This application note discusses the guidelines for the determination of compound recovery on Waters AutoPurification System.
Whether target compounds are isolated from naturally-occurring sources or chemically synthesized in multi-step strategies, the purification of intermediates and products, with high yield and purity, is crucial for the efficient completion of isolation protocols. Maximum compound recovery at each process step ensures a fast turnaround time and streamlines the overall product purification workflow. Although the calculation of recovery on a preparative chromatography system provides useful information about the compound synthesis and isolation strategy, several factors must be considered for accurate determination of sample yield. Factors which impact sample recovery include: system operating parameters, fraction collection parameters, compound peak shape, and the robustness of the analytical technique employed by the scientist performing the experiments. Any one of these elements, whether examined alone or in combination, may affect the compound recovery result. Guidelines for the determination of compound recovery on the Waters AutoPurification System are specifically discussed in this application note, but the fundamental principles for data acquisition and the accurate calculation of sample yield can be applied to any chromatographic system.
|
Analytical column: |
XBridge BEH C18, 4.6 x 50 mm, 5 μm (p/n 186003113) |
|
Analytical flow rate: |
1.46 mL/min |
|
Prep column: |
XBridge BEH C18, OBD Prep, 19 x 50 mm, 5 μm (p/n 186002977) |
|
Prep flow rate: |
25 mL/min |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
Methanol, neat |
|
Weak wash: |
95% Water: 5% Methanol |
|
Strong wash: |
95% Acetonitrile: 5% Water |
|
Makeup solvent: |
Methanol, neat |
|
Cone voltage: |
30 V |
|
Probe temp.: |
600 °C |
|
Ionization mode: |
ES+, centroid |
|
Sampling frequency: |
10 Hz |
|
Scan range: |
100–600 amu |
|
Wavelength: |
254 nm |
|
Gradients and injection volumes: |
as noted in figures |
|
Sample: |
Acetaminophen, 10.03 mg/mL |
|
Waters AutoPurification System: |
2545 Binary Gradient Module, 2767 Sample Manager, System Fluidics Organizer, 8–30 mL Flow Splitter, two 515 HPLC pumps, 2998 Photodiode Array Detector, SQ Detector 2; controlled by MassLynx Software |

Maximizing target compound recovery in product isolation reduces the time required to generate sufficient material for subsequent experiments, decreases solvent usage, and liberates personnel to perform other tasks to yield improvement in overall process efficiency. The chromatographic system must be configured properly and performing optimally before any compound isolation can commence. Even with a perfectly tuned system, however, several ancillary factors influence the calculation of sample recovery which include, but are not limited to, sample separation, column choice, peak shape, injection volume, and the quality of chemist’s analytical technique.
NOTE: This procedure was performed to demonstrate the calculation of compound recovery during the isolation process. All values are for illustrative purposes only and are not intended to provide system specifications. This protocol may be modified for use with a different sample solution, mass detector, column, or flow rate; however, the principles outlined remain constant irrespective of these changes.
From the MassLynx Help window located in the AutoPurification System software, select the “Search” tab, then type “testing system performance” in the keyword dialog box. Select this topic from the list and then click “Display”. If the system configuration has been changed, that is, the plumbing, tubing, or flow rate is different from the time when last used, then the pressure balancing and timing tests must be performed. If the timing test is completed, perform the chromatography performance test with the dye solution (p/n 716000765) to assess the accuracy of fraction collection. The resulting chromatograms should be similar to those shown in Figure 2. Adjust the fraction collection parameters, if necessary, to achieve comparable results.
A 10 mg/mL acetaminophen stock solution was prepared according to the following protocol.
Note: The exact values noted below were used for demonstration purposes only. The masses used in this procedure do not have to exactly match the ones that are obtained each time this procedure is performed. The values reported here are only meant to be a guideline for the preparation of the acetaminophen stock solution. It is important to note all masses and measures each time the procedure is executed.
A. Weighed approximately 250 mg acetaminophen into a 20 mL scintillation vial. (The mass used in this set of experiments was 250.87 mg.)
B. Prepared 100 mL of 95% water/5% methanol (mixed 5 mL methanol and 95 mL HPLC grade water).
C. Dissolved acetaminophen in ~22 mL of 95% water/5% methanol. Vortexed and sonicated the solution to completely solubilize the analyte.
