
In this application note, we provide an overview of the physical and chemical characteristics of Waters’ patented RapiGest SF and illustrate selected application areas. First introduced in 2002 as an enzyme-friendly surfactant to assist in-solution protein digestion, RapiGest SF is an innovative product created to improve protein solubility during sample preparation.
In this application note, we provide an overview of the physical and chemical characteristics of Waters’ patented RapiGest SF and illustrate selected application areas. First introduced in 2002 as an enzyme-friendly surfactant to assist in-solution protein digestion, RapiGest SF is an innovative product created to improve protein solubility during sample preparation.
The mechanism by which RapiGest SF improves the speed and completeness of digestion is illustrated in Figure 1. Mild protein denaturation opens protein structure and exposes the proteolytic sites to enzymatic cleavage. Enzymes are more resistant to denaturation than common proteins and remain active in RapiGest SF solutions. More complete denaturation of globular proteins can be accomplished by heating in a RapiGest SF solution at elevated temperatures prior to the addition of enzymes and incubating the sample with the enzyme at 37 °C.1,2
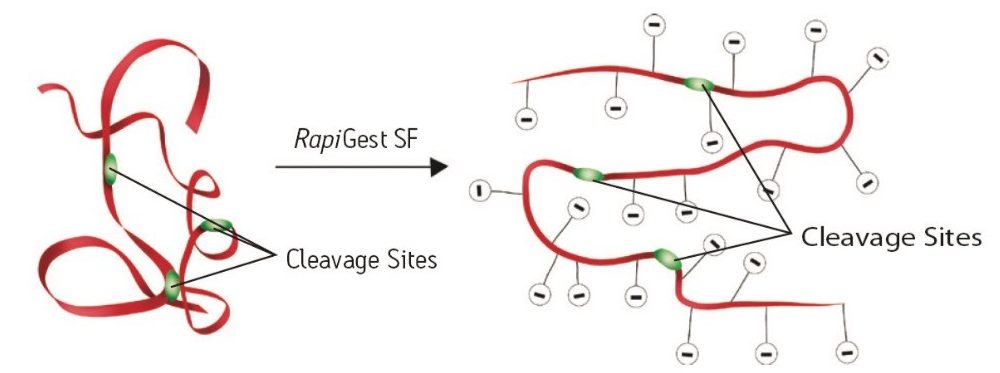
More than 200 peer-reviewed journals have cited the use of RapiGest SF for general sample solubilization purposes, mostly for proteomic applications. Recently, many pharmaceutical laboratories have adopted RapiGest SF for biotherapeutic protein characterization. Because of improved digestion and easy surfactant removal prior to LC and MS analysis, RapiGest SF has become widely accepted for many applications, including high sequence coverage LC-UV/MS peptide mapping of therapeutic proteins.
|
LC System: |
Waters ACQUITY UPLC System |
|
Column: |
ACQUITY UPLC BEH 300 C18, Peptide Separation Technology Column, 2.1 x 100mm |
|
Column temp.: |
40 °C |
|
Sample injected: |
2 μL (10 pmol) |
|
Solvent A: |
0.1% formic acid in water |
|
Solvent B: |
0.1% formic acid in acetonitrile |
|
Flow rate: |
200 μL/min |
|
Gradient: |
0 to 2 min: 2% B 2 to 92 min: 2 to 35% B 92 to 102 min: 35 to 50% B 102.1 to 105 min: 90% B 105.1 to 110 min: 2% B |
|
MS System: |
SYNAPT MS (V mode) |
|
Capillary voltage: |
3.2 kV |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
350 °C |
|
Desolvation gas: |
700 L/hr |
|
MS scan rate: |
1 sec/scan |
|
