
This application note describes the results of a successful validation of the analysis of shrimp tissue for tetracyclines, sulfonamides, trimethoprim, ormetoprim, and dapsone using the Waters ACQUITY UPLC I-Class PLUS System coupled to the Xevo TQ-S micro.
Veterinary drugs are used in animal husbandry and aquaculture for therapeutic or disease-preventive reasons and, in some cases, to promote growth of livestock. However, when specified withdrawal periods are not observed, unsafe antibiotic residues, or their metabolites, may be present in edible products such as milk, eggs, and meat. To meet growing demand, shrimps and other seafood are often cultivated by aquafarms, where many animals are kept in relatively small spaces, making them more prone to diseases. In order to preserve animal health as well as to ensure production and to increase yields, antibiotics are used on a large scale. Residues of these antibiotics in foods of animal origin are a major concern because they are harmful to the consumer’s health and could induce pathogens to develop resistance.1
Authorities regulate the use of veterinary drugs by setting the maximum residue limits (MRLs) or by prohibiting the use of many substances to ensure the safety of the food and facilitate international trade between countries. In the EU, there are MRLs for a number of substances, which apply to all food-producing species; sulfonamides, expressed as the combined total residues of all substances within the sulfonamide group, and for tetracyclines, which relate to the sum of parent drug and its 4-epimer except for doxycycline (DC).2 Dapsone, demeclocycline, and ormetoprim are not approved for use on food-producing animals in the EU, and as such, have no MRLs, although dapsone has a recommended concentration (RC) of 5 μg/kg.3 When the substance is prohibited, the CCα and CCβ limits should always be as low as reasonably achievable (ALARA).
Surveillance combined with the effective enforcement, investigation, and inspection activities ensures the safe and effective use of veterinary medicines in various parts of the world. For example, EU countries must implement residue-monitoring plans to detect the illegal use or misuse of authorized veterinary medicines in food producing animals. These countries must also investigate the reasons for residue violations. Non-EU countries exporting into the EU must also implement a residue monitoring plan that guarantees an equivalent level of food safety.
It is important, therefore, to develop a simple but accurate method for the determination of residues of a range of antibiotics in seafood. This application note describes the results of a successful validation of the analysis of shrimp tissue for tetracyclines, sulfonamides, trimethoprim, ormetoprim, and dapsone using the Waters ACQUITY UPLC I-Class PLUS System coupled to the Xevo TQ-S micro.
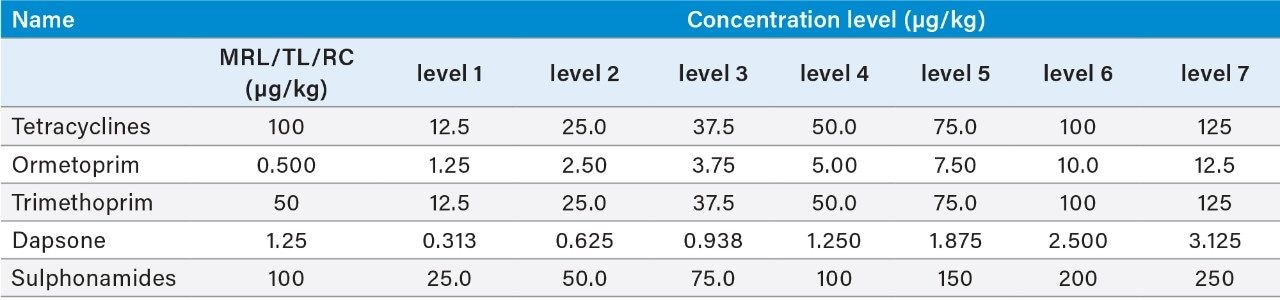
