
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates that the characteristically low carryover performance of the ACQUITY UPLC I-Class System is ideally suited for highly sensitive MS methods which require quantification across multiple orders of magnitude.
The ACQUITY UPLC I-Class Sample Manager with the flow-through needle (FTN) and fixed-loop (FL) injection platforms was designed to deliver the ultimate in low carryover and leverage the performance of highly sensitive mass spectrometers.
Today, highly sensitive mass spectrometers can deliver linear dynamic ranges as great as five orders of magnitude. In order to validate methods that extend across this entire range, a method must not only demonstrate linearity, but also show that the carryover after the upper limit of quantification (ULOQ) is at least 20% below the lower limit of quantification (LLOQ). Therefore, a quantification range of 0.005% up to 100% for an analyte is achievable that also demonstrates carryover below 0.001% (or below LOD). Inlet technology typically limits this range, as sample injection carryover performance may not meet this difficult requirement. Carryover can propagate from either the instrument hardware or the analytical method, and distinguishing between the two sources can be challenging. When injection is demonstrated as the source of carryover, optimizing the wash solvents and wash conditions can dramatically improve carryover performance. However, instrument design can also be a very important contributor in the reduction of carryover.
The ACQUITY UPLC I-Class System was designed to deliver the ultimate in low carryover performance. It has two different sample delivery designs that fulfill the needs of different applications and work flows. Both were designed to deliver excellent carryover performance. To demonstrate that both platforms meet the requirements for quantification from 0.005% to 100%, a method was developed to analyze omeprazole using the Xevo TQ-S. Carryover on the ACQUITY UPLC I-Class System (FTN) is shown in Figure 1. This injector style is inherently well-suited for managing carryover.
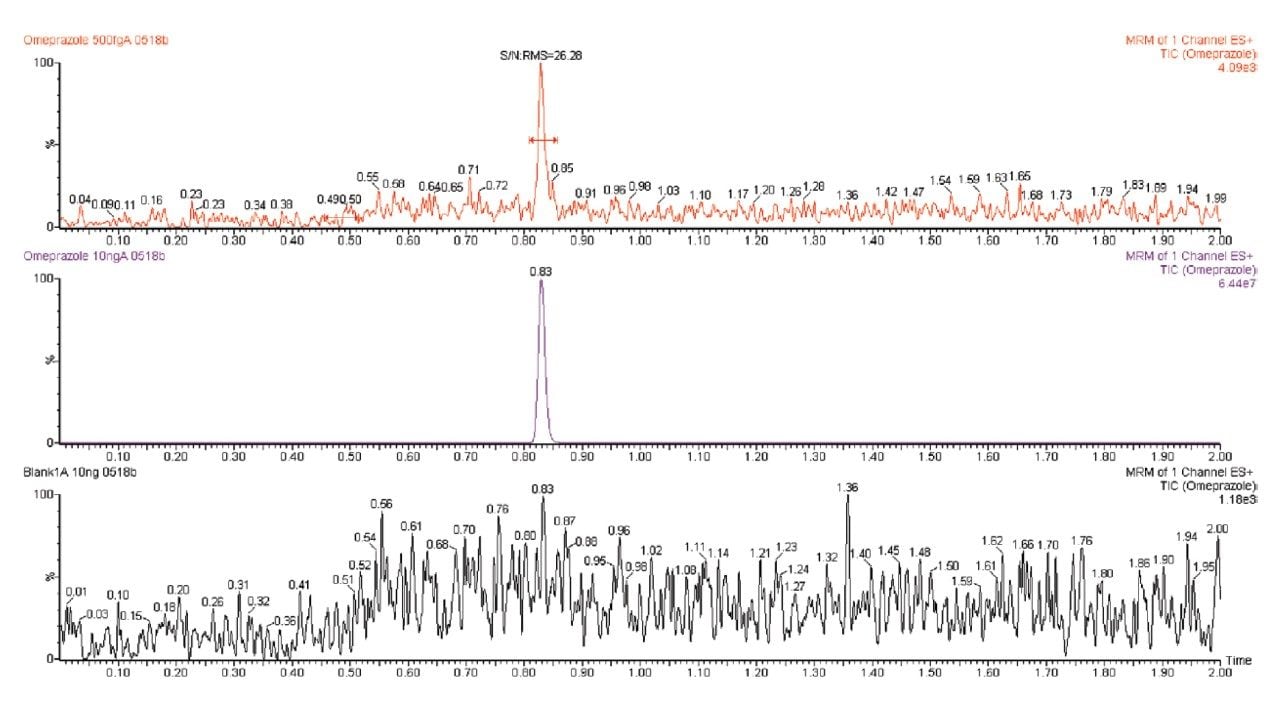
The inside of the needle is washed by the gradient while the exterior of the needle is washed during the run; therefore, the wash solvent and duration can be aggressive without adding cycle time. Sample carryover of omeprazole was below both the LOD and the 0.001% requirement for validation.
