This application note demonstrates the use of ASAP, in conjunction with a tandem quadrupole mass spectrometer (ACQUITY TQ Detector) for examining starting materials, monitoring the progress of reductive amination and amide formation reactions, and identifying components in solid-phase extraction fractions.
Without reaction workup or sample preparation, and in less than one minute, structural information can be obtained and informed decisions made without delay. As a result, the organic synthesis workflow efficiency is greatly enhanced, saving time and reducing the cost of analysis.
To maximize return on investment, the chemical industry is challenged to bring new products to the market while improving productivity. In many cases, developing a new product or a new process with only a small percentage increase in the yield can result in significant increases in earnings for the company. To develop new chemicals and improve organic synthesis efficiency, timely chemical analysis data on the progress of reactions and the product purification processes are required.
In organic synthesis labs, reactions are often monitored by TLC, which does not provide chemical structure information and requires mobile phase optimization. Conventional analytical instruments such as NMR, LC, or LC-MS require timeconsuming reaction workup procedures including extraction, filtration, separation, and evaporation. Frequently, it takes hours to obtain the critical chemical structure data. Moreover, when analyzing new reactions and new compounds, method development and expert data interpretation are needed. Therefore, technologies that can quickly provide structural information are highly desirable.
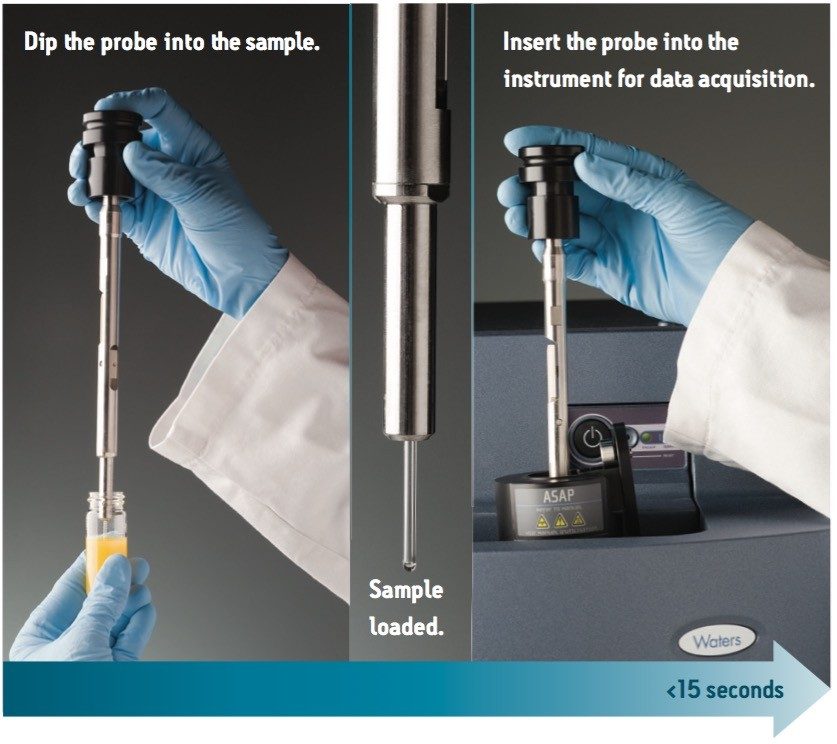
The Waters Atmospheric Solids Analysis Probe (ASAP) is an ambient desorption ionization technique for mass spectrometric (MS) analysis and is capable of ionizing a wide range of volatile and semi-volatile analytes.1-3 The ionization mechanism is similar to atmospheric pressure chemical ionization (APCI). The technique uses heated nitrogen desolvation gas to vaporize the sample and corona discharge for ionization. Solid and liquid samples are directly loaded on to ASAP for MS acquisition, as shown in Figure 1.
