
A variety of applications may require the use of relatively long, shallow gradients. The LC system ideally should deliver the same compositional gradient from one injection to the next, as well as from analysis to analysis, which may extend over a period of days to years depending on the aim of the analysis. This application note highlights the high performance level of the ACQUITY UPLC I-Class PLUS System, specifically the consistent gradient formation which enables the generation of reproducible retention times.
Repeatable retention time delivery of long, shallow gradients.
One of the goals of liquid chromatography method development is to develop a method which provides reliable and reproducible results. There are many factors that will affect the reproducibility of a method including, but not limited, to mobile phase composition, mobile phase pH, column packing material, gradient conditions, etc. The robustness of a method can also be impacted by design elements of the LC system itself. For example, methods which require the use of long and shallow gradients, or those that impart a small change in solvent composition per column volume, can be challenging. For these types of methods, a binary high-pressure mixing system where each solvent is delivered by a dedicated pump can deliver more reproducible gradients than ternary or quaternary low-pressure mixing systems which employ a gradient proportioning valve to generate the gradient.
A variety of applications may require the use of relatively long, shallow gradients. Some of these common application areas include biomolecule analyses, specifically peptide mapping1 and glycan analysis,2 as well as profiling of natural products including traditional Chinese medicines.3 This paper will evaluate the performance of a high pressure binary LC system for the generation of reproducible retention times for these types of separations. The LC system ideally should deliver the same compositional gradient from one injection to the next, as well as from analysis to analysis, which may extend over a period of days to years depending on the aim of the analysis. This study will highlight the retention time reproducibility of the ACQUITY UPLC I-Class PLUS System for applications which require the use of long, shallow gradients.
Waters MassPREP Enolase Digestion Standard (p/n: 186002325) was reconstituted in 100 µL of 0.1% trifluoroacetic acid in water (per the Care and Use Manuals). Samples and mobile phases were prepared daily.
|
LC systems: |
ACQUITY UPLC I-Class PLUS System with CH-A |
|
Detection: |
ACQUITY UPLC TUV Detector |
|
Sample: |
MassPREP Enolase Digestion Standard and MassPREP Peptide Mixture |
|
Column: |
ACQUITY UPLC Peptide, BEH C18 130Å 1.7 μm, 2.1 × 100 mm (p/n: 186003555) |
|
Column temp.: |
65 °C |
|
Mobile phase A: |
0.1% Trifluoroacetic acid in water |
|
Mobile phase B: |
0.1% Trifluoroacetic acid in acetonitrile |
|
Flow rate: |
0.200 mL/min |
|
Injection volume: |
10 μL |
|
Wavelength: |
214 nm |
|
Collection rate: |
10 Hz |
|
Needle wash: |
25/75 Mobile phase A/mobile phase B |
|
Seal wash: |
80/20 Water/methanol |
|
Chromatography software: |
Empower 3, FR2 |
Long, shallow gradients are typically used to resolve complex samples such as peptide digests. For these particular samples, each combination of protein, digest reagent, and method conditions, among other variables, will result in a unique peptide mapping fingerprint. Thus, a peptide mapping separation can be used to identify, track, or validate processes. In addition, the complexity of the resulting peptide digest will depend on multiple factors including initial protein size and digestion enzyme. For very complex peptide digests, where the separation of many peaks is required, run-to-run retention time reproducibility is of the utmost importance for tracking peaks.
However, the reproducibility of any method is dependent on many factors, including sample preparation, column, LC method conditions, as well as the LC instrumentation used. In order to focus on the impact of the LC instrument in this study, all other variables were controlled. For example, the sample consisted of a purchased standard and a single lot of columns were used for the study. To reduce the impact of solvent evaporation, low-evaporative bottle caps were used. Lastly, to ensure proper performance of the instrument, the system was qualified prior to the study.
As stated in the introduction, due to the complexity of peptide maps, LC methods typically consist of very shallow gradients run at low flow rates in an effort to separate as many components as possible, leading to improvements in characterization. In addition, conditions commonly start with a low % of organic solvent, often times less than 5%, to retain highly polar or early eluting peptides. It is commonly known that binary pumps, which utilize high pressure mixing, offer better compositional accuracy and precision than do their low pressure mixing counterparts. The compositional accuracy and precision of a binary pump depends only on the flow accuracy for each pump head. Conversely, for a tertiary or quaternary mixing pump, the compositional precision and accuracy depends on the ability of the gradient proportioning valve or apparatus to perform in a consistent and correct manner. For these reasons, the binary ACQUITY UPLC I-Class PLUS System was selected for the study.