D. Transferred the whole solution to a 25 mL volumetric flask.
E. Rinsed the vial 3 times with small amounts of diluent and added these rinses to the volumetric flask.
F. Brought the sample volume to 25 mL with additional diluent.
G. Inserted stopper into the volumetric flask and mixed well.
H. Calculated the acetaminophen stock solution concentration:
Acetaminophen concentration = Mass of acetaminophen/volume of diluent
For this example, then: 250.87 mg/25 mL = 10.03 mg/mL
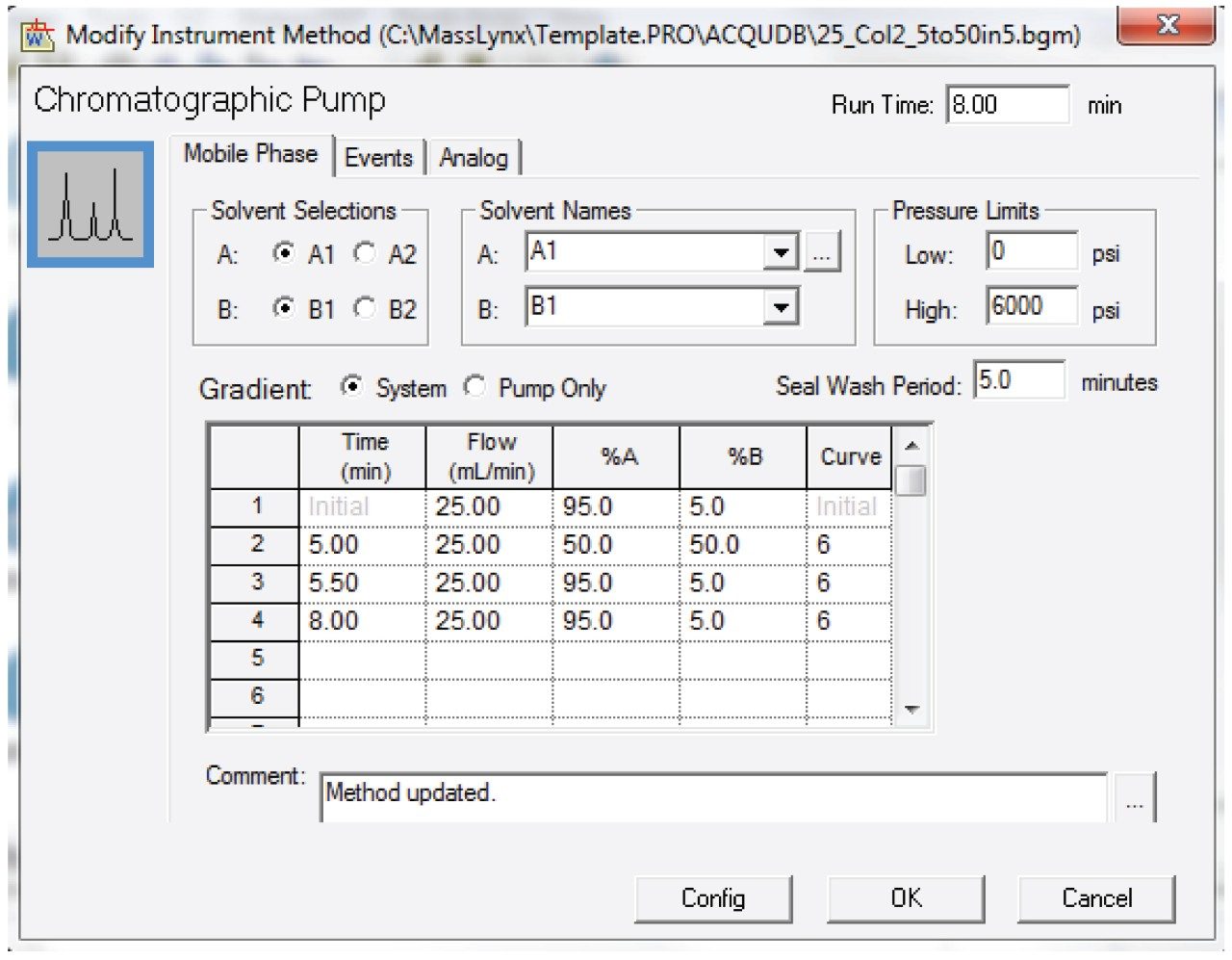
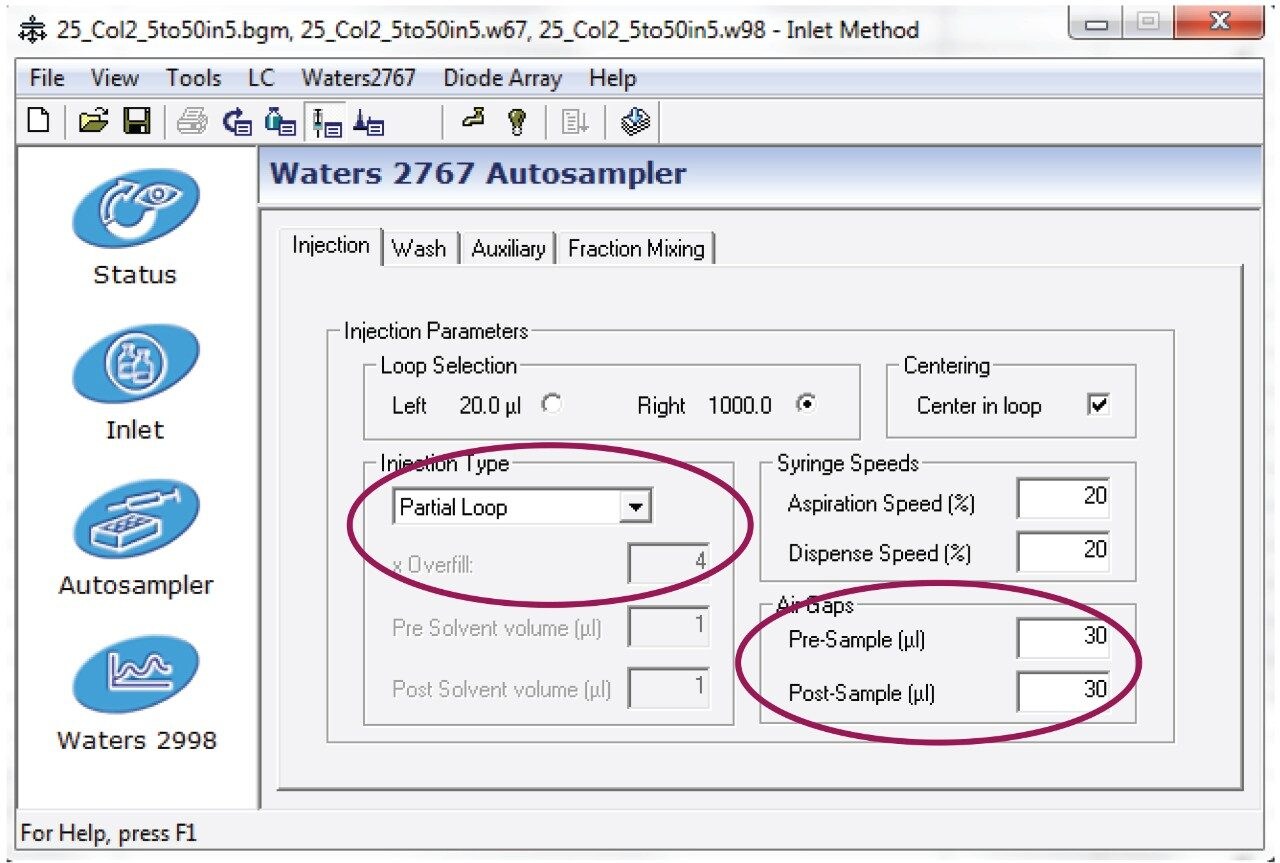
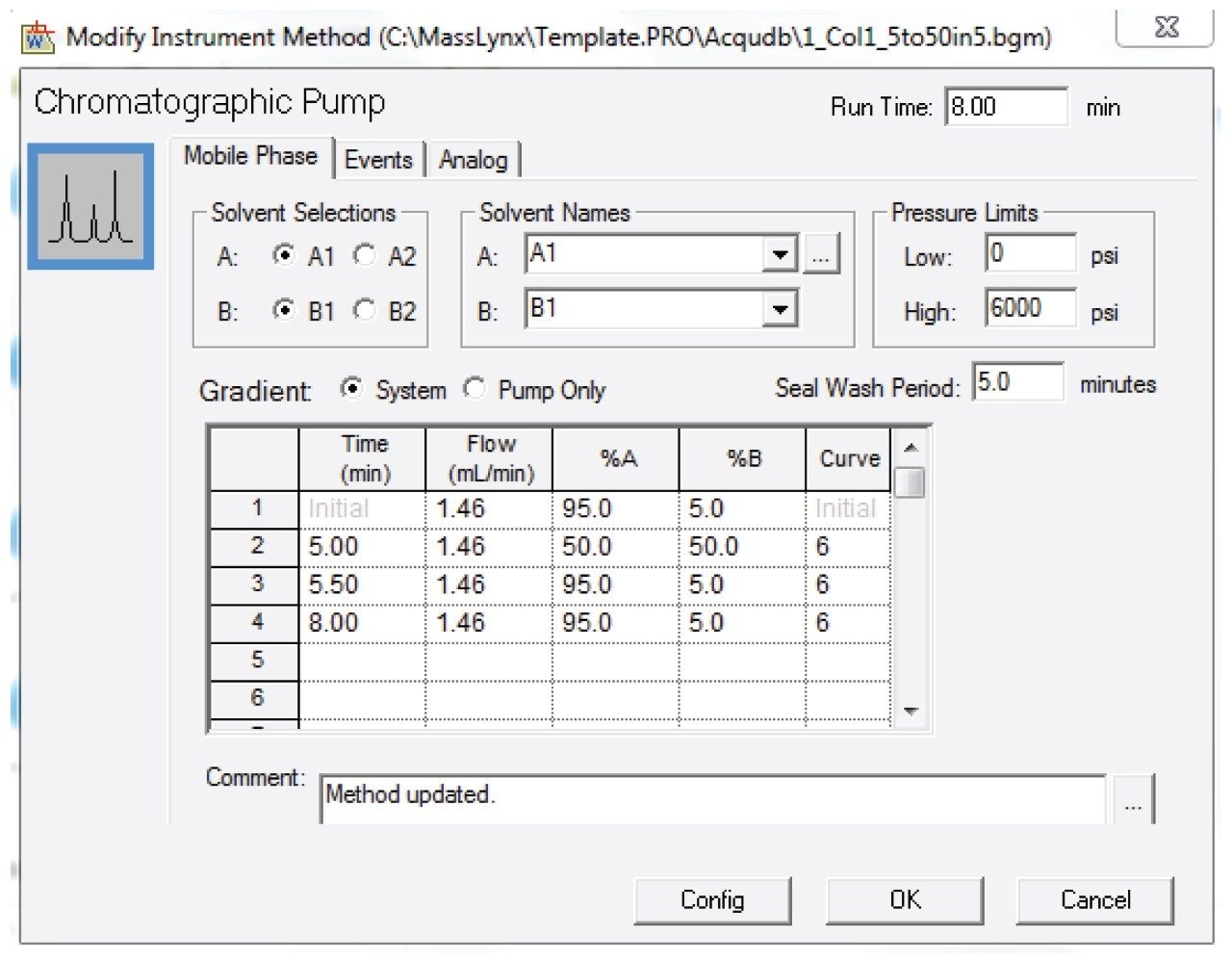
Entered the HPLC method as shown in Figures 3 and 4.
Note: This method was prepared for use on a 19 x 50 mm column. For columns with different dimensions, scale the method and the injection volume accordingly.1 Note that the injection type was partial loop, centered with 30 µL air gaps pre and post sample.