This application note demonstrates the use of ASAP, in conjunction with a tandem quadrupole mass spectrometer (ACQUITY TQ Detector) for examining starting materials, monitoring the progress of reductive amination and amide formation reactions, and identifying components in solid-phase extraction fractions. Without reaction workup or sample preparation, in less than one minute, structural information can be obtained and informed decisions made without delay. As a result, the organic synthesis workflow efficiency is greatly enhanced, saving time, and reducing the cost of analysis.
Acetophenone (0.218 g, 1.8 mmol) and aniline (0.169 g, 1.8 mmol) were added to a glass vial containing 3.5 mL of methanol. To the vial, 1.2 mL of 1.5 N HCl in methanol was added. The mixture was stirred at room temperature for two minutes, and then 10 mL of a 11.3 mg/mL solution (1.8 mmol) of NaBH3CN in methanol was added to start the reaction. The progress of the reaction was monitored using ASAP coupled to the ACQUITY TQD.
Acetylimidazole (416 mg, 3.78 mmol) was dissolved in 5 mL of acetonitrile. Phenylphenylamine (170 mg, 1 mmol) was dissolved in 5 mL of acetonitrile in a second vial. The two solutions were then mixed and the reaction progress was examined with ASAP, coupled with the ACQUITY TQD.
To a glass vial containing 10 mL of acetonitrile, 4-dimethylaminopyridine (0.257 mg, 2.1 mmol) and methyloxybenzylamine (285 mg, 2.1 mmol) were added and the mixture was stirred at room temperature for one minute. An excess amount of acetic anhydride (320 mg, 3.1 mmol) was added to start the acylation reaction. Upon completion of the reaction, 8 mL of the mixture was gravity loaded on to a Waters 20 mL PoraPak Rxn CX Cartridge. The product fraction was eluted with 10 mL of methanol, and the base catalyst (4-dimethylaminopyridine) was eluted with 20 mL of 5% ammonium hydroxide in methanol.
The ASAP coupled the ACQUITY TQD was used for the analyses. The sampling procedure is shown in Figure 1. First, the sample was loaded onto the sealed glass melting point capillary tube of the ASAP probe by dipping the tip into the reaction mixture. The ASAP probe was then inserted into the sealed MS source enclosure and the desolvation gas was rapidly heated to 400 ˚C for MS acquisition. MS full scan data from 60 to 500 amu were acquired for 0.5 min using a 0.2 sec scan duration with the following MS tune page parameters:
|
Ionization mode: |
ESCi positive |
|
Corona current (APcI): |
5 μA |
|
Cone voltage: |
2 V |
|
Extractor: |
1 V |
|
Desolvation temp.: |
400 ˚C |
|
Source temp.: |
120 ˚C |
|
Desolvation gas: |
500 L/Hr |
|
Cone gas: |
0 L/Hr |
|
Collision gas flow: |
0.1 mL/min |
To avoid in-source fragmentation and pyrolysis, a low cone voltage (2 V) and low desolvation temperature (400 °C) were applied. To minimize the formation of radical cations (M+), a 2 mL glass vial filled with water was placed inside the source chamber. As a result, highly reproducible mass spectra were obtained. Prior to each analysis, a new glass capillary tube was inserted into the source and baked for approximately 15 sec to minimize any background ions. The acquisition was initiated while the newly baked capillary was still in the source to obtain background reference scans. The probe was removed from the source and sample was applied. The probe was then re-inserted while acquisition continued. Combined mass spectra were obtained by subtracting the baseline of the reference scans from the total ion current profiles of the samples.
Reductive amination reactions are widely used in multiple-step organic syntheses for making agricultural chemicals, food additives, drugs, and building blocks for high performance materials. A reductive amination reaction for making phenyl(phenylethyl)amine (Scheme 1) was conducted using the same conditions reported by Borch et. al.4 The reaction mixture could not be analyzed by LC or LC-MS techniques without workup procedures to remove NaBH3CN since the reducing agent would plug ESI capillary tubes and damage LC columns. In contrast, using ASAP, the reaction mixture was directly loaded onto the melting point capillary of the probe for MS analysis without removing the reducing agent.