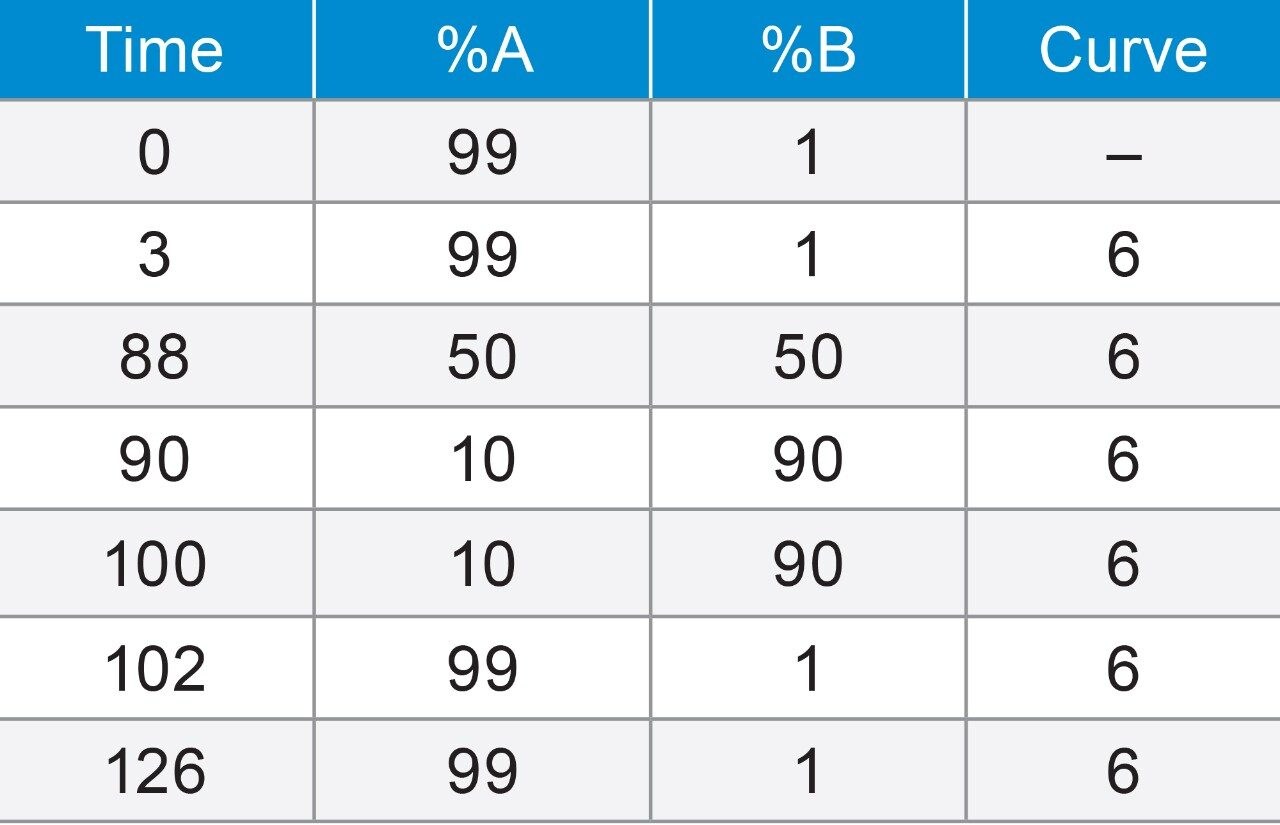
Prior to running analysis on the ACQUITY UPLC I-Class PLUS System, a generic peptide mapping liquid chromatography method was developed for the analysis of the MassPREP Enolase Digestion Standard. The method was designed to impart a 0.66% change in organic solvent per column volume of mobile phase delivered. A flow rate of 0.200 mL/min was employed to account for the relatively slow diffusion of peptides. Finally, to ensure maximum peptide map coverage, a gradient method was employed to start at 1% organic to retain highly polar or early eluting peptides. Because the conditions chosen include the use of TFA, an ion-pairing reagent, as an additive, columns were equilibrated by running the method gradient for 24 hours. Each individual batch was comprised of two blank injections (mobile phase A) followed by eight to twelve replicate injections of the reconstituted enolase digest (Figure 1).
