In this application note, a flexible analytical system comprised of a Xevo G2-XS QTof with APGC and ACQUITY UPLC I-Class was used in conjunction with a well-defined informatics workflow to elucidate degradation markers in Li-Ion battery electrolytes in both the volatile and non-volatile chemical space.
The Li-Ion battery (LIB) is a key technology underpinning society’s increasing reliance on consumer electronics for telecommunications, computing, transportation, and navigation. Demand for LIB technology is set to increase with recent efforts from the automotive industry and sovereign governments to achieve sustainability targets by reducing carbon emissions and reliance on petroleum-based fuels.1 A transition to electric powered vehicles (EV’s) and away from internal combustion engines is one way to meet these sustainability targets.2 As a result, LIB manufacturers are facing increasing market demand for energy storage technology with higher capacitance, higher temperature tolerance, faster charging rates, and simpler (wireless) charging techniques, all in smaller, lighter units with increased robustness and safety.3
To successfully develop increasingly efficient technology, manufacturers require a thorough understanding of the chemistry occurring during the charge-discharge cycle that LIB technology employs. Information on the chemical composition of LIB materials and how that composition changes over time and under particular conditions aids manufacturers in increasing performance and quality of existing battery technologies. Specifically, determining the longevity and ultimate fate of proprietary additives is of particular importance.
This communication describes a protocol for determining the effect of lithium ion battery charge-discharge cycles on the composition of secondary battery electrolyte solutions and additives. The protocol utilizes a flexible, dual inlet analytical system comprising a single high-resolution mass spectrometer, the Waters Xevo G2 XS QTof, coupled to both GC and UPLC inlets via the Waters Universal Source architecture employing an APGC interface. This allows for the analysis and characterization of both volatile (analyzed by GC) and liquid extractable analytes (analyzed by LC) on a single system.
The insight into the chemistry that occurs during the charge-discharge cycle provided by this flexible system can aid secondary battery manufacturers to inform both iterative and paradigm shifting improvements in battery technology.
Li-Ion secondary battery electrolyte solution was prepared consisting of a dimethyl carbonate solution containing diethyl carbonate, ethyl-methyl carbonate, fluoroethyl carbonate, lithium hexafluorophosphate, and proprietary additives. The electrolyte solution was used to prepare secondary batteries that then underwent charge-discharge cycles at a fixed voltage. Electrolyte solution samples were extracted with dimethyl carbonate from batteries that had undergone:
Sample 1: 1 x charge-discharge cycle at fixed voltage