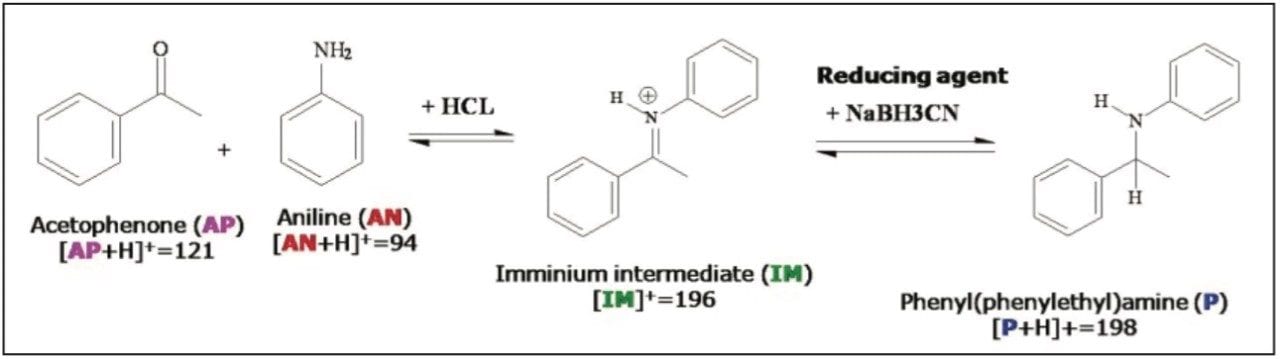
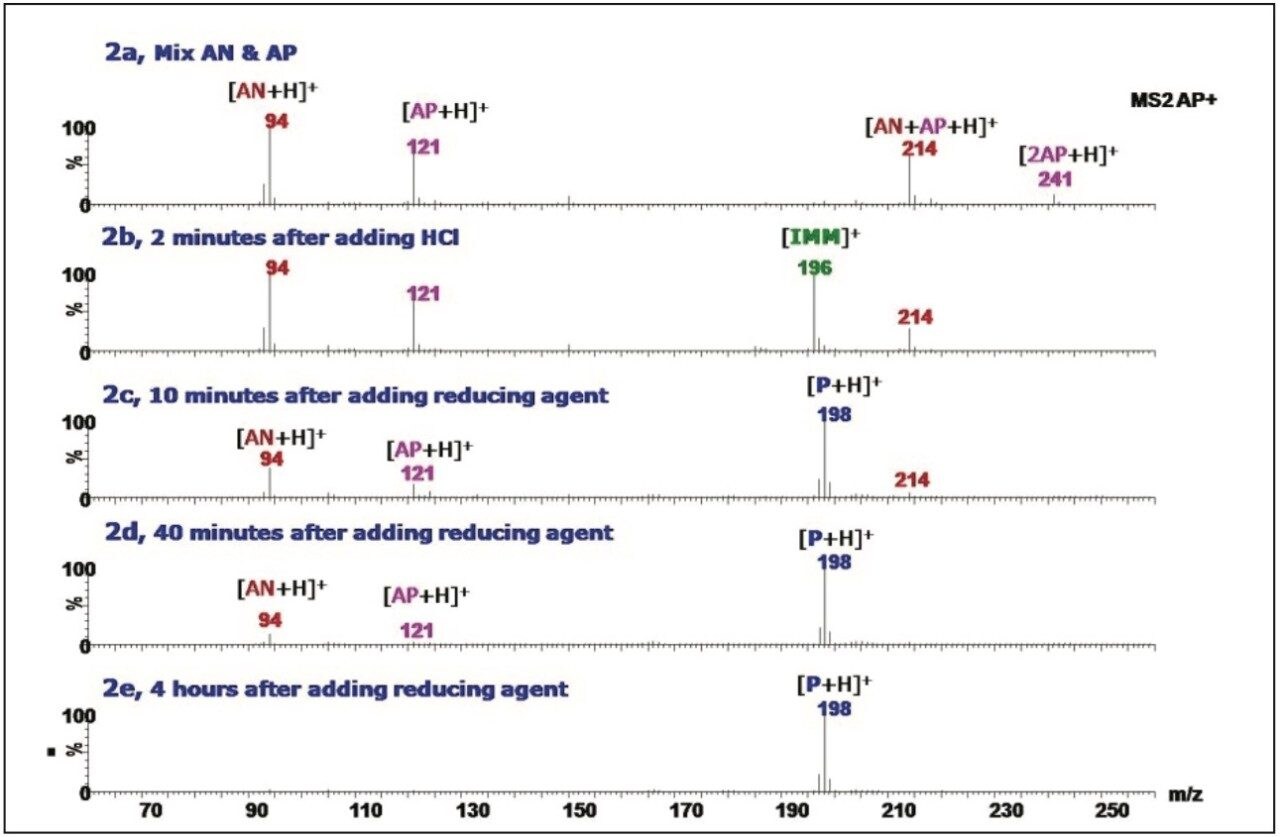
Figure 2 shows the full scan mass spectra of step-by-step reaction procedures. Spectrum 2a was acquired after mixing acetophenone (AP) and aniline (AN) in methanol. Four major ion peaks, protonated aniline ion (m/z 94), protonated acetophenone ion (m/z 121), adduct ion of aniline and acetophenone (m/z 214), and dimer ion of acetophenone (m/z 241) were observed. Their identities were confirmed using MS/MS (data not shown). 2 min after adding HCl solution, the formation of an imminium intermediate4 (IMM) with m/z 196 was apparent in the mass spectrum (Figure 2b). In Figure 2c, acquired 10 min after adding the reducing agent, the spectrum revealed the formation of a protonated phenyl(phenylethyl)amine ion (m/z 198) and the disappearance of the imminium intermediate. The increase in relative ion intensity of the product and decrease in that of the reactant ions after 40 min are shown in Figure 2d. Finally, in the spectrum acquired after 4 hours, no remaining reactants were visible (Figure 2e).