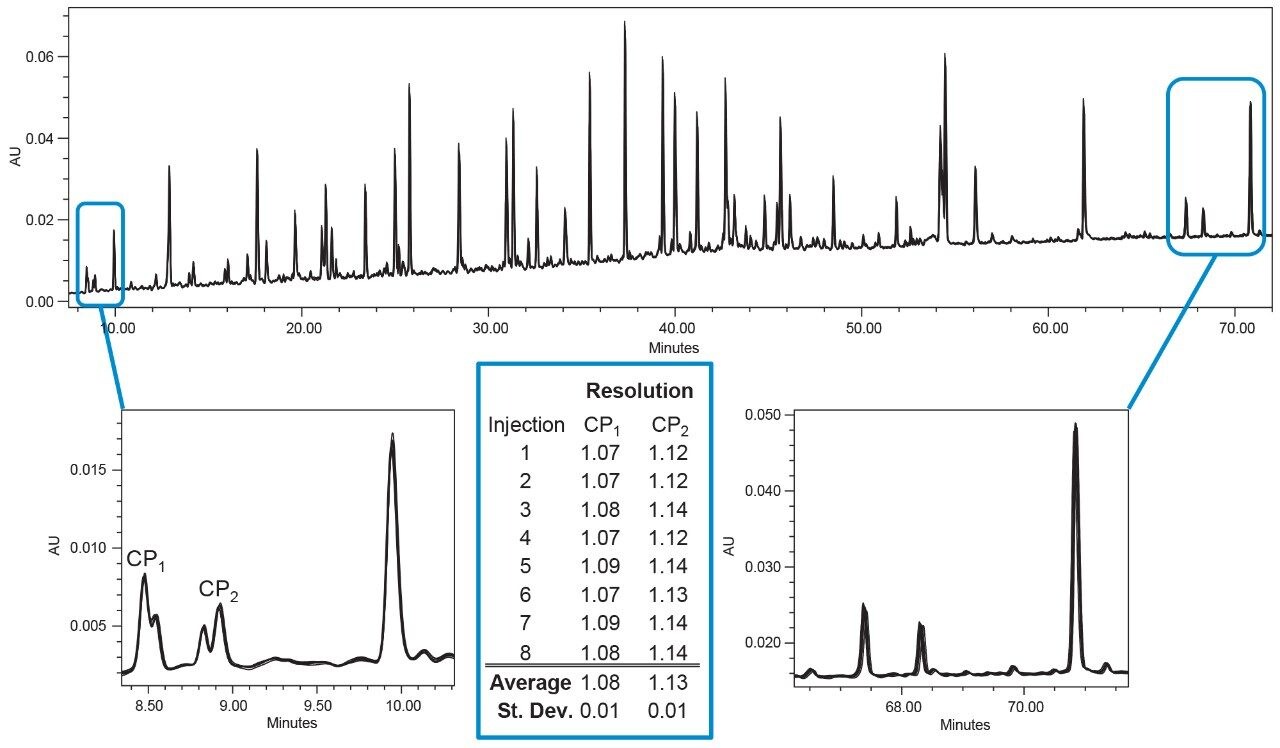
The ability of an LC system to provide precise and reproducible results over a number of injections is important not only for identification purposes, but also for quantitative purposes. For example, if gradient delivery varies slightly from injection to injection, small selectivity differences may appear. These can impact the resolution between peaks, particularly for critical pairs or peaks which are only partially separated. Peptide mapping is one example of an application area that requires separation and resolution of compounds with varying chemical properties. The retention of peptides can be greatly affected by a small change in % organic, and any compositional inconsistency of the pump between injections will be seen in the resulting retention time reproducibility. Table 1 shows the standard deviations obtained from the 8 replicate injections pictured in Figure 1.
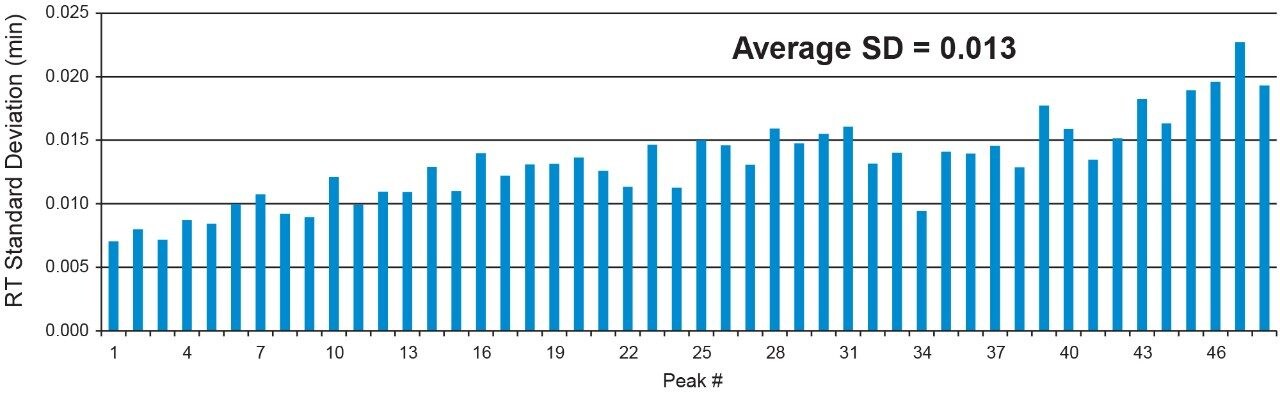
The very small standard deviation values highlight the consistent gradient delivery of the ACQUITY UPLC I-Class PLUS System. The average standard deviation across the 48 integrated peaks was calculated to be 0.013 min, or 0.78 sec, which displays the exceptional retention time reproducibility. In addition, by looking at the zoomed in portions of the chromatograms depicted in Figure 1, it is clear that there is no change in selectivity or resolution, so errors in identification or quantitation are highly unlikely.