Sample 2: 40 x charge-discharge cycle at fixed voltage
Sample 3: 180 x charge-discharge cycles at fixed voltage
Sample 4: 200 x charge-discharge cycles at fixed voltage
Sample 5: 0 charge-discharge cycles (Negative control)
|
GC parameters |
|
|---|---|
|
Column: |
DB-5 MS, 30 × 0.25 × 0.25 |
|
Carrier gas: |
He |
|
Carrier gas flow rate: |
1.5 mL/min |
|
Inlet temp.: |
280 °C |
|
Injection mode: |
Split split ratio = 10:1 |
|
Initial temp.: |
60 °C |
|
Initial time: |
0.5 min |
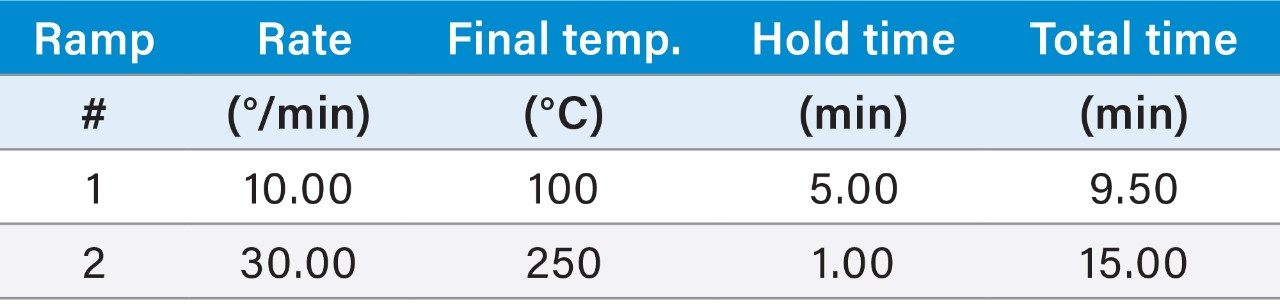
|
Column: |
HSS T3 2.1 × 100, 1.7 μm |
|
Injection volume: |
1.0 μL |
|
Mobile phase A: |
5 mM ammonium formate in water |
|
Mobile phase B: |
MeOH |

|
MS spectrometer: |
Xevo G2-XS QTof |
|
Instrument condition: |
APGC |
|
Corona: |
5 μA |
|
Sampling cone: |
15 V |
|
Source temp.: |
120 |
|
Gone gas: |
100 L/h |
|
Auxillary gas: |
450 L/h |
|
Instrument condition: |
LC-MS |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
2.5 kV |
|
Cone voltage: |
15 V |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
450 °C |
|
Cone gas flow: |
50 L/h |
|
Desolvation gas flow: |
800 L/h |
|
Informatics solution: |
UNIFI |
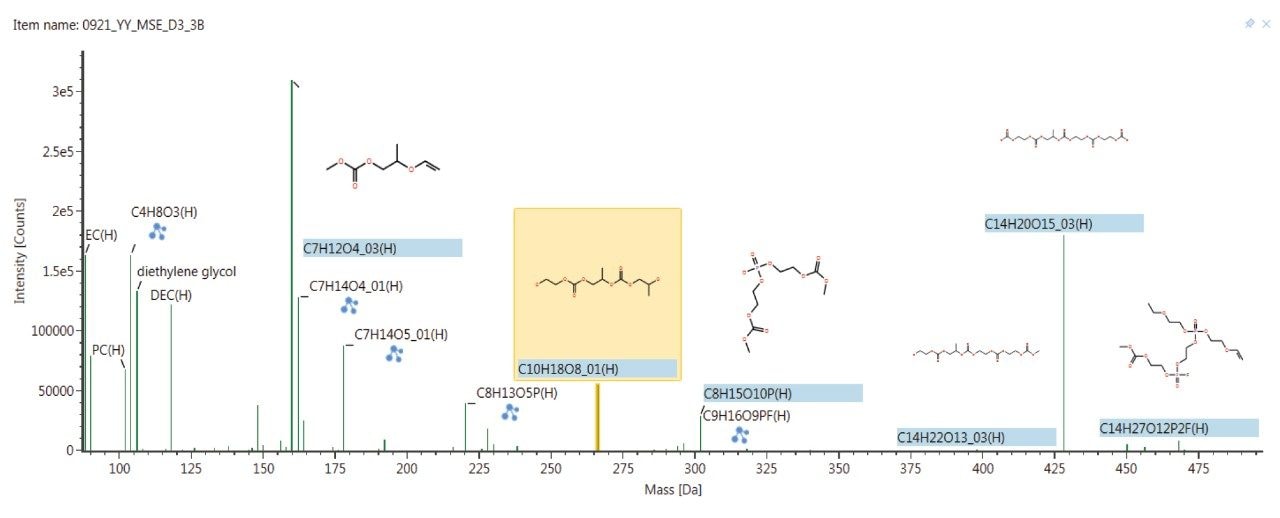
Since the components of the lithium ion battery electrolyte solution and additives were hypothesized to comprise both volatile and non-volatile chemical components, an APGC dual inlet source was used for the HRMS analysis. A comprehensive solution that could analyze both volatile and non-volatile components was deemed critical for an untargeted discovery workflow. A further challenge that was perceived is that the volatile chemical components were hypothesized to be labile, and a traditional electron ionization (EI) technique as employed in GCMS might lead to excessive fragmentation. Hence, the gentler ionization of APGC was selected in order to aid with structural elucidation. Specifically, APGC allows for the increased transmission of molecular ions over fragment ions compared to traditional electron ionization (EI) based GC-MS inlets. This molecular ion conservation allows greater selectivity and sensitivity since starting with a higher abundance ion. It also allows for structural elucidation of unknowns in a single run (as compared with EI GC-MS) by capturing spectral data on both molecular ions and fragments together. Figure 1 shows GC-MS data acquired in APGC mode on sample number one, comprised of extracted electrolyte solution exposed to a single charge-discharge cycle. Screening against an internal library of known electrolyte components, UNIFI was able to propose identification for a number of components in the sample based on accurate mass of molecular ions and fragments. EI GC-MS analysis of the same sample would have required further experiments such as repetition with Chemical Ionization (CI) – MS and fragment matching from external databases such as NIST to propose precursor structures due to the heavily fragmented nature of EI GC-MS spectra.
