This application note demonstrates the benefits of combining solid phase extraction (SPE) methodology of silica, UltraPerformance LC, and a moderately sensitive tandem quadrupole mass spectrometer, ACQUITY TQD for the development of a method for quantification of quetiapine in plasma. In addition to addressing the challenge of achieving medium to high sensitivity without using a high end TQ mass spectrometer, this method addresses several other challenges faced by the bioanalytical scientist, such as robustness, high throughput, and high selectivity.
Enables the bioanalytical scientist to address several challenges, such as medium tohigh sensitivity of a sample without using high end TQ mass spectrometers, robustness, high throughput, and high selectivity.
Quetiapine (2-[2-(4-dibenzo[b,f ][1,4]thiazepin-11-yl-1-piperazinyl)ethoxy]ethanol fumarate (2:1 salt)) is an atypical antipsychotic drug with a unique receptorbinding profile that belongs to a new chemical class, the dibenzothiazepine derivatives. Quetiapine is used for the treatment of schizophrenia and acute manic episodes associated with bipolar 1 disorder. The antipsychotic effect of Quetiapine is hypothesized to be mediated through antagonist activity at dopamine and serotonin receptors. Quetiapine also has an antagonistic effect on the histamine H1 receptor. These antipsychotics have a low incidence of extrapyriamidal side effects and tardive dyskinesias compared to older antipsychotics.
The advantages of the therapeutic profile of quetiapine have led to increased use in the clinical practice, which encourages the development of new pharmaceutical preparations. As a consequence, there are increased demands for new analytical methods to determine pharmacokinetic parameters in bioequivalence studies. Some of these methods could also be used in therapeutic drug monitoring. Quetiapine binds strongly to protein, which when combined with its low bio-availability (~9%), requires high dosage levels. It is metabolized by the liver and appears to be a major circulating species in plasma. Such high dosage strengths require development of an LC-MS assay to determine quetiapine in the range of 150 pg/mL as LLOQ (lowest limit of quantitation). Although several HPLC methods for the determination and/or quantitation of quetiapine have been reported, none of these methods are sensitive enough to address the determination in the expected drug levels. In this application note, ACQUITY TQD and Waters sample preparation chemistries were used for bioanalysis of quetiapine within a range of 150 pg/mL to 150 ng/mL.