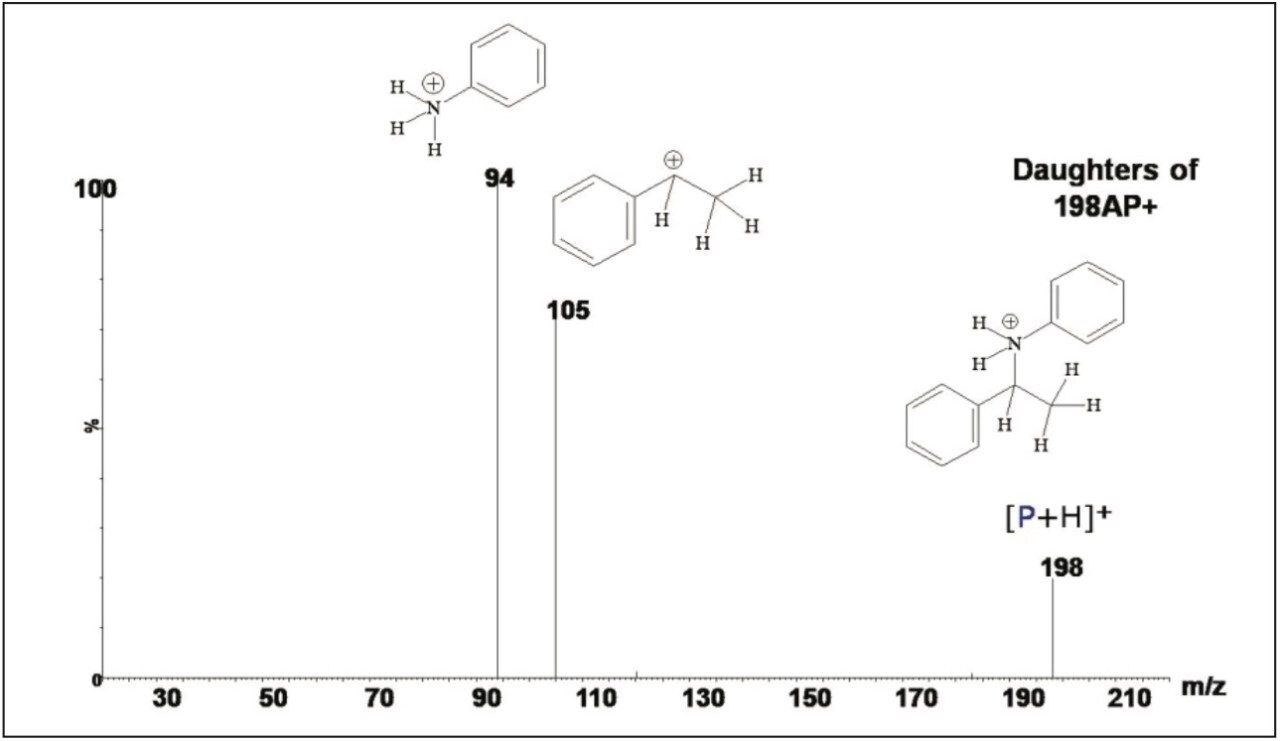
Using ASAP with the ACQUITY TQD, product ion spectra of targeted ions in reaction mixtures can be easily acquired to further confirm the identity of the ions detected in full scan mode. Figure 3 shows the product ion spectrum of m/z 198 acquired at a collision energy of 7 V. Two product ion peaks with m/z of 94 and 105 were observed, confirming that m/z 198 is indeed the protonated phenyl(phenylethyl)amine ion. The experiments demonstrate that without reaction workup or sample cleanup, ASAP rapidly provides structural information on the progress of reductive amination reactions to maximize organic synthesis efficiency and yields.
Amide bond formation through acylation reactions is another synthetic pathway of critical importance in many industries. The rate of acylation reactions depends highly on the amines, acylation reagents and catalysts used in the reaction. The workup procedures usually include