The major benefit of employing a dual inlet HRMS solution for workflows such as this is that it allows for immediate comparison, contrast and potential identification of both volatile (GC inlet) and non-volatile (LC inlet) components of samples analyzed. In the case of lithium ion battery development analysis via APGC is an effective strategy to gain insight into electrolyte solutions and their reaction products after charge and discharge. Analysis via LC-MS with ESI ionization will provide insight into the behavior of additives and other components during the charge-discharge cycle. GC/LC-MS data was acquired for all samples. An example of the total ion current (TIC) chromatograms acquired in both LC-MS and APGC MS mode comparing samples exposed to both 1 cycle and 200 cycles is shown in Figure 2. On examination of the chromatograms, it is apparent that the two complementary inlet techniques (GC and LC) capture information on discrete components within the samples across the non-volatile and volatile chemical space. The holistic data capture provided by the dual inlet system provides reassurance to researchers that changes in the performance of lithium battery technology can be extrapolated back to the correct changes in chemical composition of the electrolyte solutions and additive packages.
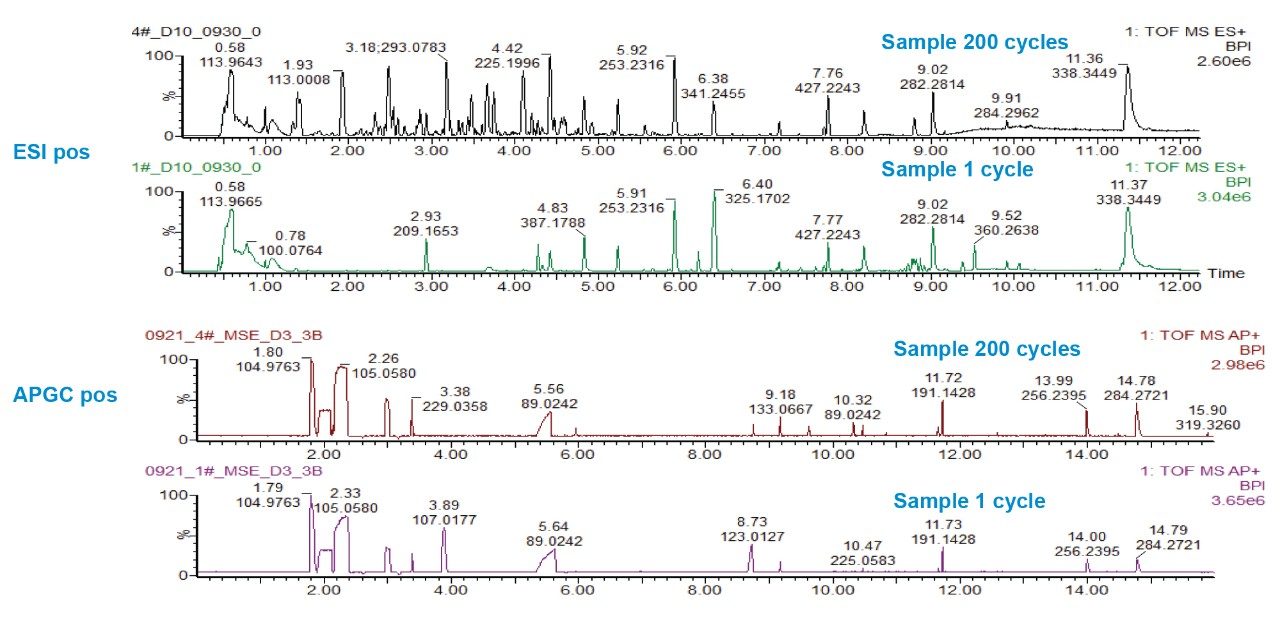
At first glance, there are obvious differences in the LC chromatogram on increasing charge cycles, particularly around retention times between 3–5 mins. The APGC chromatogram shows fewer initial differences on increasing charge cycles at first examination. For this reason, principal component analysis (PCA) is employed to determine differences to the samples not immediately apparent by visual inspection.
The first step in any attempt to determine the chemical effect of a physical process on a material is to show that a change has occurred to the material. One method of showing these changes is through principal component analysis (PCA). The PCA statistical technique groups similar samples together and dissimilar samples into separate groupings based on retention time, m/z, and ion intensity. In other words, the statistical method can help identify similarities and differences between sample injections that would not be readily apparent to the human eye by manual inspection. Figure 3 shows PCA results comparing electrolyte solution prior to the charge discharge – cycle(YY) to triplicate analyses of the samples that underwent 1, 40, 180, and 200 charge cycles (1#, 2#, 3#, 4# respectively).