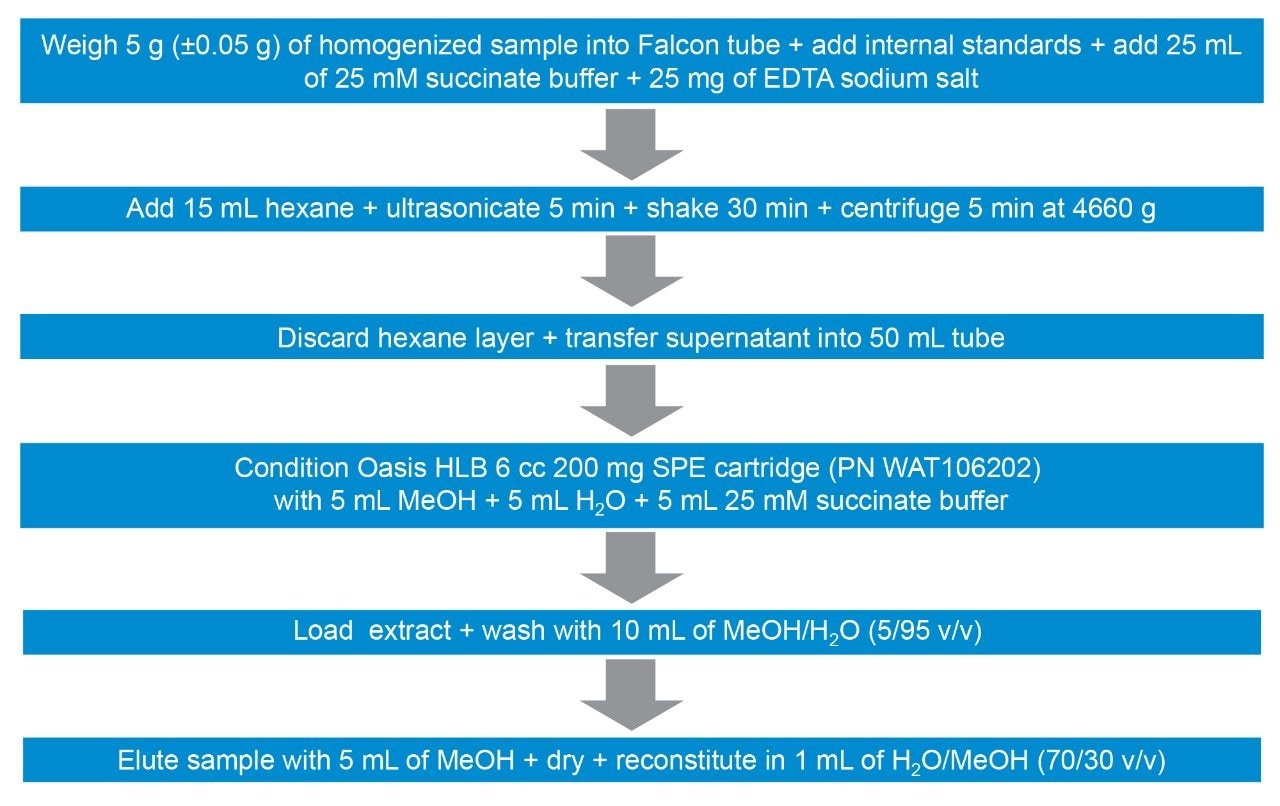
After the addition of internal standards, shrimp tissue was extracted using a mixture of chelating agents to improve extraction efficiency for tetracyclines, followed by SPE clean-up (see Figure 1 for more detail). Matrix-matched standards were prepared in shrimp tissue extract, previously shown to be blank, at the following concentrations (Table 1):
|
System: |
ACQUITY UPLC I-Class PLUS with FTN Sample Manager |
|
Column: |
ACQUITY HSS C18, 1.8 μm, 2.1 × 100 mm (p/n: 186003533) |
|
Column temp.: |
25 °C |
|
Sample temp.: |
10 °C |
|
Injection parameters: |
1 μL |
|
Mobile phase A: |
water + 0.1% formic acid |
|
Mobile phase B: |
methanol + 0.1% formic acid |
|
Sample manager wash: |
25/25/25/25 water/methanol/ isopropanol/acetonitrile with 0.2% formic acid |
|
MS system: |
Xevo TQ-S micro |
|
Polarity: |
ESI+ |
|
Capillary voltage: |
2.0 kV |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
650 °C |
|
Desolvation gas flow: |
1000 L/hr |
|
Cone gas flow: |
50 L/hr |
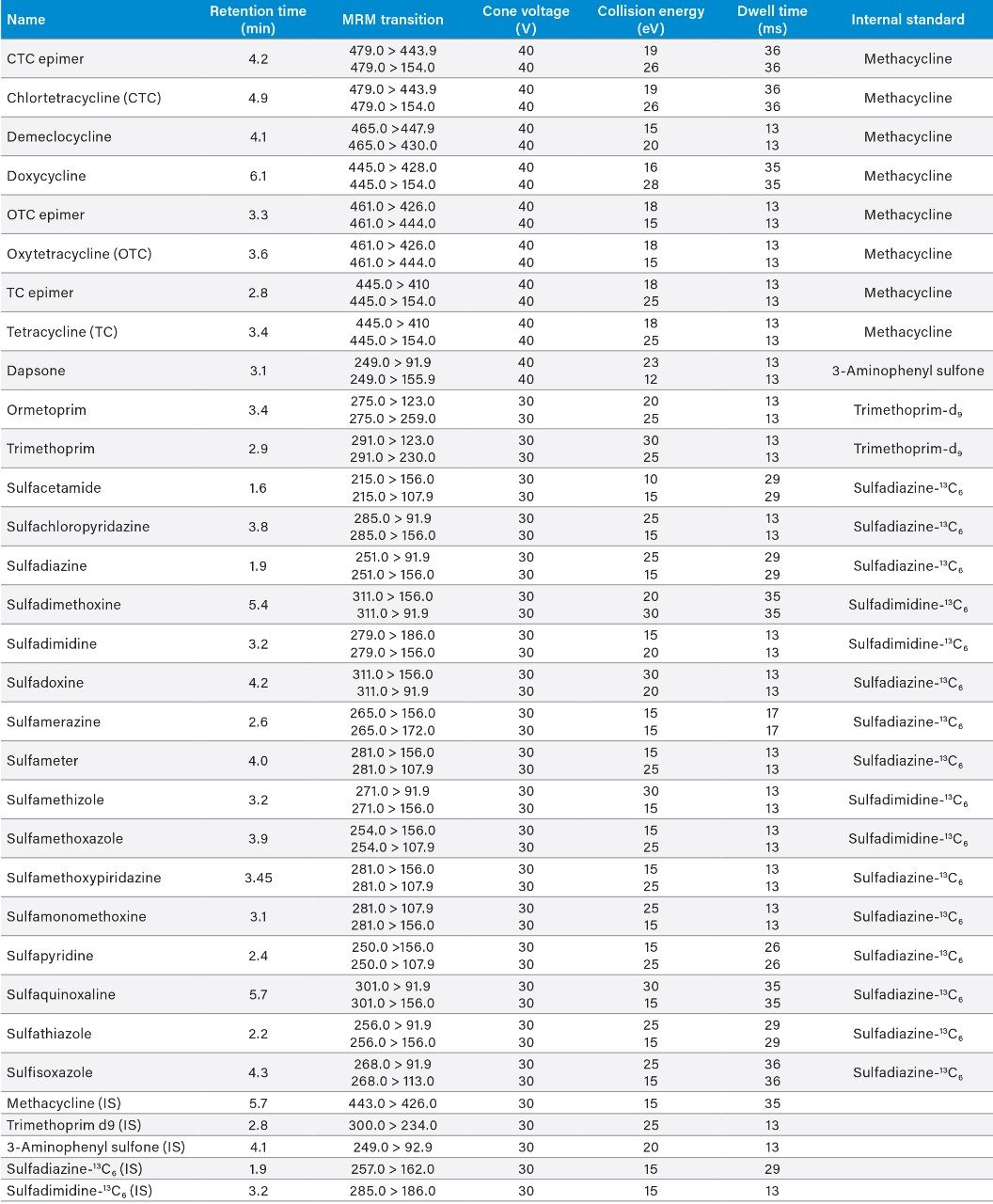
Two MRM transitions per compound were used. The dwell times were set automatically using the autodwell function to give a minimum of 12 data points across each peak. The data were acquired using MassLynx Software and processed using TargetLynx XS Application Manager. Table 2 summarizes the MRM transitions and the actual dwell time settings. The quantification traces are noted in bold.