Lock Mass channel: |
100 fmol/μL Glu-Fib peptide (m/z 785.8426, z = 2), flow rate 20 μL/min |
RapiGest SF is an acid labile surfactant that undergoes hydrolysis in acidic conditions.1 This unique feature can be utilized to remove the surfactant from solutions when desired. The structure of RapiGest SF and its byproducts from the acid hydrolysis are shown in Figure 2. The acid labile property facilitates a nearly complete surfactant degradation of within about 45 minutes at pH 2.1
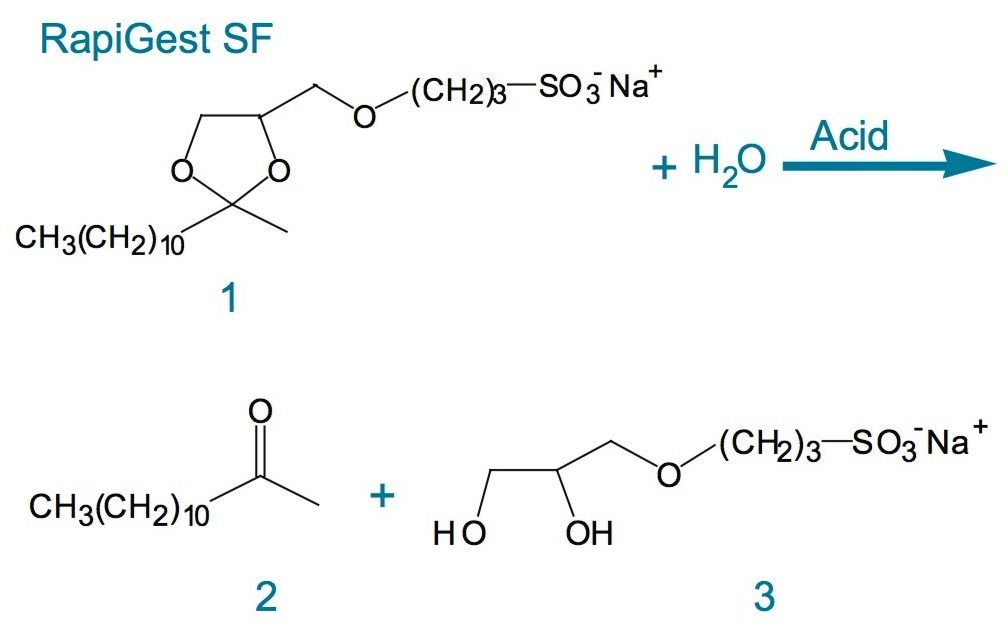
The surfactant decomposes into two products, dodeca-2-one and sodium-3-(2,3-dihydroxypropoxy) propanesulfonate. The first compound is water immiscible, and can be removed by centrifugation. The second degradant is highly soluble in aqueous solutions and practically unretained in reversed-phase LC mode. The aqueous fraction of enzymatic digest can be directly analyzed by HPLC, LC-MS, or by MALDI-TOF MS.
No additional detergent removal steps are required (e.g., dialysis) prior to sample analysis. The enzymatic digestions are typically acidified with acids such as formic acid, trifluoroacetic acid (TFA), or hydrochloric acid (HCl) to degrade RapiGest SF prior to further analysis. The recommended pH for degradation is ≤ 2.
Trypsin is the most common proteolytic enzyme used for peptide mapping and proteomic applications. We have investigated trypsin activity in the presence of RapiGest SF and compared it to most common denaturants cited in literature. The assay was based on trypsin induced hydrolysis of N-α-benzoyl-L-arginine ethyl ester (BAEE) in 50 mM ammonium bicarbonate (pH 7.9) at room temperature. Changes in trypsin activity were calculated by measuring the rate of BAEE hydrolysis at UV 253 nm. Trypsin activity in the selected denaturant solution was compared against the control sample (no denaturant). The results are shown in Table 1.
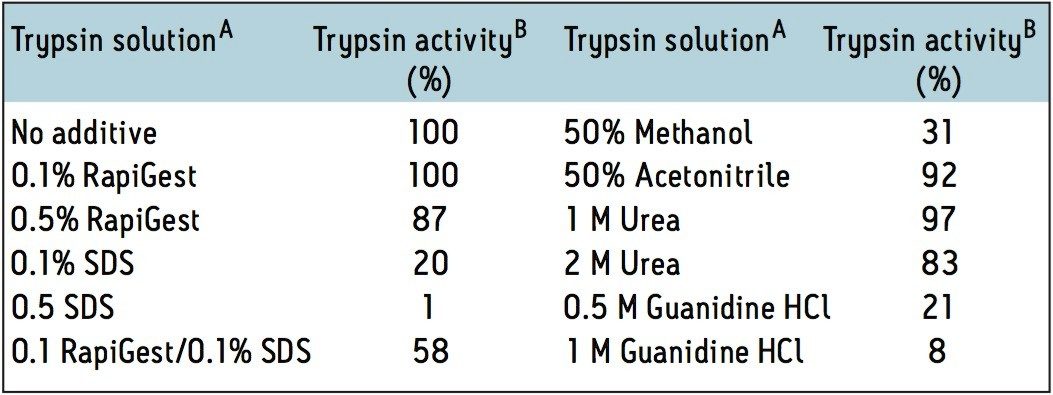
The data in Table 1 suggest that at low concentrations (0.1%), RapiGest SF does not inhibit trypsin activity. This contrasts with structurallysimilar surfactant SDS, which appears to be a strong denaturant and inactivates the trypsin. Urea, acetonitrile, or guanidine-HCl were also proposed as denaturants for tryptic digestions. However, acetonitrile is a strong eluent and interferes with reversed-phase LC analysis of digested sample. Urea is known to cause covalent modification of proteins, and Guanidine-HCl inactivates enzyme, similarly to SDS.