The assay was then run on the ACQUITY UPLC I-Class System (FL). This injector is optimized to ensure that all fittings, tubing, and connections associated with sample transfer deliver minimized sample carryover. Additionally, new software functionality allows the injection valve to be actuated at the end of the gradient to wash valve surfaces and further improve carryover. For omeprazole, no carryover was detected after the ULOQ and was, therefore, below the required 0.001%. Data from both injectors is summarized in Figure 2. Since neither injector had carryover within the linear range of the MS (ULOQ = 10 pg on column),the concentration was increased five-fold. From the data shown in Figure 3, the actual carryover is measured at 0.00041% for the FTN and 0.00052% for the FL.
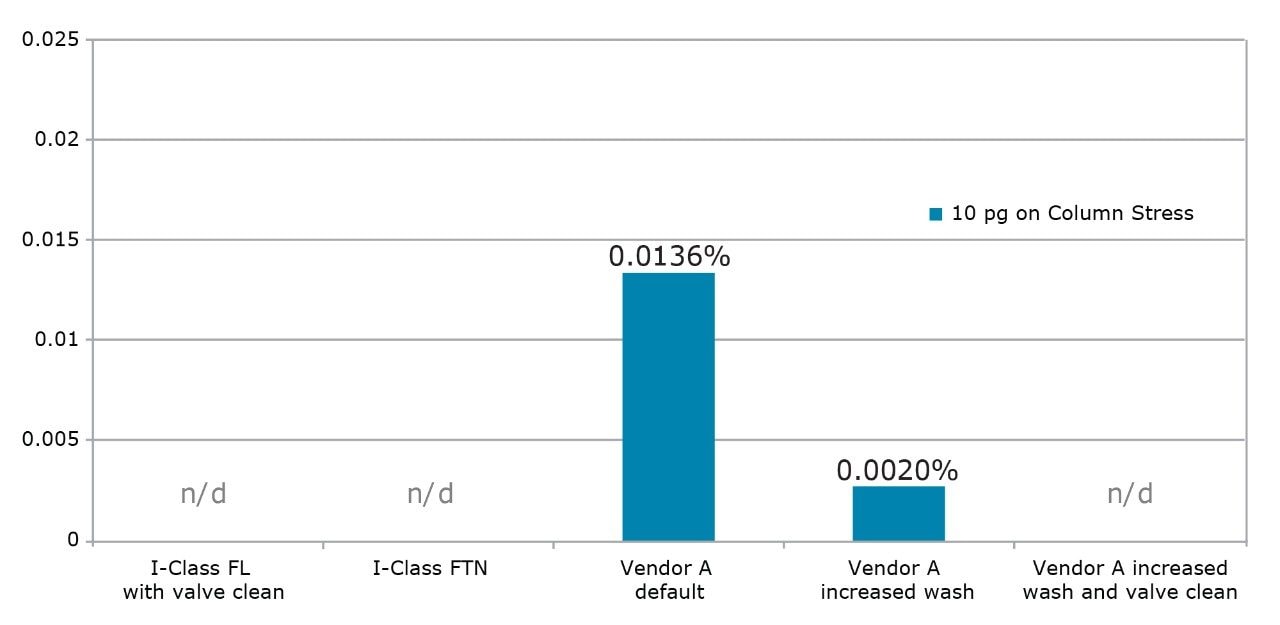
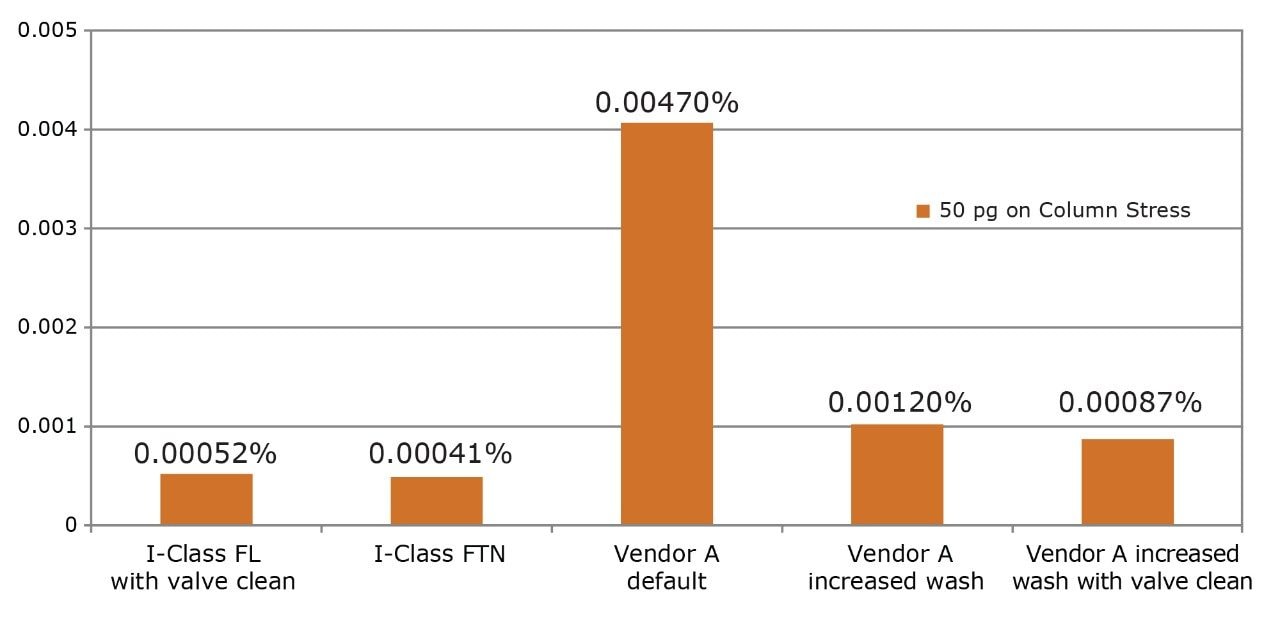
For the purpose of comparison, carryover on another UHPLC system was examined. On this UHPLC system, needlewash does not occur simultaneously with run time, adding cycle time and compromising throughput. With the default wash conditions, an excessive level of carryover is observed as seen in Figure 2. If the wash is increased to the recommended 10 s, carryover is still present. Only when the wash is increased to 20 s with valve actuations enabled does the carryover drop below LOD. When the concentration was increased five-fold to quantify carryover as shown in Figure 3, it was still up to 2x greater than that observed on the ACQUITY UPLC I-Class System; however, it required a 20 s longer injection cycle time.
When developing methods requiring highly sensitive mass spectrometers with increasingly wide linear dynamic ranges like the Xevo TQ-S, it is important to have an inlet that delivers carryover performance which can fully utilize this powerful tool. The ACQUITY UPLC I-Class System easily meets these requirements without compromising other important factors such as sample throughput.
720004140, October 2011