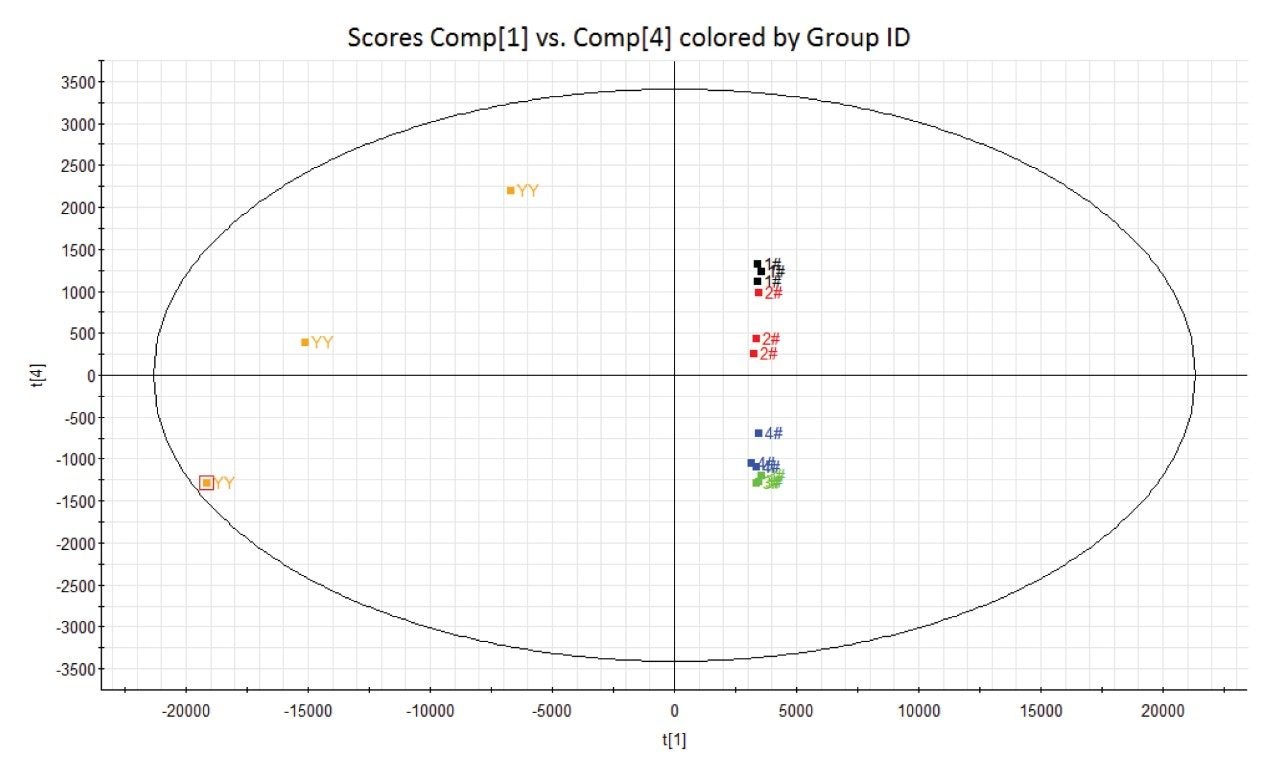
From the above PCA plot it can be inferred that there are indeed differences in the components that make up the samples based on the respective number of discharge cycles each sample underwent. It is also noticeable that the samples that underwent discharge can be placed into two distinct groups based on the repetition of discharge cycles the samples underwent, with those samples that underwent a low number (1 x and 40 x) being closely grouped in the upper right quadrant of the plot and those that underwent a high number (180 x and 200 x) closely grouped in the lower right quadrant.
The initial question is whether it is possible to identify some of the small organic molecule decomposition contaminants produced in the battery cell during the charge-discharge cycles. A basic workflow that outlines the approach made to determining which chemical species are either produced or diminished by increasing charge-discharge cycles is outlined in Figure 4.