The implication from this experiment is that enzyme proteolysis activity can be affected by the denaturant used to solubilize the protein samples. Using RapiGest SF at low to high concentrations does not alter the enzyme activity; therefore, optimum proteolysis digestion is achieved without using an excess of enzyme.
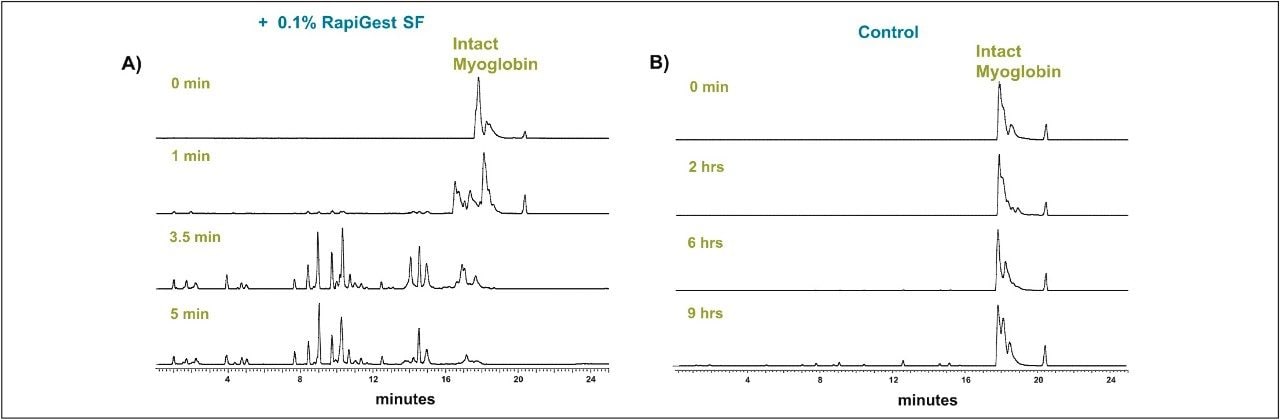
Proteins that are resistant to proteolysis can be digested within minutes using RapiGest SF. A complete protein digestion for a globular protein, horse myoglobin, was achieved within 5 minutes. The comparison of results for surfactant-aided and control digestion is shown in Figure 3. Due to its globular nature, myoglobin is known to be difficult to digest without the use of any denaturant. In the control reaction, only a small fraction of protein is digested after 9 hours of incubation with trypsin. The overall digestion efficiency significantly improved when using RapiGest SF.
RapiGest SF has been widely used in proteomics sample preparation as an effective denaturant for protein solubilization. Recently, more biopharmaceutical labs have adopted RapiGest SF in their peptide mapping protocols. Several publications document the benefits of using RapiGest SF for therapeutic protein digestion.4,5 The reported RapiGest SF concentration used ranges from 0.05 to 1% depending on the protein hydrophobicity and concentration.
We have found that a 0.05 to 0.1% concentration of RapiGest SF is sufficient to denature various sizes of proteins; higher concentration of RapiGest SF may be suited for a whole cell protein extraction type of experiment.