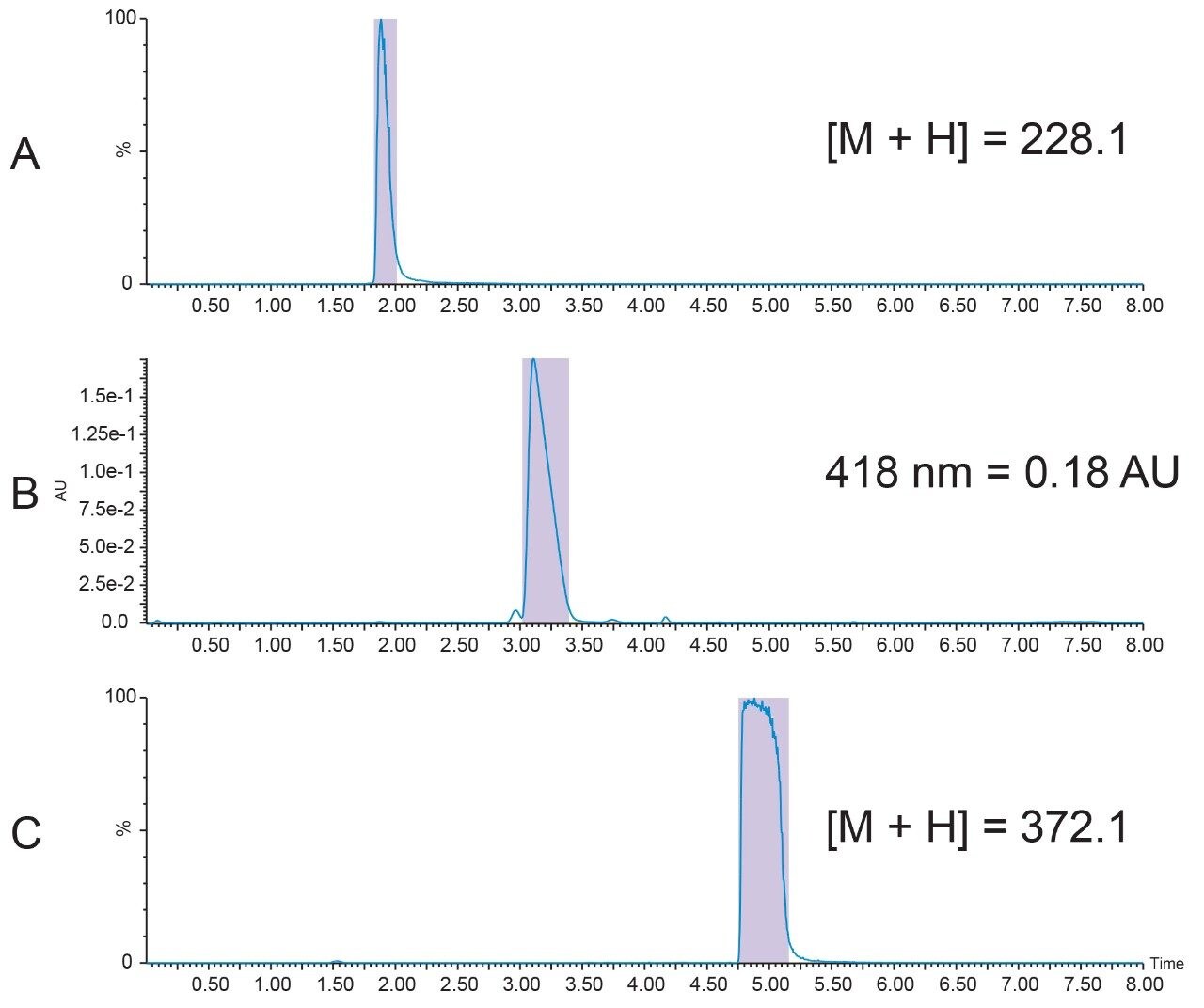
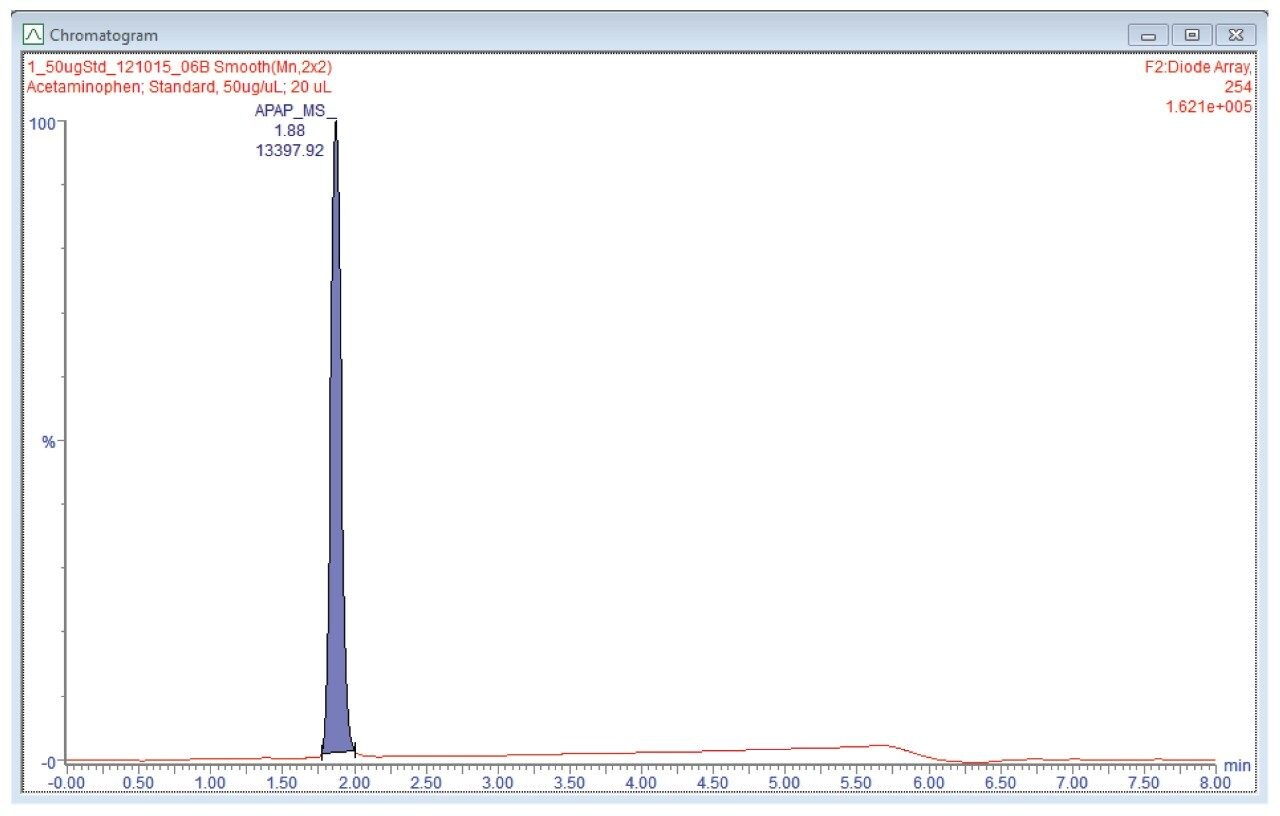
A. Collected the fraction by UV trigger at 254 nm. The chromatogram should be similar to the one shown in Figure 5.
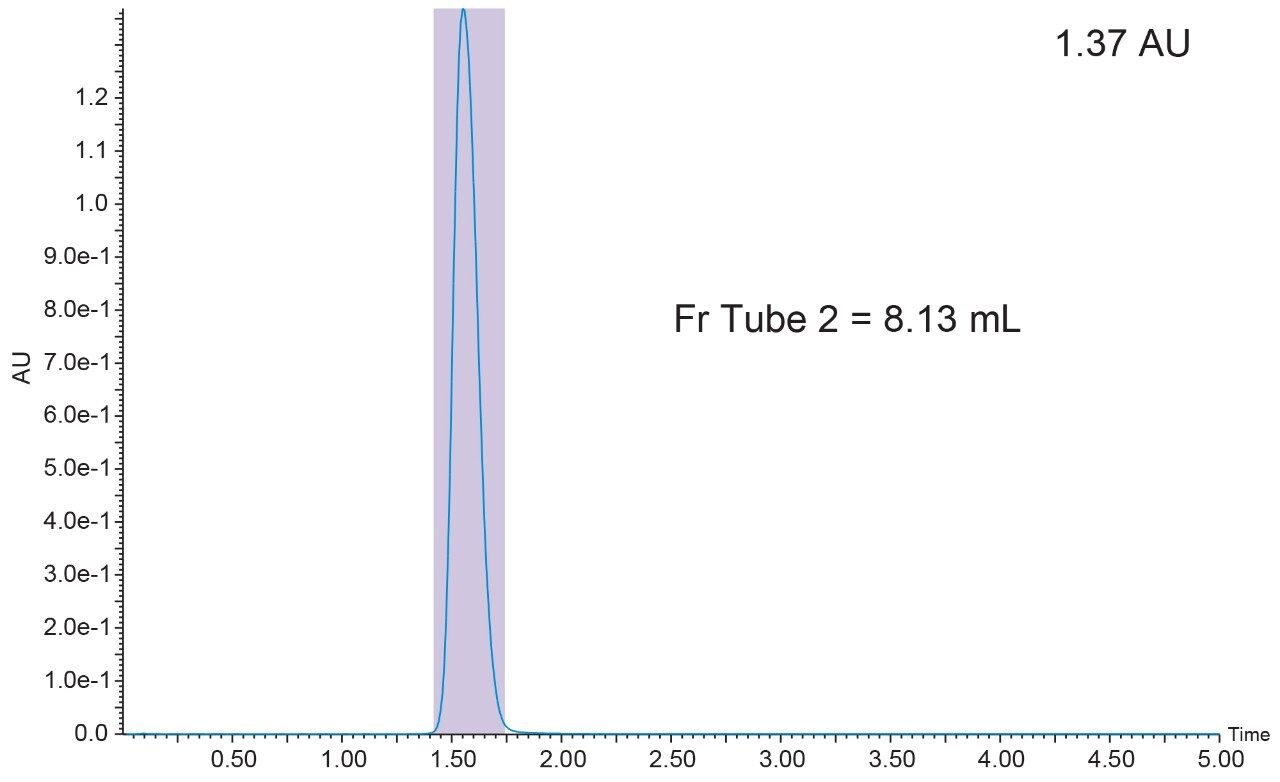