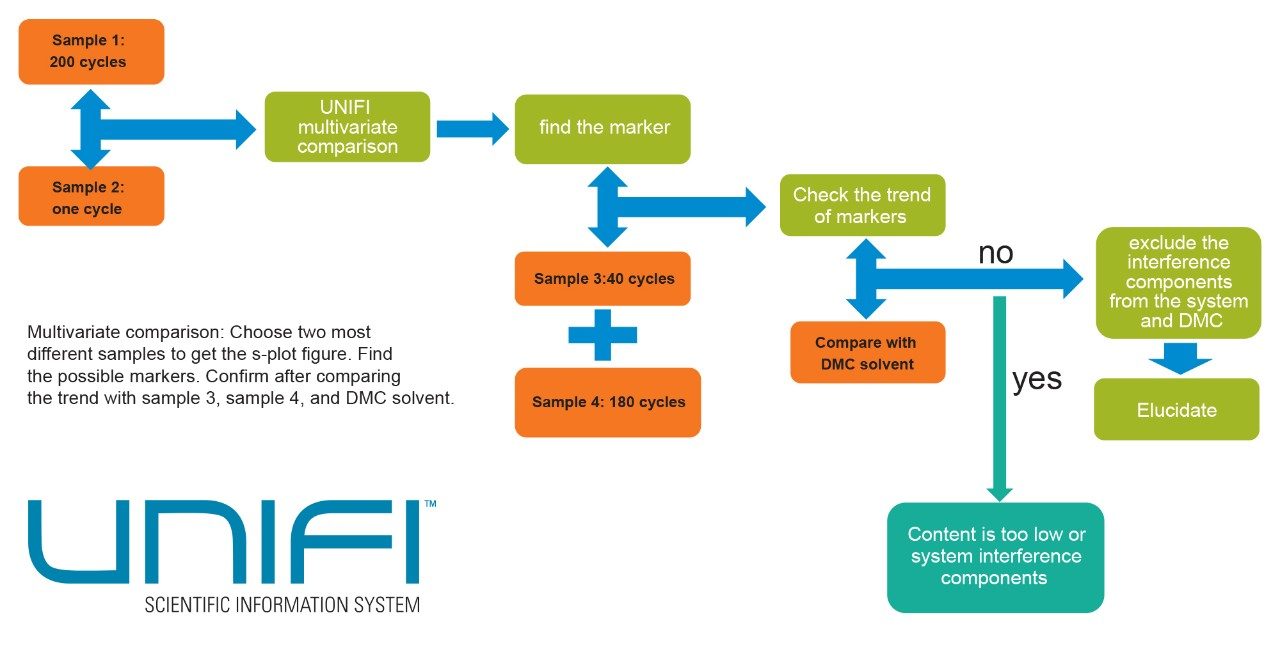
Having determined by PCA that each of the samples contained statistical differences, a second statistical method called orthogonal partial least squares discriminant analysis (OPLS-DA) was used to determine why the groups in the principal component analysis differed. The resultant s-plot from the OPLS-DA comparison between sample #1 and #4 is shown above in Figure 5.
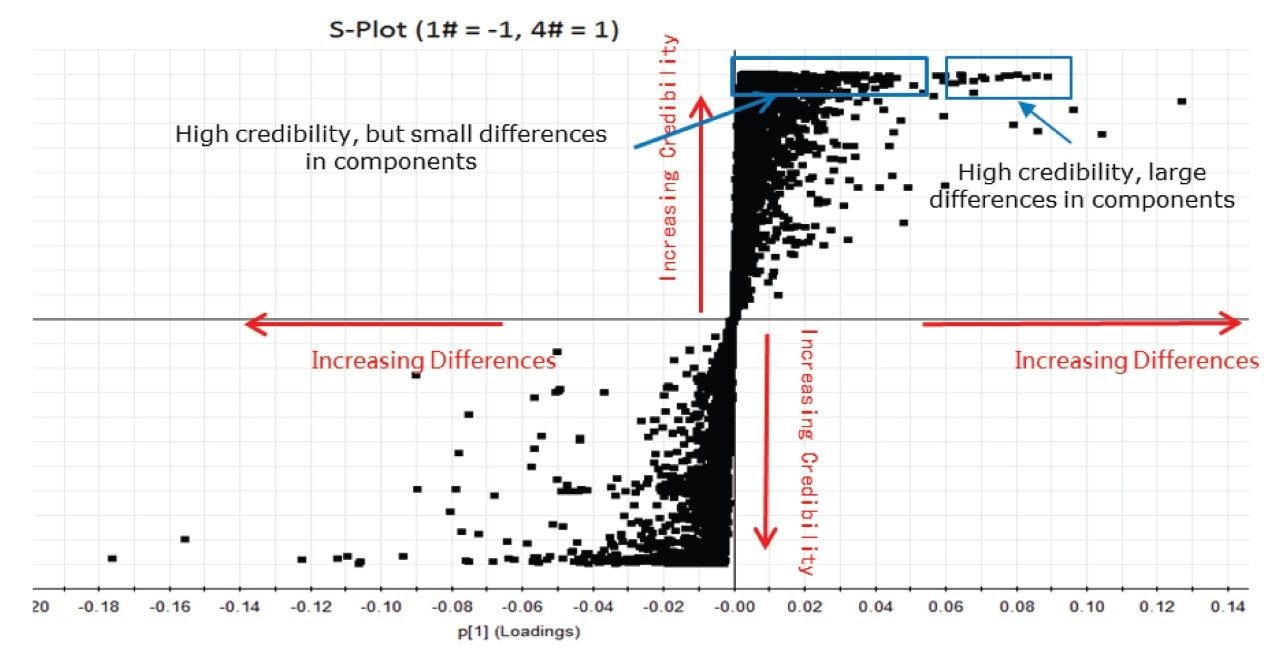
This plot helps pinpoint which components within the sample are contributing the biggest differences. This process can be automated with the UNIFI multivariate analysis workflow (Figure 6) to show what these components are using the trendplot function when they begin to appear in the electrolyte solution upon repeated charge-discharge cycles.