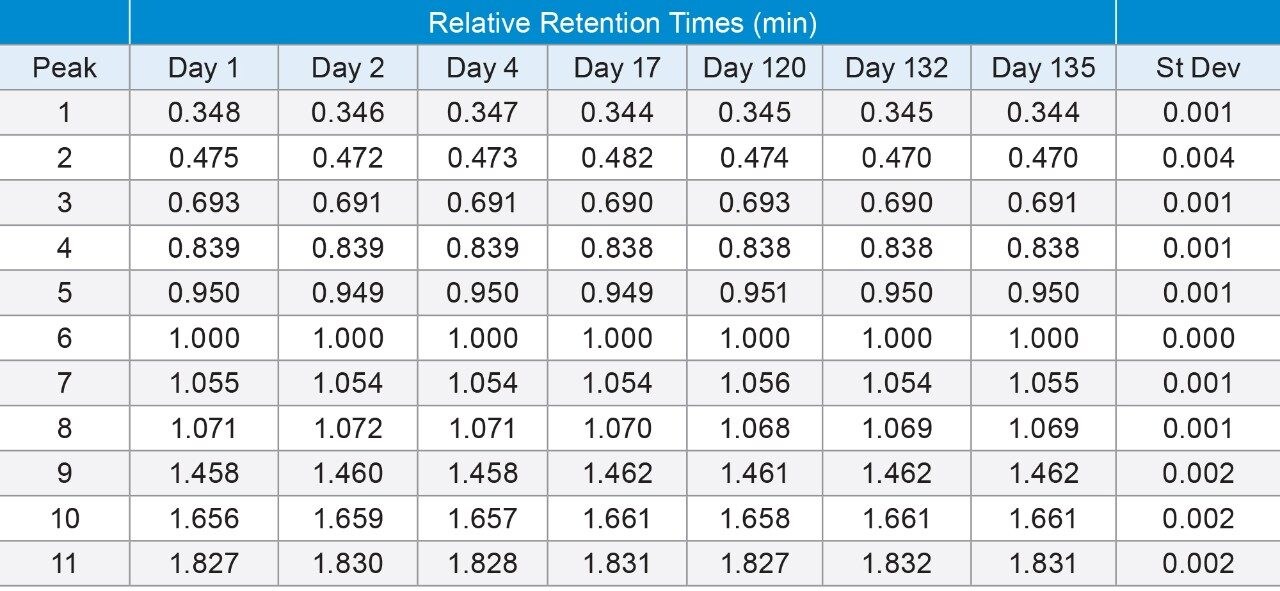
To highlight the reproducibility over extended periods of time, the same enolase digest and method were injected on seven separate days over the course of 135 days. Absolute retention times were not compared due to the use of different lots of standard, different columns, and slight differences in mobile phase preparation from one day to the next, which can all impact the resulting retention time. Table 2 shows the relative retention times (RRTs) of 11 representative peaks which span the entire elution range of the enolase digest standard. For example, peak 1 eluted at approximately 12 minutes, peak 6, which was used as the reference peak, eluted at approximately 37 minutes and peak 11 eluted at approximately 68 minutes. The largest standard deviation of the RRTs evaluated was 0.004 minutes, or 0.24 seconds. This data highlights the exceptionally consistent performance of the ACQUITY UPLC I-Class PLUS System and its ability to provide reproducible gradients over an extended range of time. This includes challenging methods where long, shallow gradients and low flow rates are needed to achieve optimal separations.
The performance and reproducibility of an LC system has the potential to impact peak identification, quantification and peak tracking when running an analytical procedure. Additionally, challenging method conditions, such as long, shallow gradients, may negatively impact retention time reproducibility. This study highlights the high performance level of the ACQUITY UPLC I-Class PLUS System, specifically the consistent gradient formation which enables the generation of reproducible retention times.
720006290, May 2018