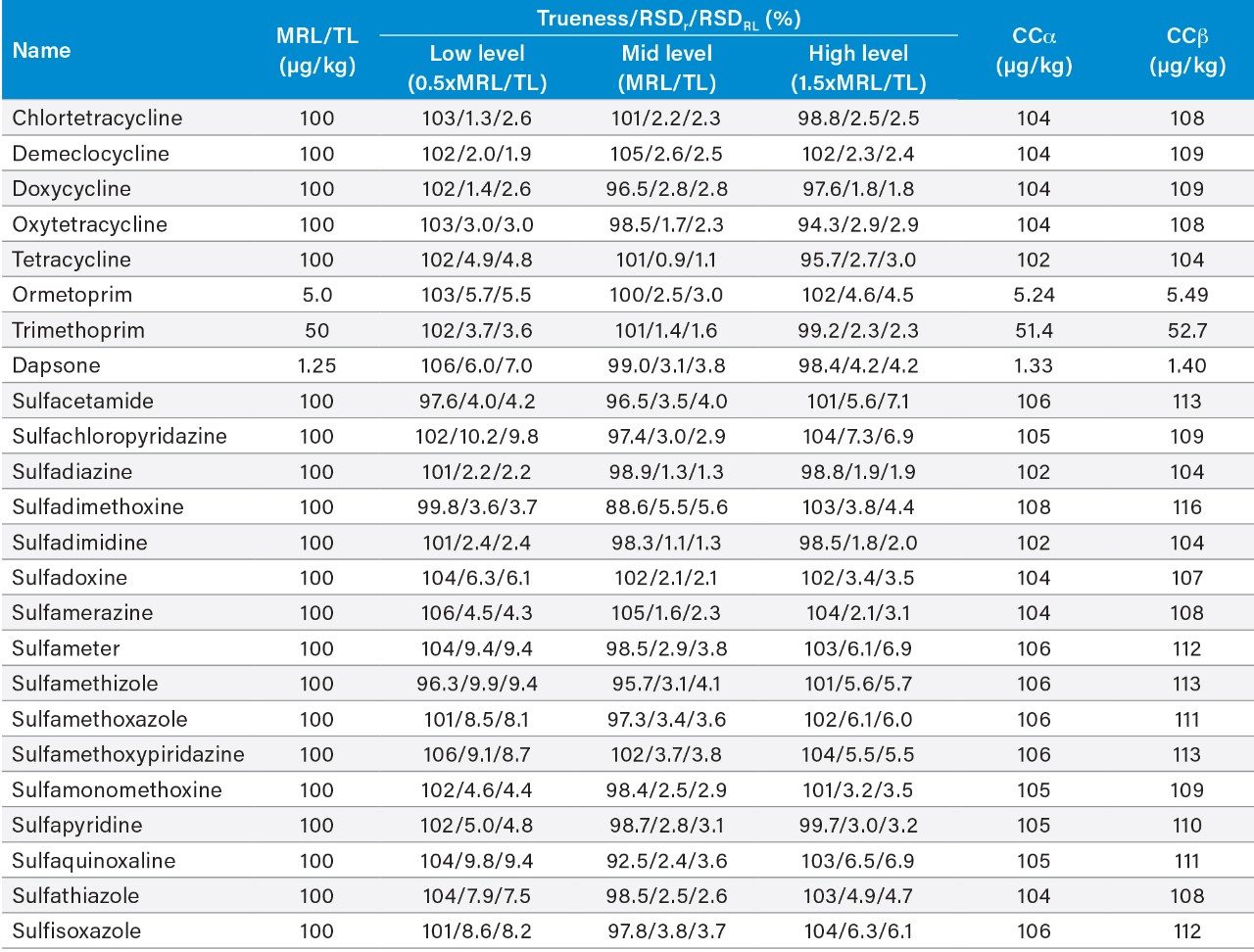
Validation was performed using spiked blank samples according to the 2002/657/EC guidelines.4 The following parameters were assessed: identification, selectivity, linearity, trueness, within-laboratory repeatability (RSDr), within-laboratory reproducibility (RSDRL), decision limit (CCα), and detection capability (CCβ). Identification was assessed by examining retention times, ion ratios, and identification points. The selectivity of the method was verified by testing blank shrimp tissue samples to check the presence of any interferences eluting at and around the retention times of the analytes. The linearity of the curves and individual residuals were checked. Replicate spiked samples of shrimp tissue were prepared and analyzed on three separate days by the same analyst. For MRL substances, samples were spiked at 0.5, 1.0, and 1.5 times the MRLs (see Table 1). However, as MRLs for tetracyclines relate to the sum of parent drug and its 4-epimer, spiking concentration were halved to 25, 50, and 75 μg/kg for each compound, and the calculated concentrations for parent and epimer summed prior to calculation of the validation parameters. For those compounds with no MRL, assessment was made at 0.5, 1.0, and 1.5 times a target level (TL). CCα and CCβ were calculated from the RSDRL as defined in 2002/657/EC.