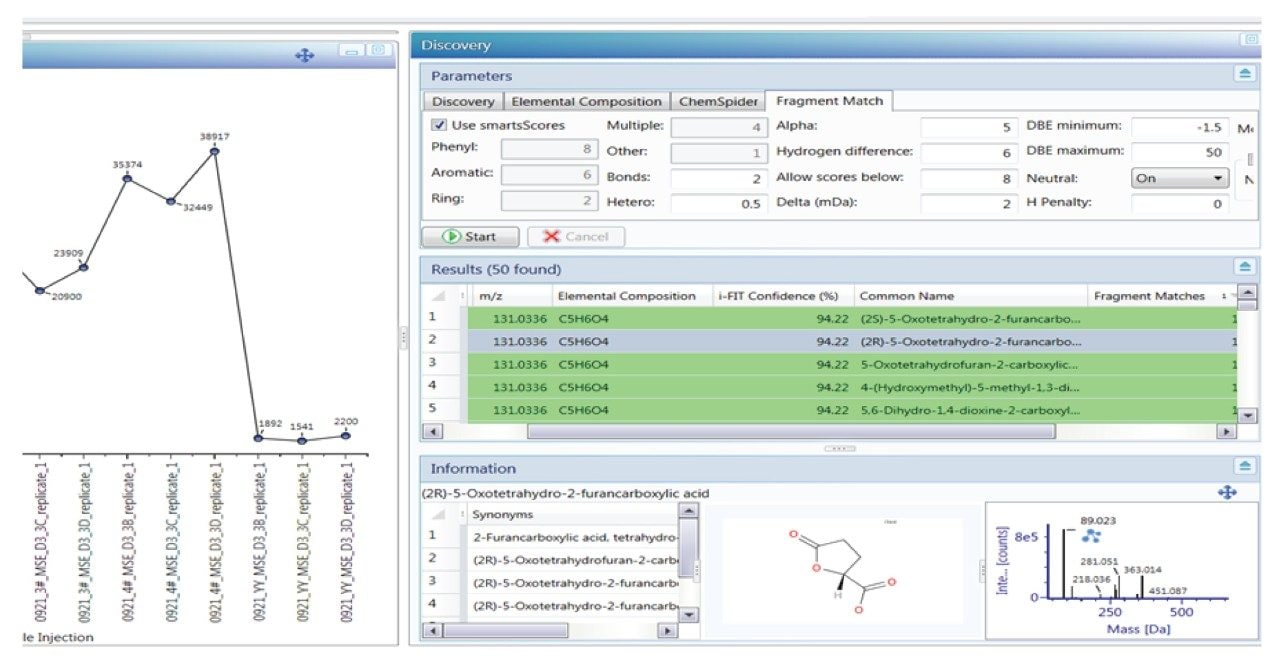
The Marker Summary window in the UNIFI workflow can be used to rapidly highlight the presence or absence of components over time. The Marker Summary window in Figure 5 shows that one component with m/z 131.0336 is absent from the electrolyte solution until after it has undergone 40 cycles. It then increases in concentration when the solution has undergone 180 cycles and increases further upon 200 cycles. The component is not present in electrolyte solution that has not undergone charge-discharge cycling (sample #5).