Quetiapine in spiked plasma samples was extracted using solid phase extraction (SPE) using an Oasis HLB (3 cc, 60 mg) Cartridge. A 300-μL aliquot of plasma was diluted with 4% o-phosphoric acid, and loaded onto the SPE cartridge previously conditioned with organic solvent and water. The plasma solution was then washed with water followed by an organo-aqueous solution, eluted in aqueous basic buffer, followed by a wash with acetonitrile. The eluted samples were injected on to the system directly.
|
LC system: |
ACQUITY UPLC |
|
Column: |
ACQUITY UPLC BEH C18 1.7 μm, 150 mm x 2.1 mm |
|
LC column elution: |
90% organic solvent, 10% basic aqueous buffer over 3 min followed by a high concentration aqueous wash in the post run phase for 2 min, then the elution conditions were changed back to initial isocratic conditions |
|
Column temp.: |
40 °C |
|
Flow rate: |
0.275 mL/min |
|
MS system: |
ACQUITY TQD |
|
MS mode: |
ESI positive |
|
MRM transition: |
383.59 → 337.32 |
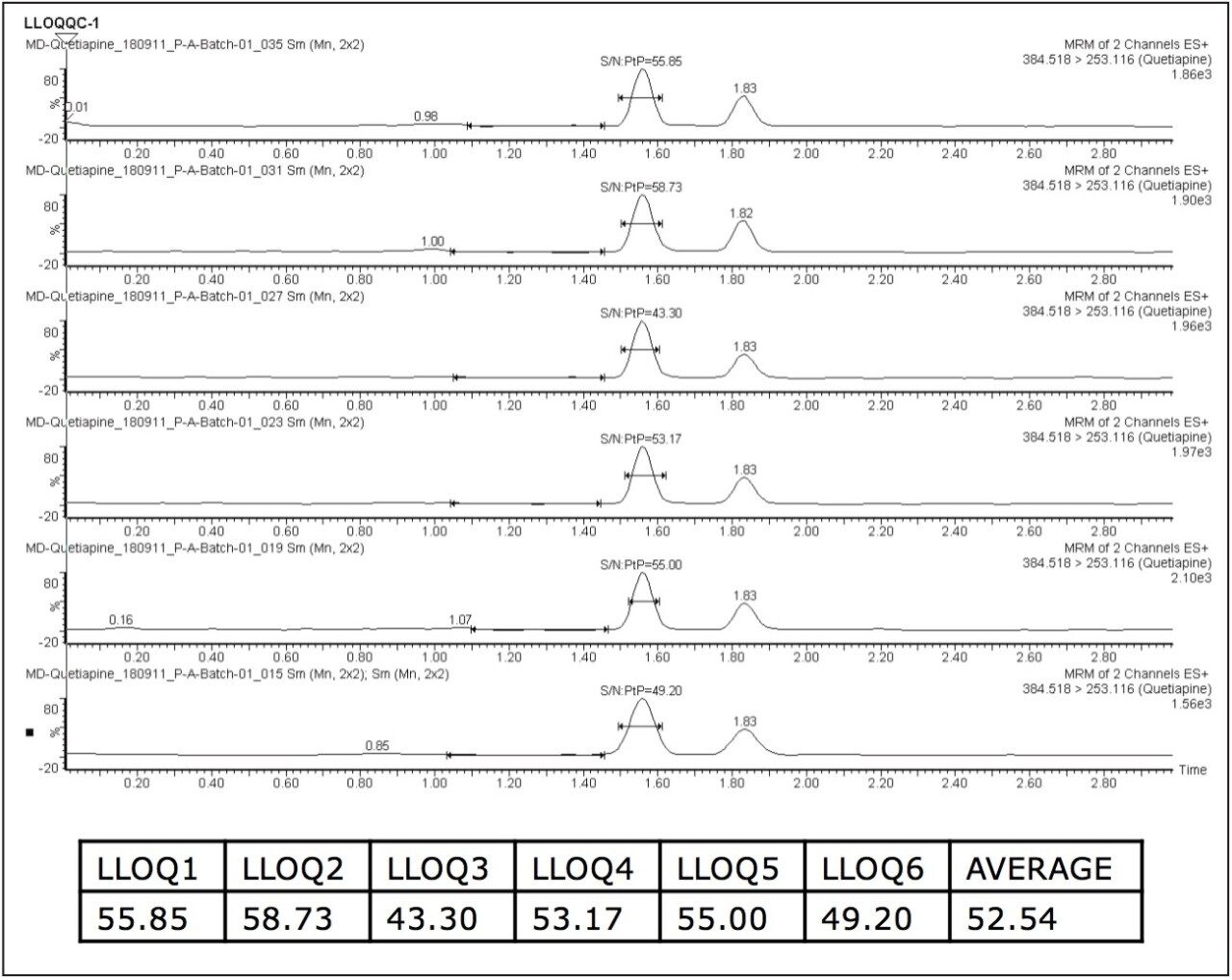
The chromatographic method was obtained using an ACQUITY UPLC System and an ACQUITY UPLC BEH C18 (1.7 μm, 150 mm x 2.1 mm) Column, which provided resolution for quetiapine from other possible co-eluting plasma endogenous peaks present in the samples. Quetiapine eluted at 1.54 min with a peak width of 12 s at the base. Quetiapine gave a signal-to-noise (S/N) ratio of approximately 53 for an average of six replicates of plasma samples at the lower limit of quantification (LLOQ), as shown in Figure 2.
The ACQUITY TQD is a triple quadrupole mass spectrometer that offers an LC-MS/MS system solution with medium- to high-range sensitivity, and it is suitable for fast-paced, routine analysis in multi-user laboratory environments.
For the assay detailed in this application note, quetiapine exhibited a linear calibration curve over a range of 150 pg/mL to 150 ng/mL with an r2 value of 0.998. The back-calculated concentrations of the standards were within ±7 % of the nominal concentration, as shown in Table 1. This assay was performed within a 3 min injection-to-injection time scale in isocratic mode (inclusive of a post-run gradient wash), which allowed for high throughput analysis with high precision.