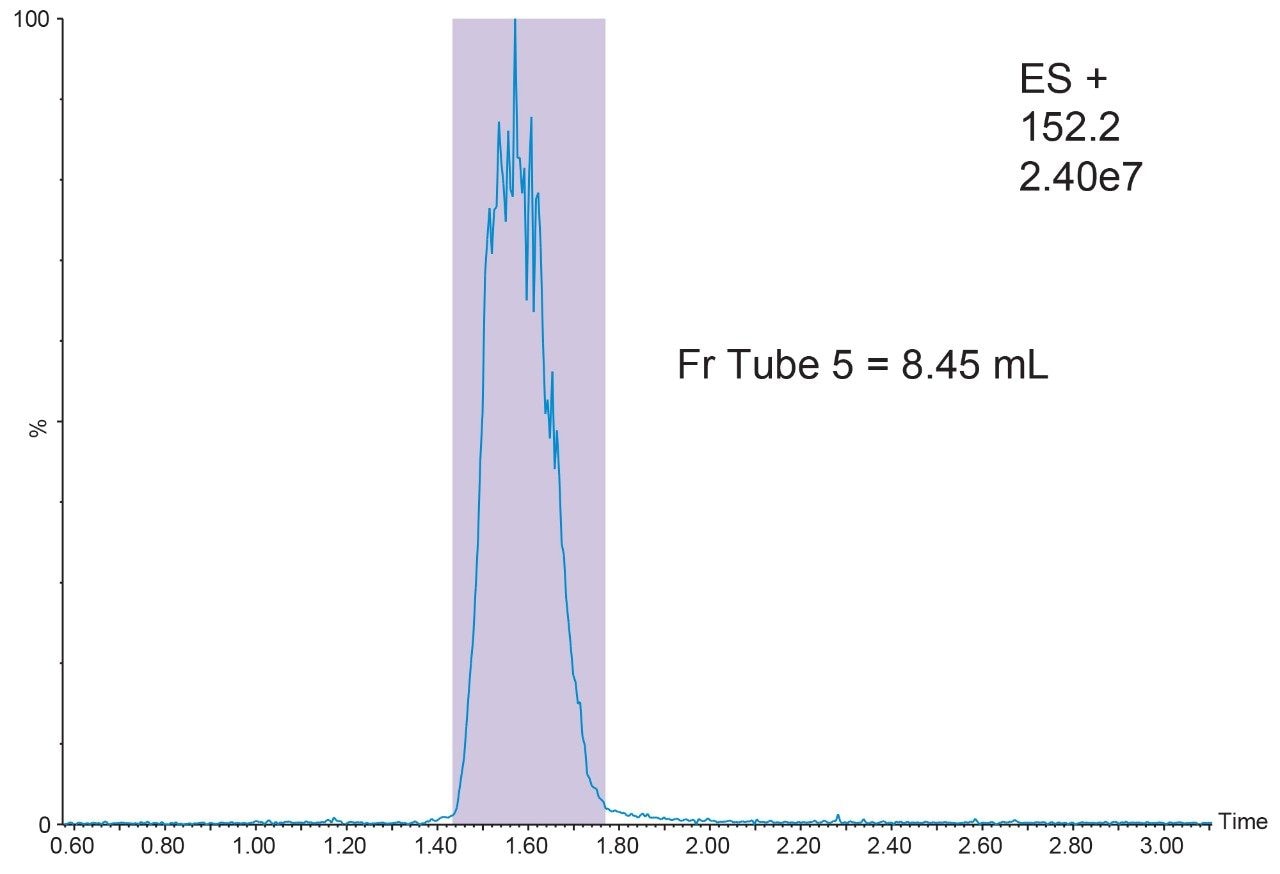
B. Injected another 200 μL of sample and collected the fraction by MS trigger (ES+, m/z = 152.2) in centroid mode.
The chromatogram should be similar to the one shown in Figure 6.