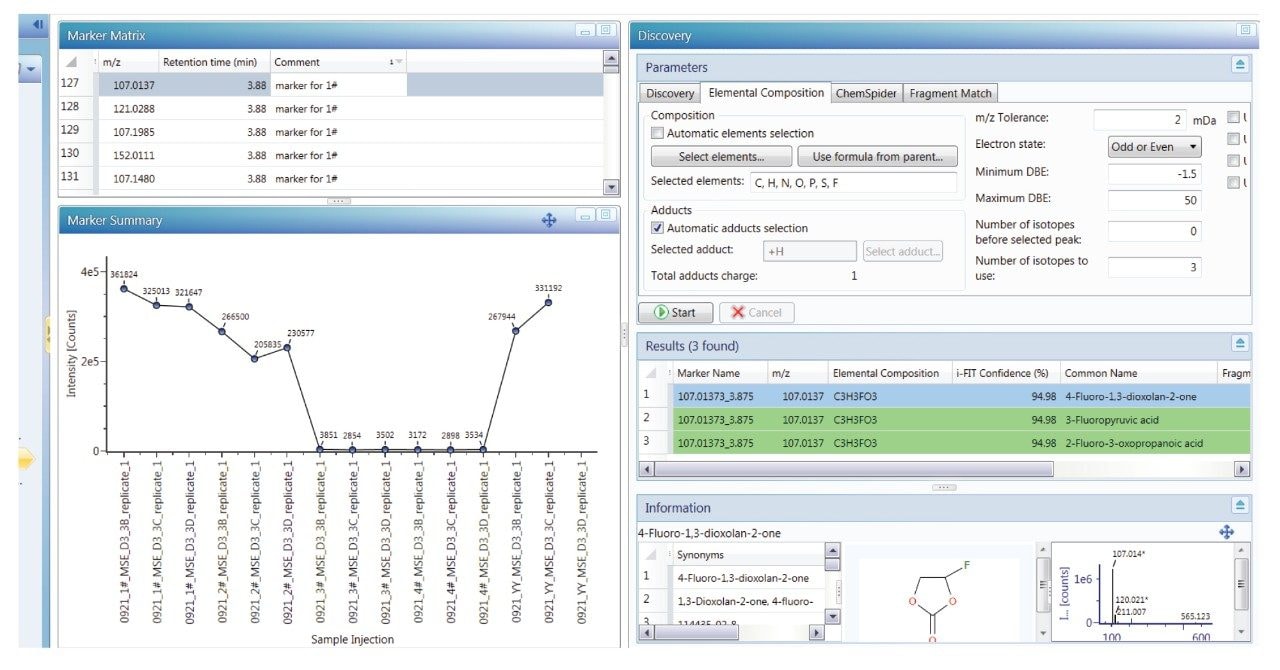
In addition to workflow tools that can pinpoint when a component begins causing changes in a physical process, UNIFI possesses tools that can aid in identification of those components.
Applying the discovery workflow tool to the component with m/z of 131.0336 led to a suggested elemental composition of C5H6O4 with 94% confidence based on isotope matching (i-Fit score, discovery results window, Figure 6). UNIFI can automatically search the Chemspider database (Royal Society of Chemistry) for matching chemical entities with the proposed elemental composition. In this case there is evidence to suggest that the component with m/z 131.0336 is a degradation product of fluoro-ethyl carbonate. The proposed structure represents replacement of the fluorine group in fluoroethyl carbonate (FEC) with carbon monoxide due to electrochemical reaction within the lithium ion battery (Figure 7A).
