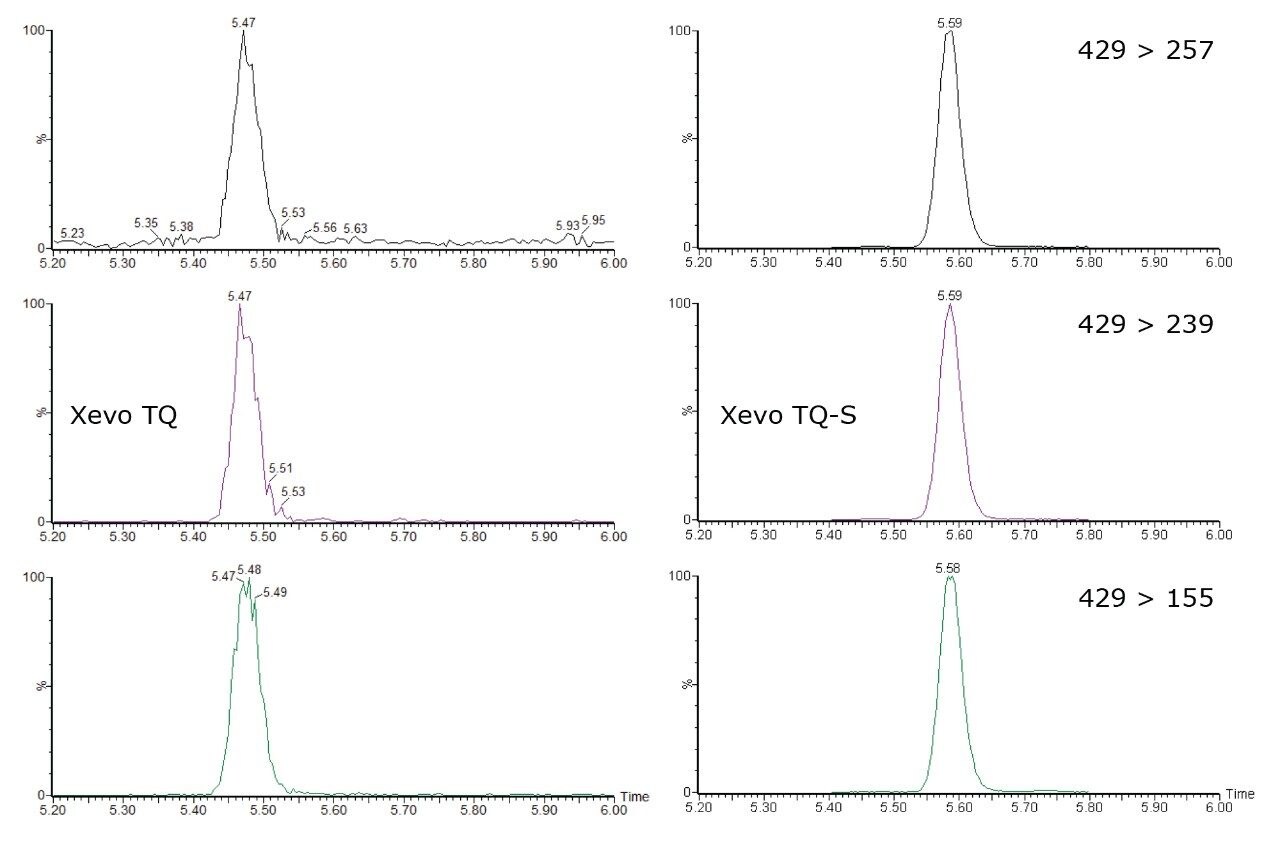
One way of minimizing interference is to decrease the amount of matrix that is injected onto the system. This can be achieved by using smaller injection volumes or diluted samples. However, for both approaches an instrument with absolute sensitivity is required to meet the required levels dictated by legislation. The increased sensitivity of Xevo TQ-S compared to Xevo TQ is illustrated in Figure 4 for nandrolone decanoate. The results show an increase in signal-to-noise for all three MRM chromatograms on raw, unsmoothed data. This allows lower concentration levels to be reached, or alternatively, for less matrix to be injected onto the system, while maintaining the required level of performance.