liquid-liquid extraction and evaporation. The products are often purified using flash chromatography or preparative LC. Scheme 2 is an example of acetamide formation using acetylimidazole (AI) as the acylation reagent to yield 4-(phenylphenyl)- acetamide (PPA).
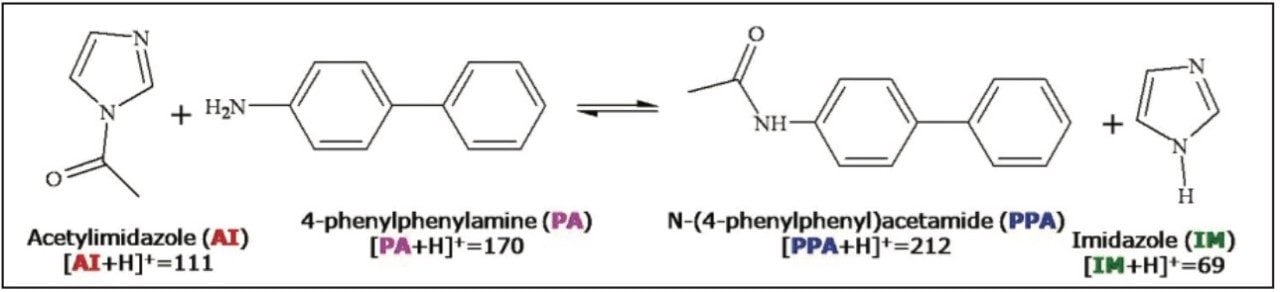
Figure 4 shows the acylation reaction monitored using ASAP with ACQUITY TQD. Spectrum 4a was acquired after dissolving acetylimidazole (AI) in acetonitrile. The molecular ion and dimer ion of acetylimidazole are apparent at m/z 111 and m/z 221, respectively. The observation of imidazole ions (m/z 137, m/z 69) indicates that some acetylimidazole had decomposed. Since acetylimidazole can be hydrolyzed to form imidazole upon exposure to moisture in the atmosphere, its decomposition was not unexpected. Spectrum 4b was acquired after dissolving phenylphenylamine (PA) in acetonitrile. Only protonated molecular ions of 4-phenylphenylamine (m/z 170) and the dimer ion of 4-phenylphenylamine (m/z 339) were observed.