The HSS C18 Column provides excellent retention and peak shape for all the analytes without the need for an ion pair reagent in the mobile phase for tetracyclines.5 The combination of the HSS C18 Column with methanol rather than acetonitrile and a low column temperature resulted in complete separation of the parent and epimeric forms (Figure 2).
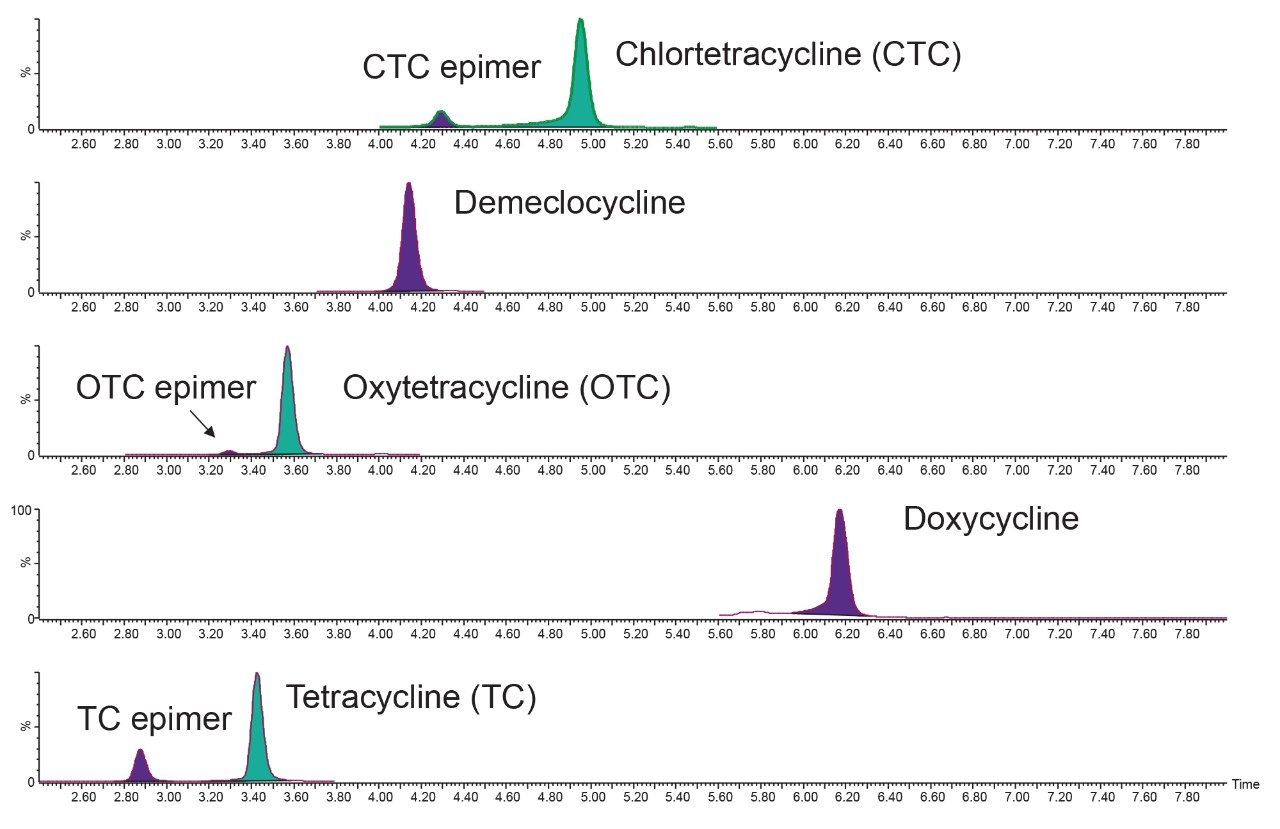
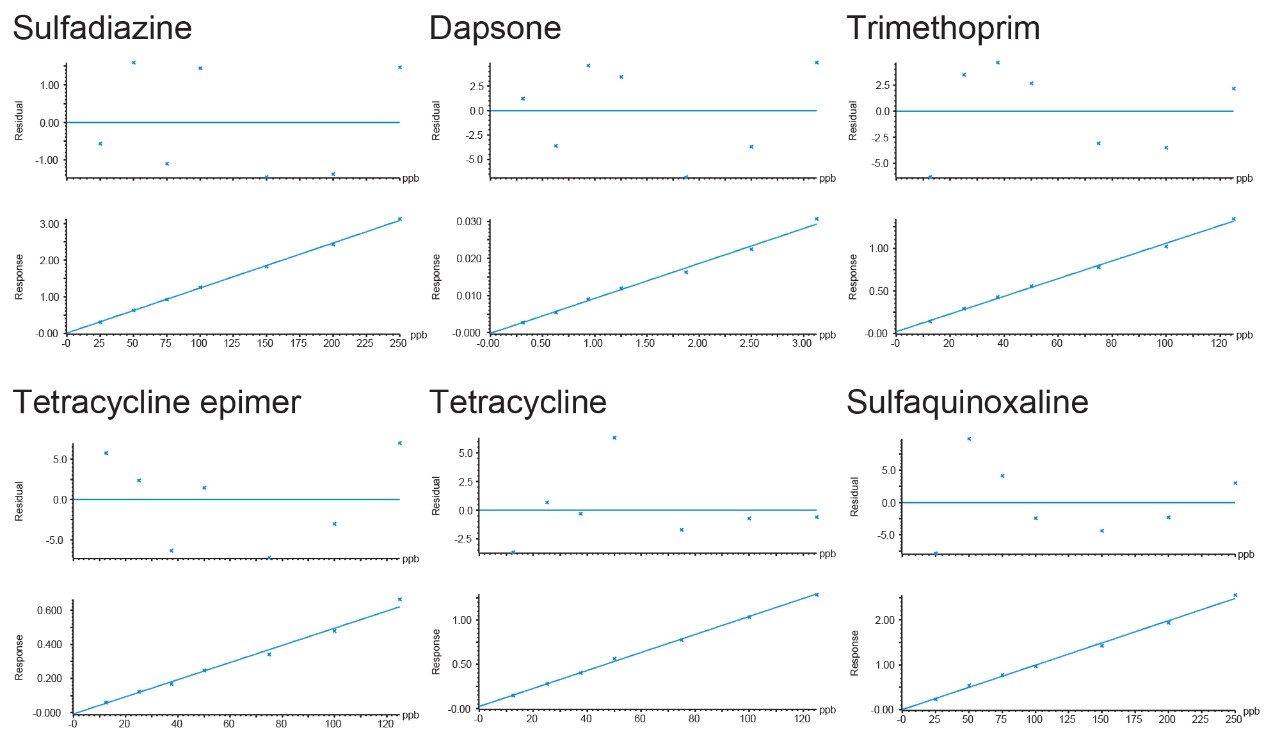
Seven blank shrimp samples were prepared each of the three days and analyzed. No signal was detected in the extracts that could lead to detection of false reporting of non-compliant samples. Some compounds were detected at trace levels but were estimated to be at concentrations much lower than the lowest standard. A 7-point calibration curve was prepared in matrix extract and acquired on each day. The two transitions for each analyte, enough to meet the required identification points (three for MRL substances and four for banned substances), gave peaks with ion ratios and retention times within the recommended tolerances when compared with the standards. Linear fit with 1/x weighing was applied and all correlation of determination (R2) values from the calibration graphs were >0.99, with individual residuals all <20% (with most <5%), demonstrating reliable quantification. Some examples of typical calibration curves are given in Figure 3.