A. Transferred the full fraction collected by UV trigger to a 10 mL volumetric flask. Rinsed the collection tube with a small amount of diluent (95% water/5% methanol) and quantitatively transferred the rinsate to the volumetric flask.
B. Brought the volume to 10 mL with 95% water/5% methanol. Inserted stopper into the volumetric flask and mixed well.
C. Repeated steps A and B for the fraction collected by mass trigger.
D. Created a collection standard by diluting 200 μL of the 10 mg/mL analyte stock solution to 10 mL in a third volumetric flask and mixed well.
A. The 10 mg/mL acetaminophen stock solution is equivalent to 10 µg/µL.
B. Diluted 1 mL of the 10 mg/mL acetaminophen stock solution to 10 mL in a volumetric flask with 95% water/5% methanol to make a 1 mg/mL solution (equivalent to 1 µg/µL).
C. 1 mL calibration standards for injection were prepared:
50 µg/µL = 0.05 mg/mL; 1 mg stock/0.05 mg = 20x dilution
Diluted 50 µL of the 1 mg/mL solution to 1 mL.
(50 µL + 950 µL diluent)
100 µg/µL = 0.1 mg/mL; 1 mg stock/0.1 mg = 10x dilution
Diluted 100 µL of the 1 mg/mL solution to 1 mL.
(100 µL + 900 µL diluent)
200 µg/µL = 0.2 mg/mL; 1 mg stock/0.2 mg = 5x dilution
Diluted 200 µL of the 1 mg/mL solution to 1 mL.
(200 µL + 800 µL diluent)
500 µg/µL = 0.5 mg/mL; 1 mg stock/0.5 mg = 2x dilution
Diluted 500 µL of the 1 mg/mL solution to 1 mL.
(500 µL + 500 µL diluent)
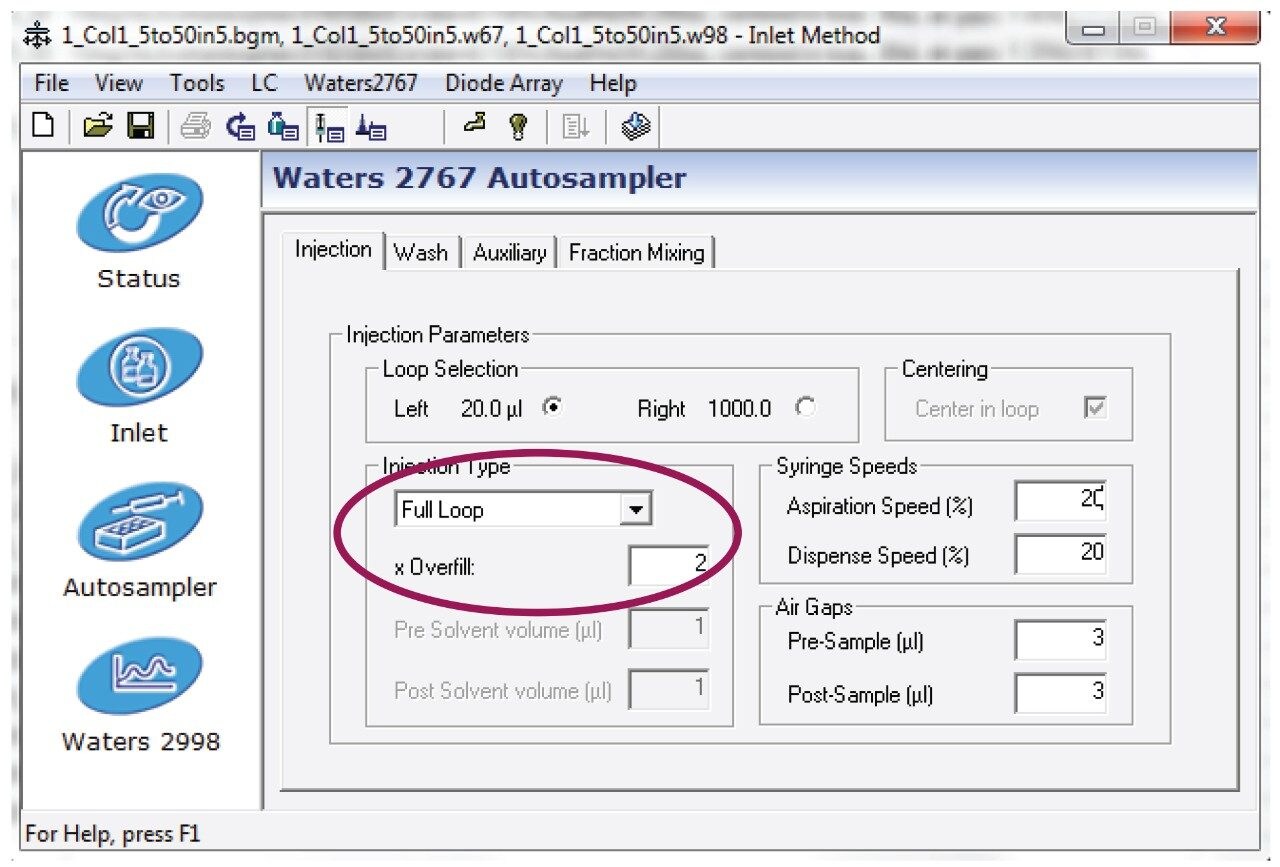
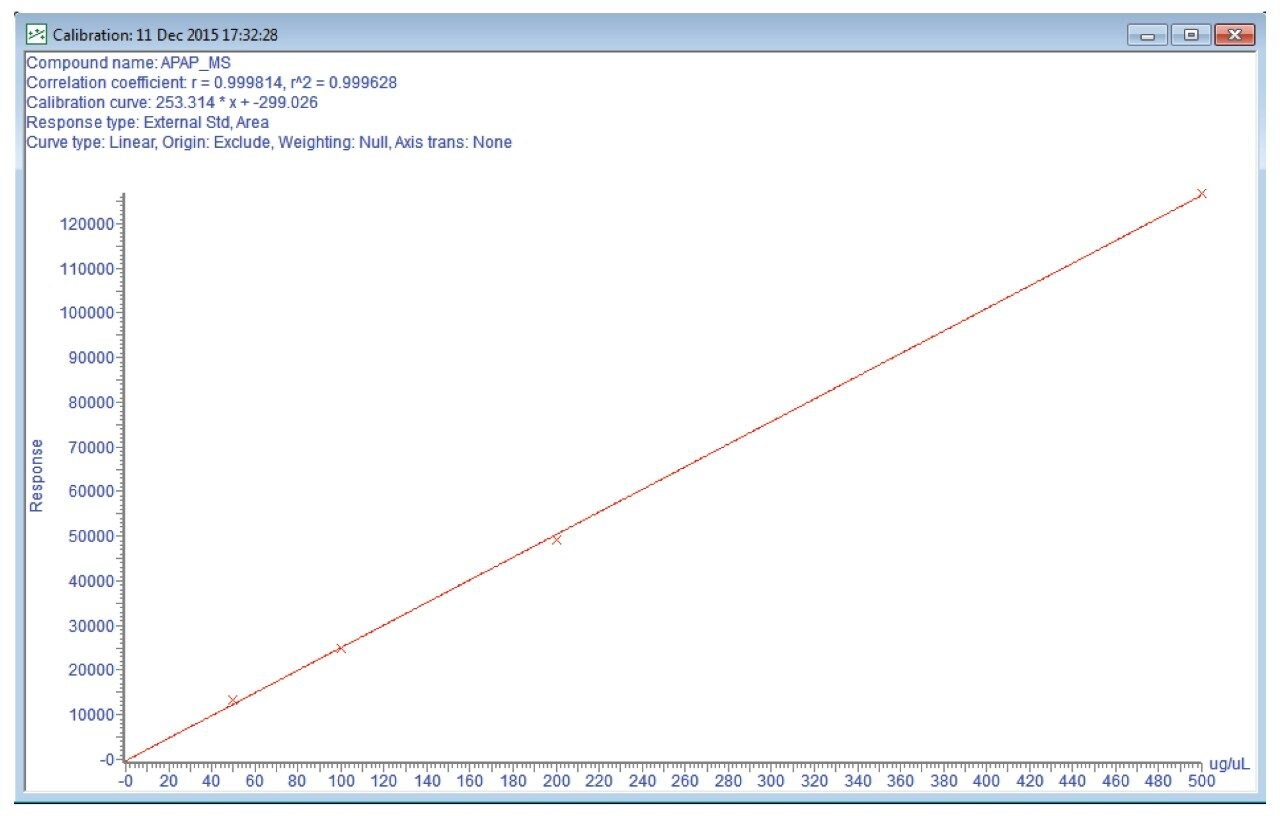
Entered the HPLC method as shown in figures 7 and 8.