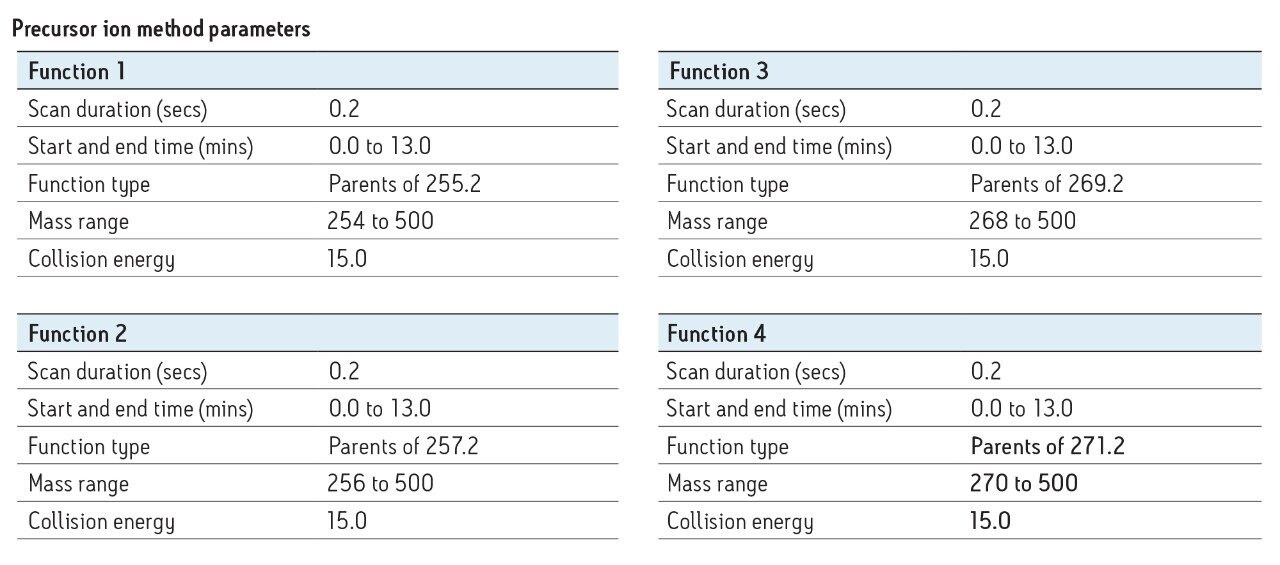
Fragmentation was observed with [Msteroid – H2O + H]+ appearing at the lowest collision energies (10 to 20 eV) that is specific to each steroid. The ester groups gave fragments under specific collision conditions of m/z 255 (estradiol), m/z 257 (nandrolone), m/z 269 (boldenone), and m/z 271 (testosterone). The non-targeted screening of steroid esters by precursor ion scanning was envisaged and tested on real samples to conclude its efficiency in hair.
Spiked samples (same concentrations as in Figure 3) and blank samples were analyzed using precursor ion scanning to assess the performance of such a screening approach, particularly in terms of sensitivity and specificity. The results are shown in Figure 5. The total ion chromatograms (TICs) are very similar if the spiked and blank samples are compared. This means that it remains very difficult to highlight a suspicious compound in this way. However, if a little targeted knowledge is applied to the sample, and a selected precursor ion is extracted, such as m/z 401 from precursor ion 271, which corresponds to testosterone enanthate at 3.90 min, a diagnostic signal clearly appears from the noise.