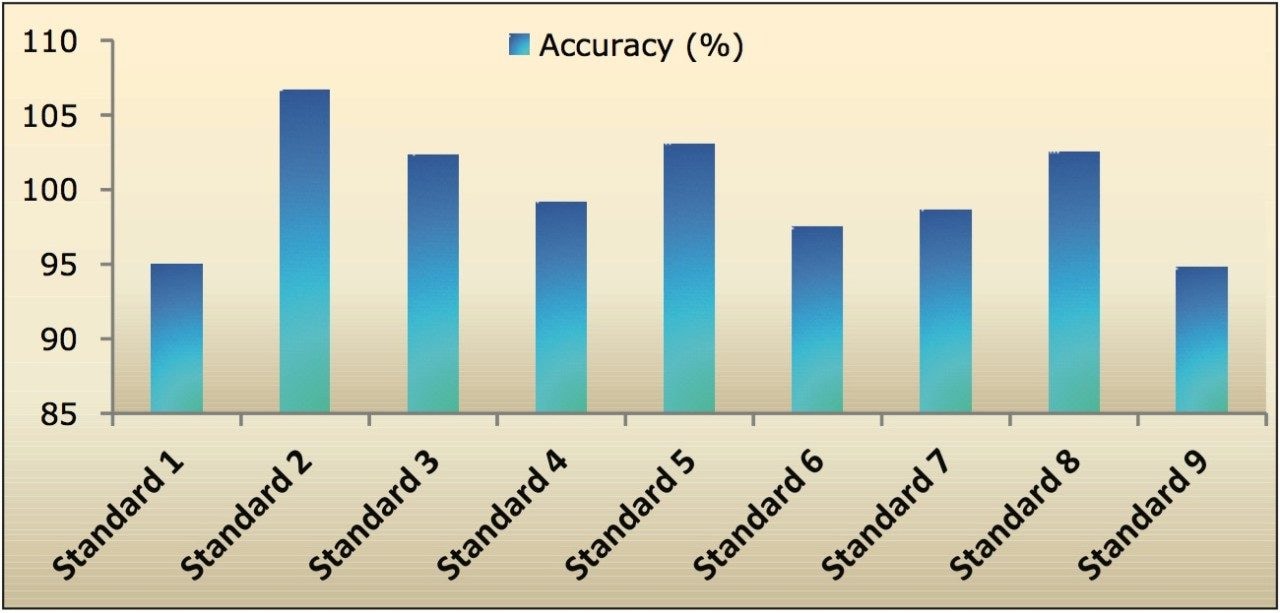
Recovery of the analyte and IS was also performed by comparison of extracted QC samples against neat samples, which was found to be approximately 65% throughout the range. The % accuracy of these measurements was also found to be satisfactory, as shown in Figure 3.
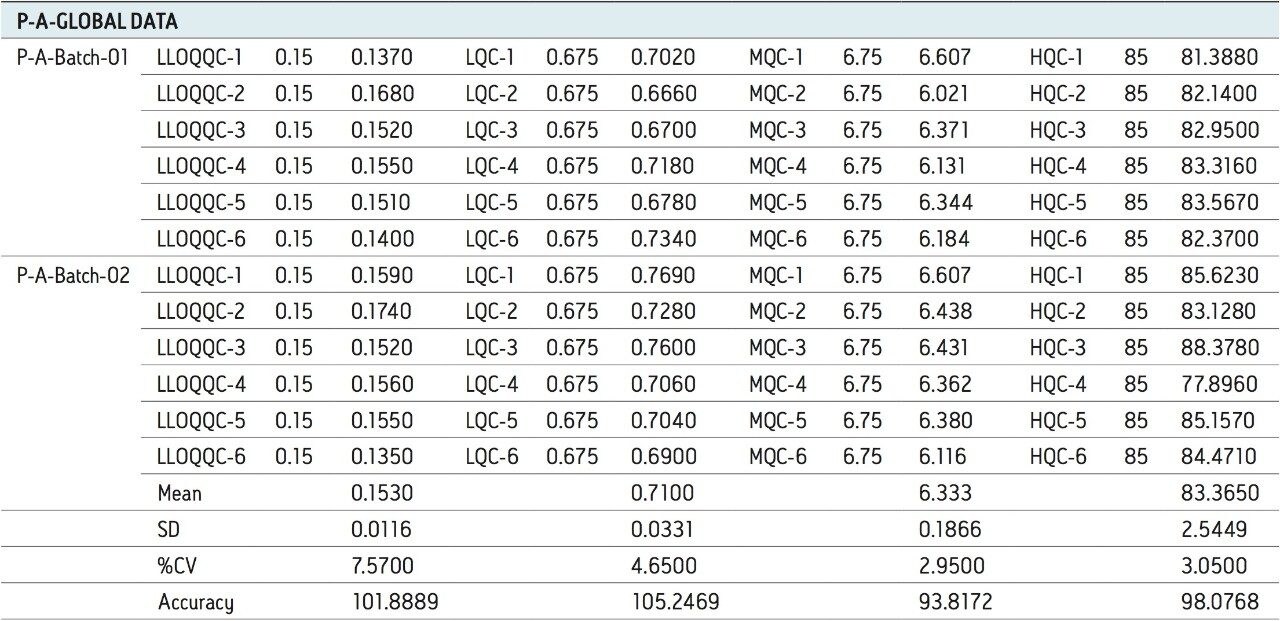
For a comparison of samples within the global batches, two separate batches were prepared with six samples in each batch for LLOQQC, LQC, MQC, and HQC concentration levels. The data showed excellent agreement between the six samples in all the three batches, as shown in Table 2.
720004410, June 2012