Note: This method was prepared for use on a 4.6 x 50 mm column. For columns with different dimensions, scale the method and the injection volume accordingly. Note that the injection type was full loop with 2x overfill.
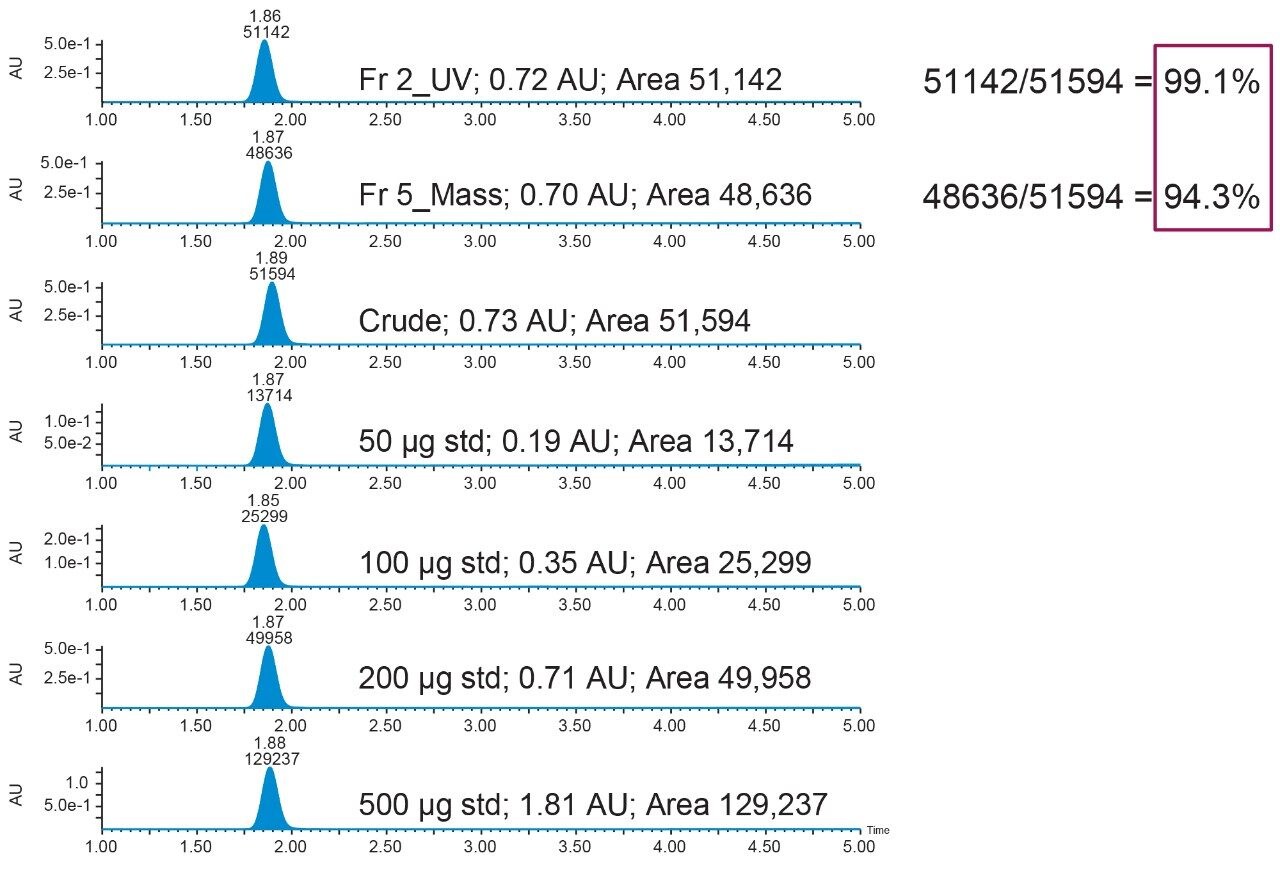
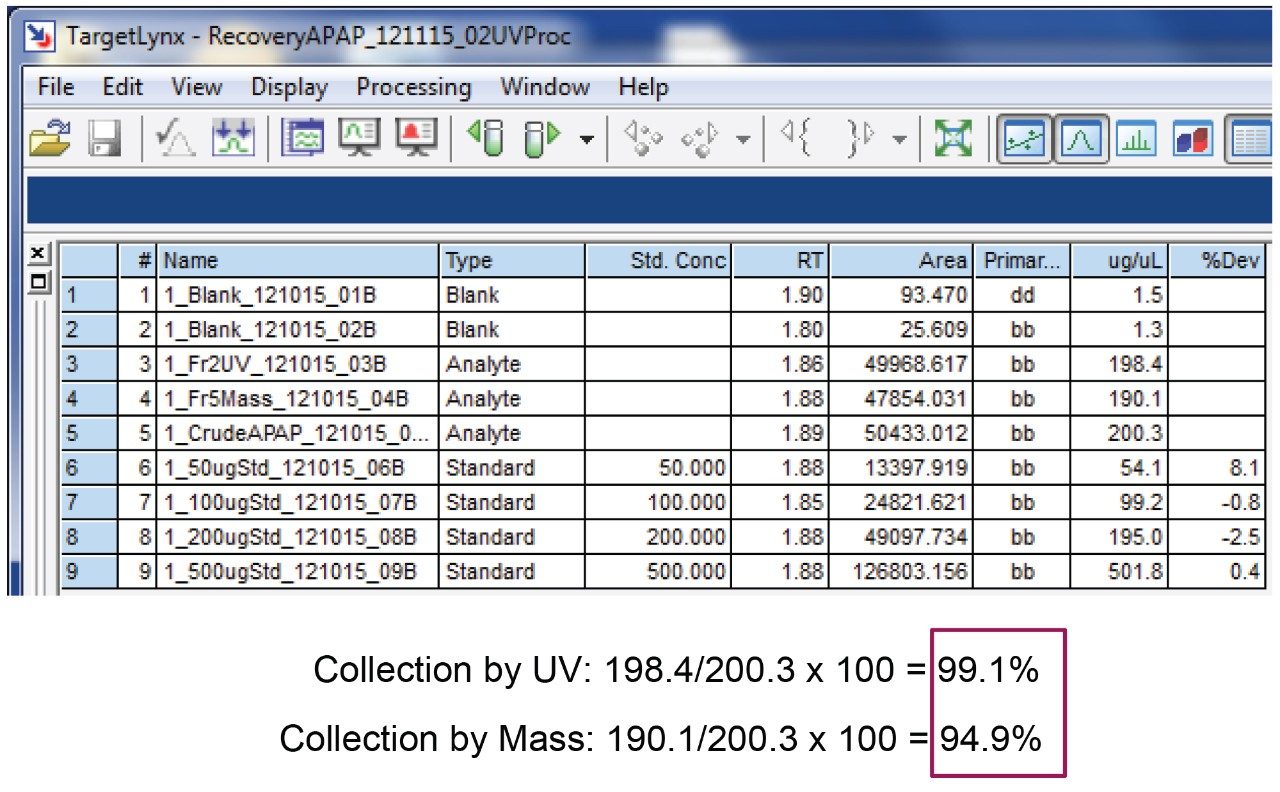
Calculated the recovery for the fraction collection using the following formula:
% Recovery = Area acetaminophen in fraction/Area acetaminophen in collection standard x 100. The chromatogram labelled "Crude" is considered the collection standard in figures 9 and 10.
Results for this experiment are shown in figures 9 and 10.

Several factors may impact the calculated recovery on a chromatographic system.
A. System configuration: Ensure that appropriate methods specific for the mass detector and/or UV detector are used for data generation.
B. Timing: Ensure that the system is timed correctly for fraction collection at the specified purification flow rate.
C. Plumbing: Ensure that the tubing diameter and flow rate is appropriate for the specified purification flow rate.
D. Sample loop: Inject no more than approximately half the installed loop volume and use sample loops with narrow ID tubing for reduced band broadening.
E. Column: Ensure that the sample is sufficiently retained and elutes with good peak shape with defined peak start and peak termination points for unambiguous integration. Samples that tail chromatographically are not recommended.
F. Inject port: Ensure that the needle depth is set to the optimum depth to avoid sample loss during injection.
G. Passive splitter configuration: Ensure that the Waters splitter is appropriate for the purification flow rate and that it is pressure balanced.
H. Flow to the mass detector probe: Ensure that the flow to the probe is optimized for the mass detector configured in the system.
A. Injection parameters: Ensure that the options selected are the ones that will be used in the isolation process. The recommended injection parameters for establishing baseline recovery on the system include partial loop, centered, with 30 μL air gaps preand post- sample for prep, and full loop with 2x overfill for analytical runs.
B. Peak start/terminate: Optimize the settings for each detector in the system.
C. Minimum intensity threshold: Optimize the setting for each detector in the system.
A. Use only excellent quality, calibrated pipettors/volumetric flasks for sample handling.
B. Ensure the efficient quantitative transfer of analyte by rinsing tubes, etc., with a small amount of diluent and accurately transferring the rinsate to the receiving vessel.
C. Dilute all samples “up to” the calibration line on the volumetric flask. The bottom of the meniscus should touch the calibration line.
D. Ensure adequate mixing to assure sample homogeneity prior to analysis.
E. If desired, consider performing multiple injections for a more complete statistical analysis of the samples and standards.
720006140, November 2017