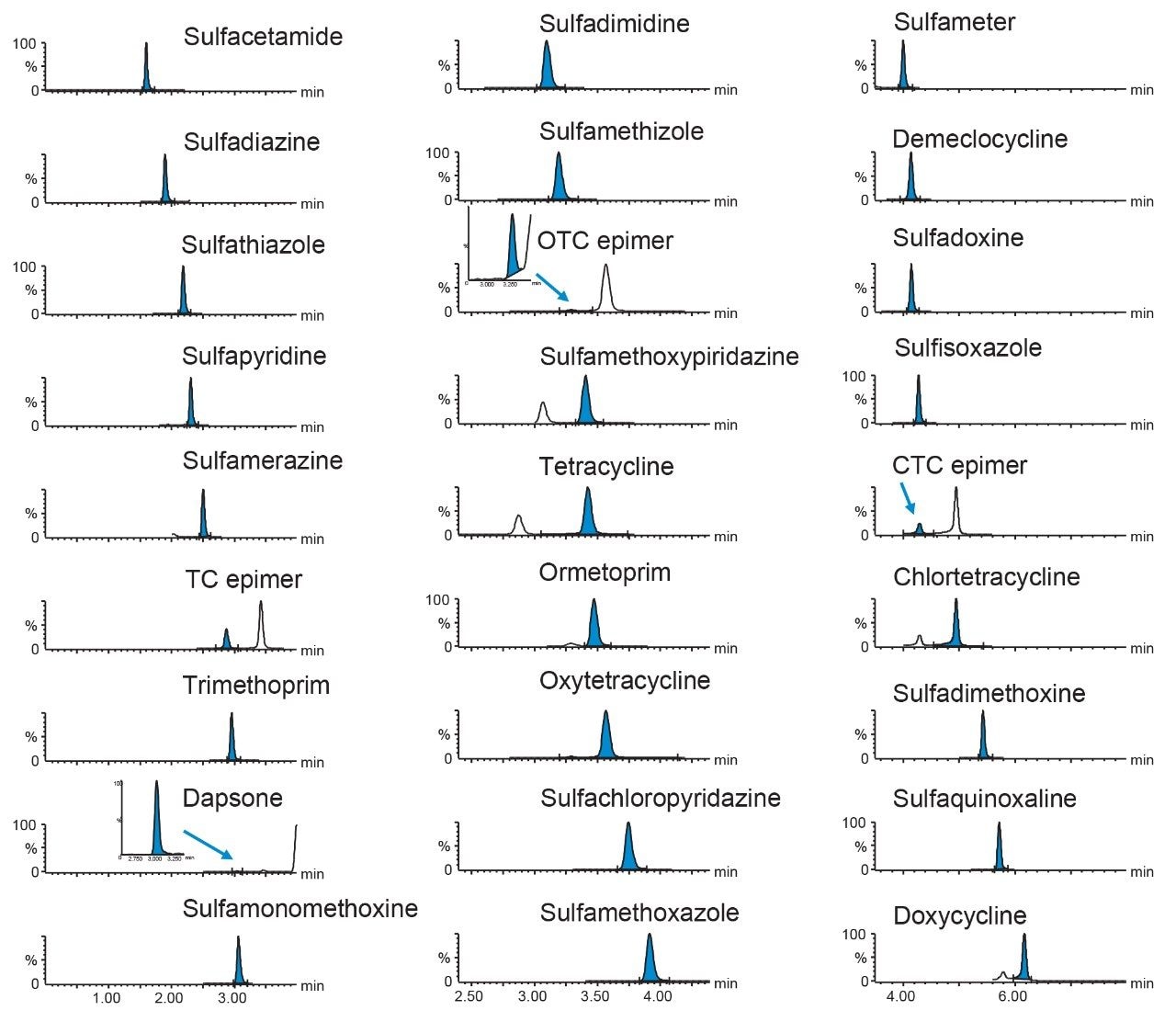
Excellent sensitivity was demonstrated from the analysis of matrix-matched standards. Figure 4 shows typical chromatograms for a selection of the antibiotics from the analysis of the matrix-matched standard at the lowest concentration, which indicates that the method is capable of detection of these antibiotics in extracts at much lower concentrations or final extracts could be diluted further prior to LC-MS/MS.