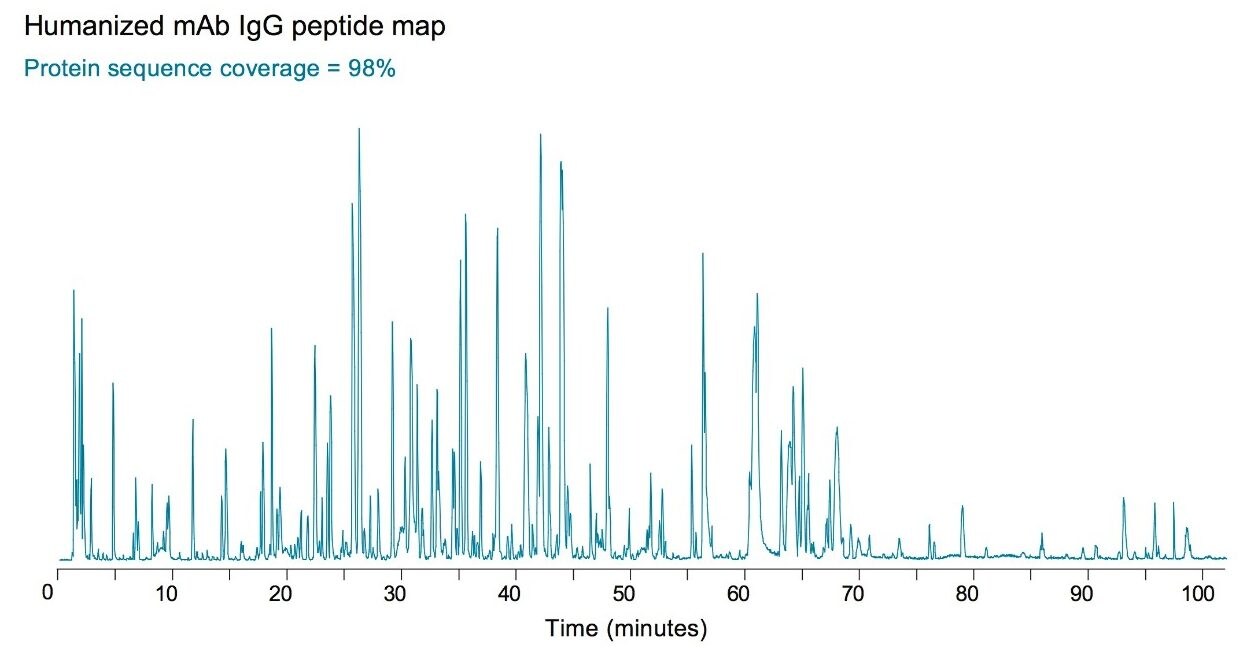
Peptide mapping of monoclonal antibodies (mAbs) is challenging due to the difficulty of digesting these large and hydrophobic proteins. The goal of peptide mapping analysis is to confirm the protein sequence and identify all present post-translational modifications (PTMs). Figure 4 shows an example of RapiGest SF-assisted digestion of humanized mAb. The parameters of sample preparation and analysis by UPLC and quadrupole time-of-flight MS are listed as guidelines.
The overall sequence coverage in the experiment shown in Figure 4 was 98%. Data analysis was performed with BiopharmaLynx Software, v.1.2. The high sequence coverage (98%) indicates a complete digestion of mAb. No intact protein or large miscleaved peptides were detected in LC-MS analysis. The remaining 2% of unaccounted sequence belong to a few two amino-acid-long peptides or to a single amino acid (R or K) that are unretainable on the reversed-phase column.
Humanized mAb sample (10 μL, 21 mg/mL) was solubilized in 50 μL 50 mM ammonium bicarbonate containing 0.1% (w/v) RapiGest SF. 2 μL of 0.1 M dithiothreitol (DTT) was added to the sample, and the sample was heated at 50 °C for 30 minutes. 4 μL of 0.1 M iodoacetamide was added to the sample, after it was cooled to room temperature, and the sample was placed in the dark for 40 minutes.
8 μg of trypsin was added to the sample (trypsin conc. = 1 μg/μL) and the sample was incubated at 37 °C overnight. The digested sample was mixed with 1% formic acid in 10% acetonitrile (1:1, v:v). The sample was diluted to 5 pmol/μL with Milli-Q water (Millipore) prior to LC-MS analysis.