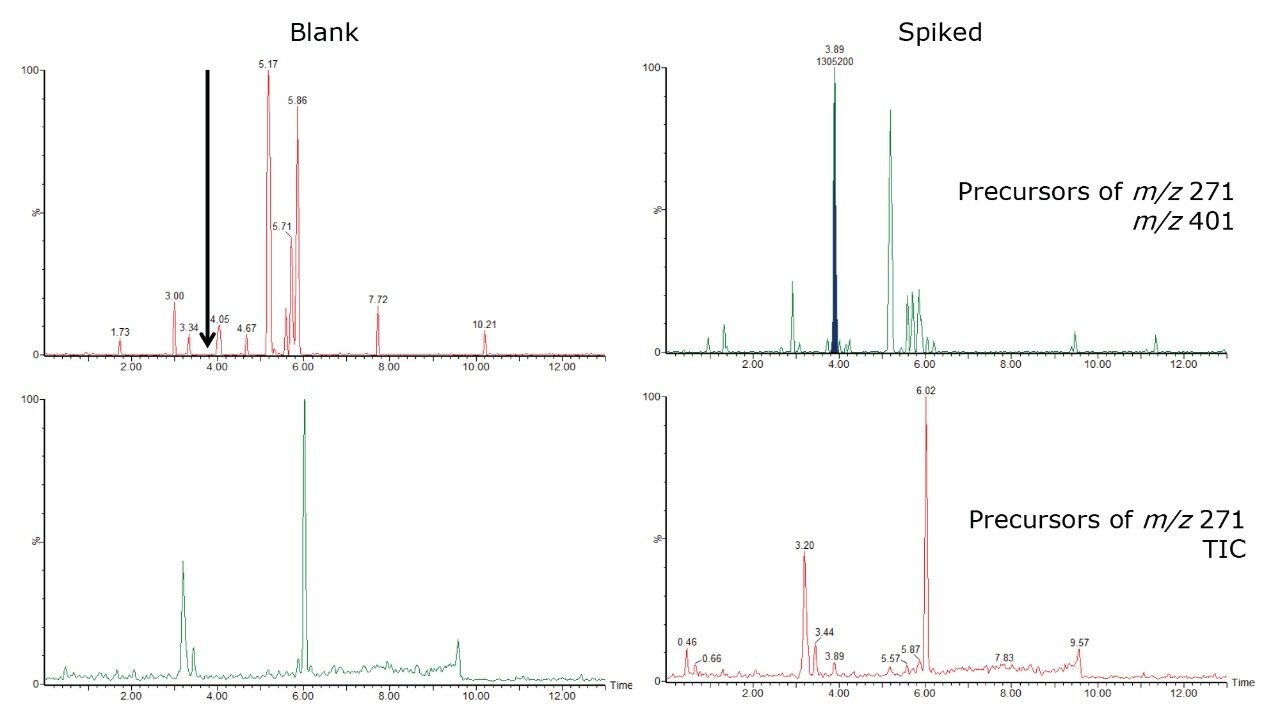
Figure 6 illustrates the precursor ion scanning approach on Xevo TQ-S, where nandrolone propionate could be successfully detected in complex hair samples at less than 10 ng/g. This approach would allow for the non-target screening of new “designer drugs”, while maintaining the required level of performance for compounds at extremely low concentrations (low ppb range) in samples of limited size (e.g., 100 mg hair).
The high sensitivity offered by Xevo TQ MS allows unambiguous determination of steroid esters at ng/g level in cattle hair. This offers an answer to the problem of lack of methodology of detecting the compounds themselves without having to rely on the detection of the metabolites in urine that are the same as from endogenous steroids.
Higher sensitivity offered by Xevo TQ-S also allows the potential to improve the detection of all banned substances in hair by reducing matrix interference, and in this way improving the control of growth promoters in breeding animals.
Relevance of precursor ion scanning on Xevo TQ and Xevo TQ-S facilitates the development of new generic screening approaches to the field of steroid ester analysis.
720003892, March 2011