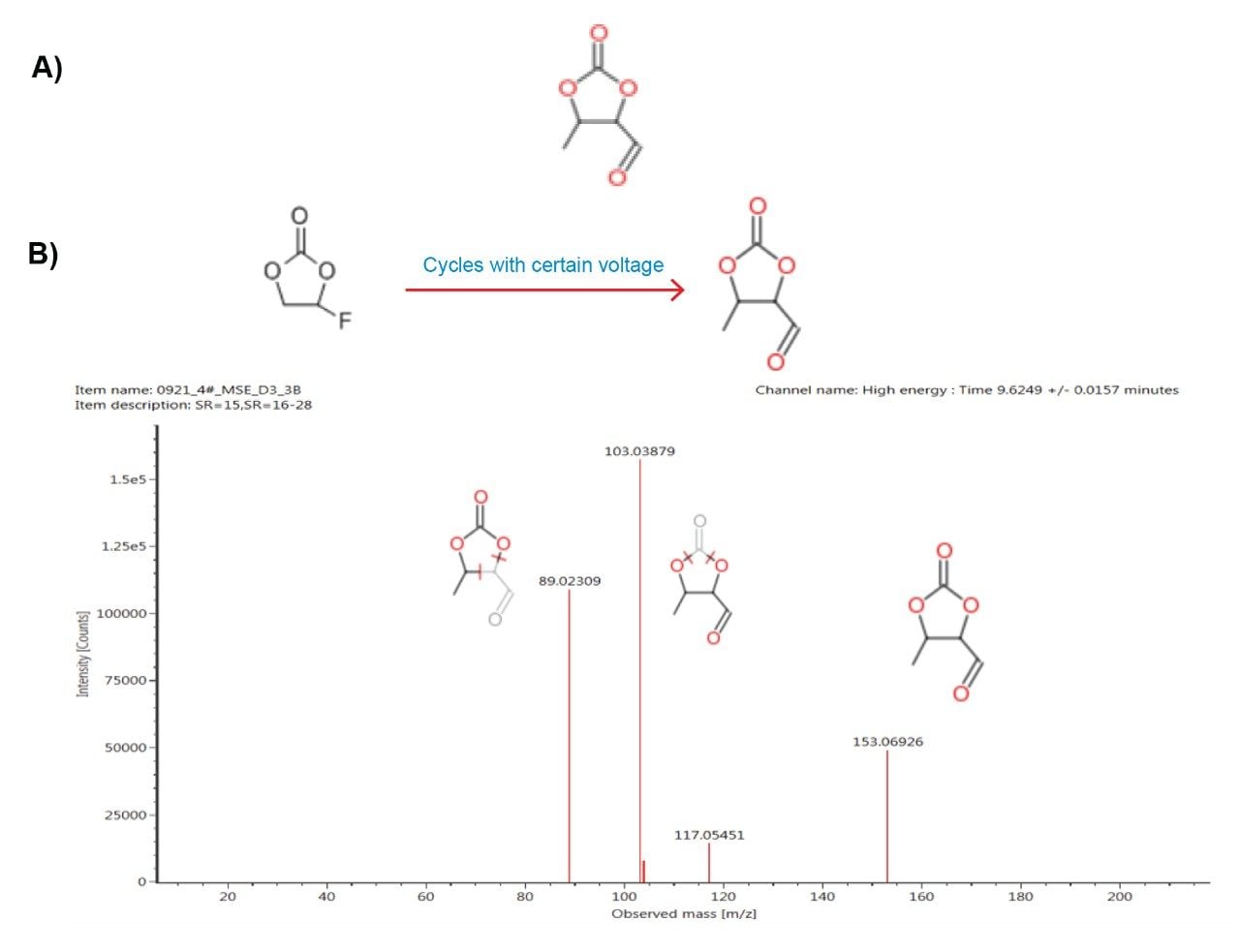
When running the massfragment workflow in UNIFI comparing to the proposed structure (Figure 7B) there is excellent correlation between the proposed structure and the fragment peaks in the high energy mass spectrum of observed component with m/z 131.0336.
Further evidence that this component is due to degradation of fluoroethyl carbonate is shown in Figure 8 highlighting decreasing concentrations of FEC at the same points in the cycle timeline as the unknown component increases.
Li-Ion Battery manufacturers require comprehensive information on the chemical composition of battery substrates such as electrolytes and additives to provide insight into the chemistry that occurs during the charge-discharge cycle. This knowledge will effectively direct research efforts towards improved energy storage technology.
In this application note, a flexible analytical system comprised of a Xevo G2-XS QTof with APGC and ACQUITY UPLC I-Class was used in conjunction with awell-defined informatics workflow to elucidate degradation markers in Li-Ion battery electrolytes in both the volatile and non-volatile chemical space.
Multivariate analysis in UNIFI was able to pinpoint potential organic impurity chemical markers that increase with increasing incidence of charging cycles on battery substrates.
Combining HRMS data, MSE, and powerful library and database searching offered by UNIFI enables the identification of structural elucidation workflow degradation markers. With this information available, a structure was proposed that enabled battery manufacturers to determine which components in electrolyte mixtures were implicated in battery lifecycle reduction. The information also helped guide direct research efforts into modifying or replacing these components to improve battery performance and reliability.
720006756, March 2020