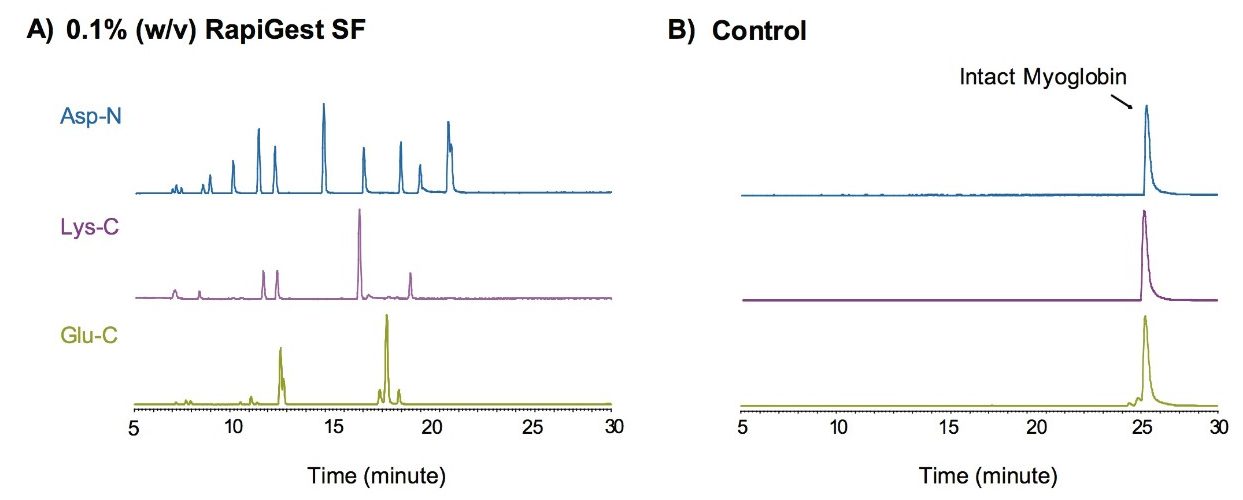
We tested RapiGest SF compatibility with multiple proteolytic enzymes, for example, Asp-N, Lys-C, and Glu-C. Efficient digest results were obtained using RapiGest SF to denature the protein prior to proteolysis (Figure 5).
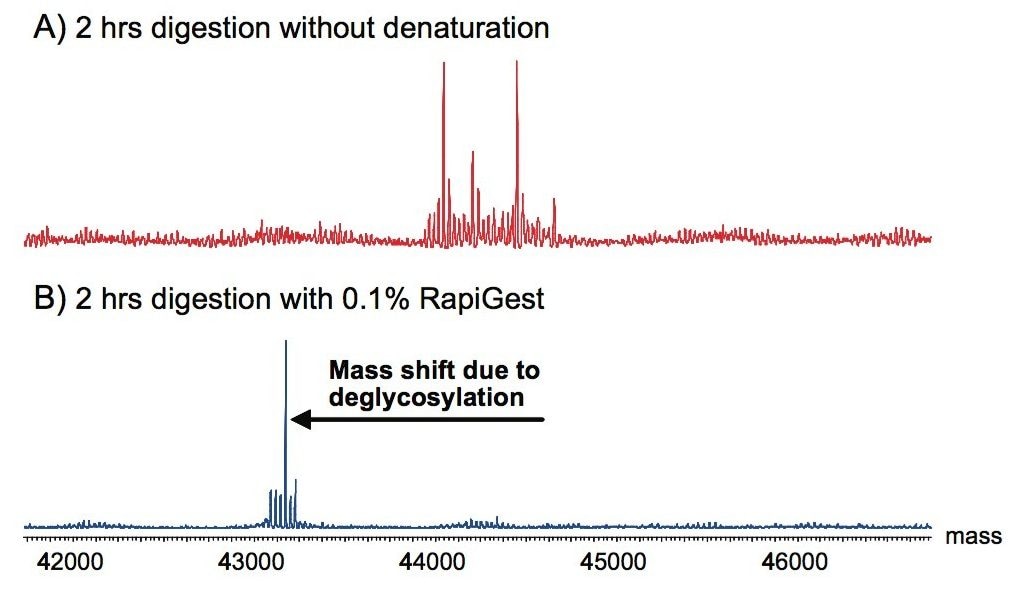
RapiGest SF was also tested with other enzymes such as PNGase F, which is used to cleave N-linked glycans from glycoproteins.2 Figure 6 illustrates the deglycosylation of chicken ovalbumin. Complete deglycosylation was observed after 2 hours in RapiGest SF-mediated digestion with PNGase F.
720003102, June 2009