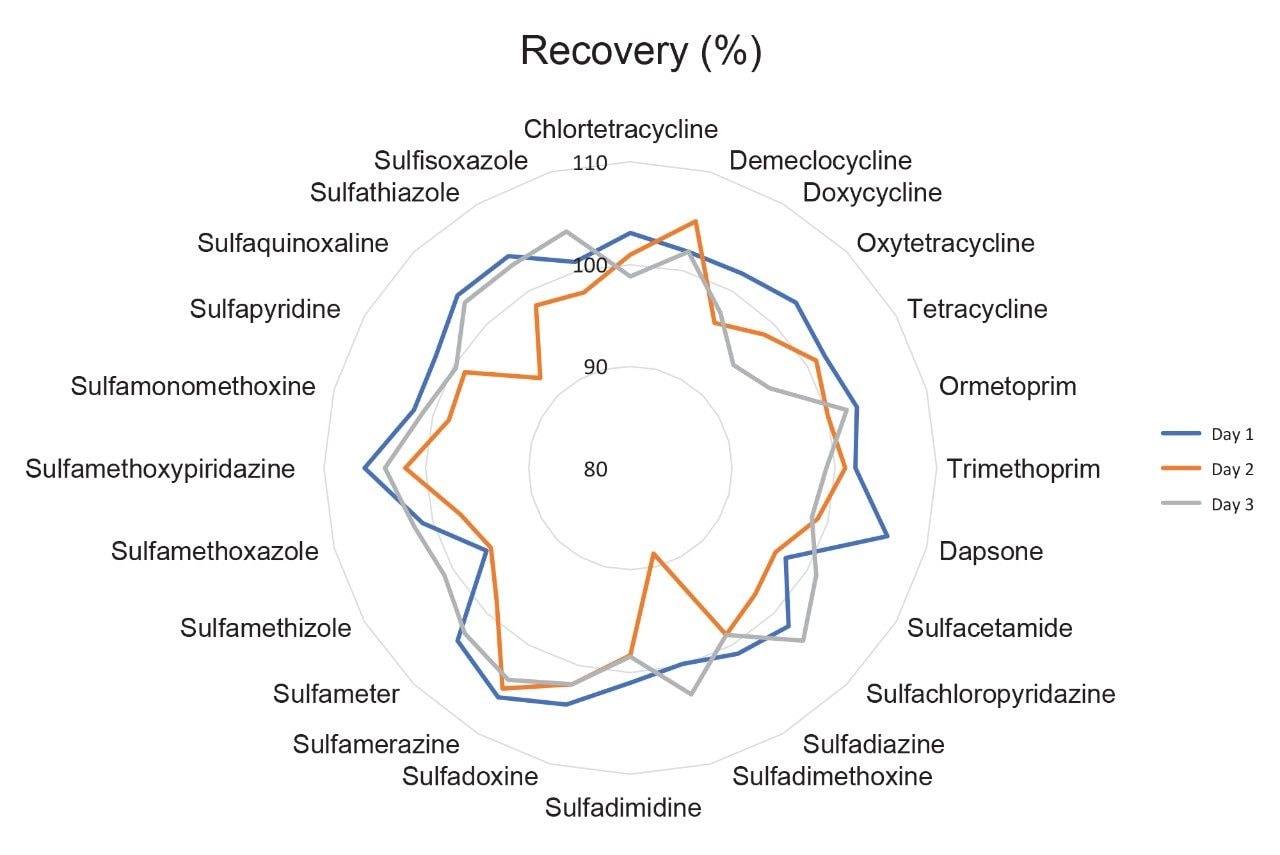
The trueness, expressed by measured recovery, was evaluated using the data from the analysis of the spiked samples over the three days. The mean recoveries for each set of seven spikes, at the three concentrations, prepared and analyzed over three days, were within the range 88.6% to 106%. As such, they were within the criteria set out in Commission Decision 2002/657/EC. The repeatability of the method was also satisfactory for all analytes in both RSDr (0.9%–10.2%) and RSDRL (1.1%–9.8%) studies. Trueness and repeatability are shown in Figures 5 and 6, and in Table 3, which also provides values for CCα and CCβ.

The method described here proved to be a sensitive and robust multiresidue method for the determination of a series of different antibiotics, namely tetracyclines, sulfonamides, trimethoprim, ormetoprim, and dapsone antibiotics, using an ACQUITY UPLC I-Class PLUS System coupled to a Xevo TQ-S micro MS/MS system. The method allows for a fast and reliable quantitation down to concentrations well below typical MRLs and was successfully validated according the European Commission Decision 2002/657, presenting satisfactory results for tetracyclines, sulphonamides, and related antibiotics in shrimp tissue. The procedure can also be applied to other animal and fish tissues after suitable validation. This cost-effective method can be easily implemented in routine testing laboratories, has been demonstrated as suitable for checking compliance with MRLs, and has the potential for screening at much lower concentrations, such as for food business operators’ due diligence testing.
720006789, Revised October 2020