The spectrum in Figure 4c was acquired 10 min after mixing the solutions of AI and PA. Four new ions (m/z 212, 280, 381, and 423) were observed. MS/MS experiments (data not shown) were consistent with the structures labelled in Figure 4c. Three hours after mixing the solutions of acetylimidazole and 4-phenylphenylamine (Figure 4d) the AI/IM adduct (m/z 179) was not detected and after 3 days (Figure 4e) the limiting reagent (PA) was no longer evident.
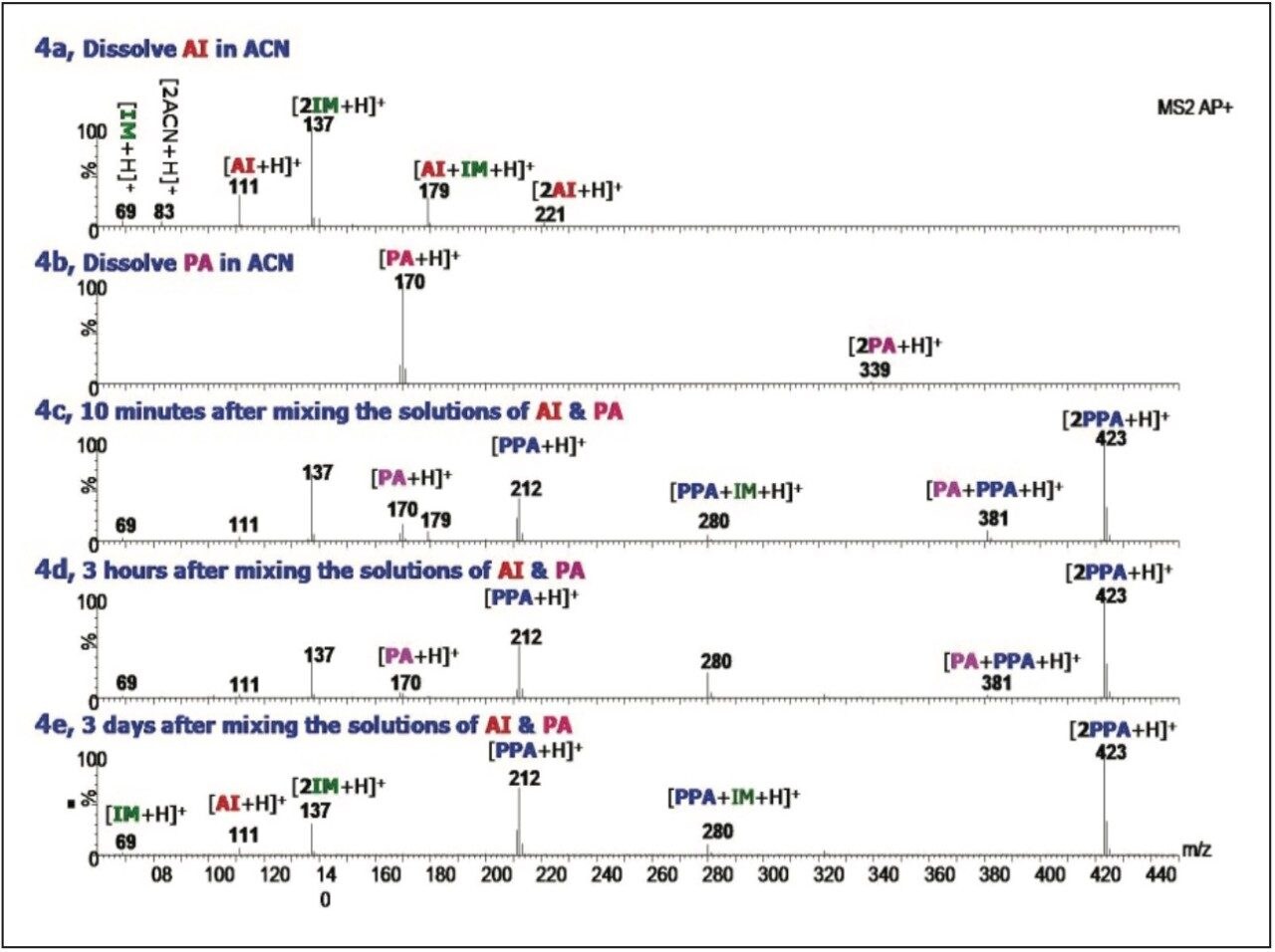
Scheme 3 shows another example of acetamide formation reaction using acetic anhydride as the acylation reagent and 4-dimethylaminopyridine (DMAP) as the base catalyst to produce N-[(4-methoxyphenyl)methyl] acetamide (MAA).
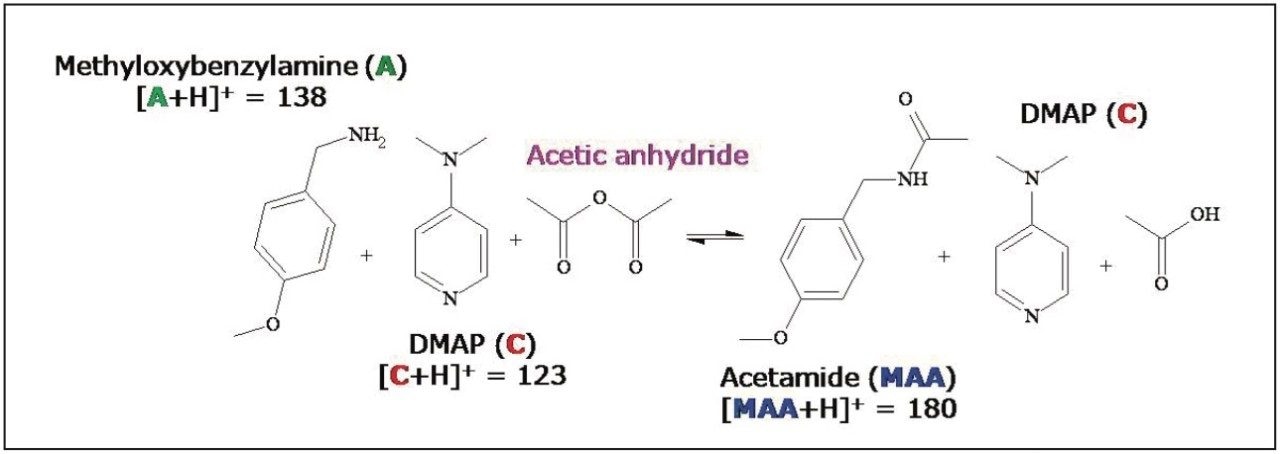
Figure 5 shows the full scan mass spectra of the step-by-step reaction and separation procedures of the acylation reaction. Spectrum 5a was acquired after mixing methyloxybenzylamine and the base catalyst DMAP in CH3CN. Four major ion peaks from the starting material and the catalyst were identified, as shown in Figure 5a. 5 min after adding an excess amount of acetic anhydride, ions from the formation of the protonated N-[(4-methoxyphenyl) methyl] -acetamide ion (m/z 180) and its dimer (m/z 359) were apparent (Figure 5b) and the reactant peaks had disappeared. The results suggest that the reaction was completed in five min.
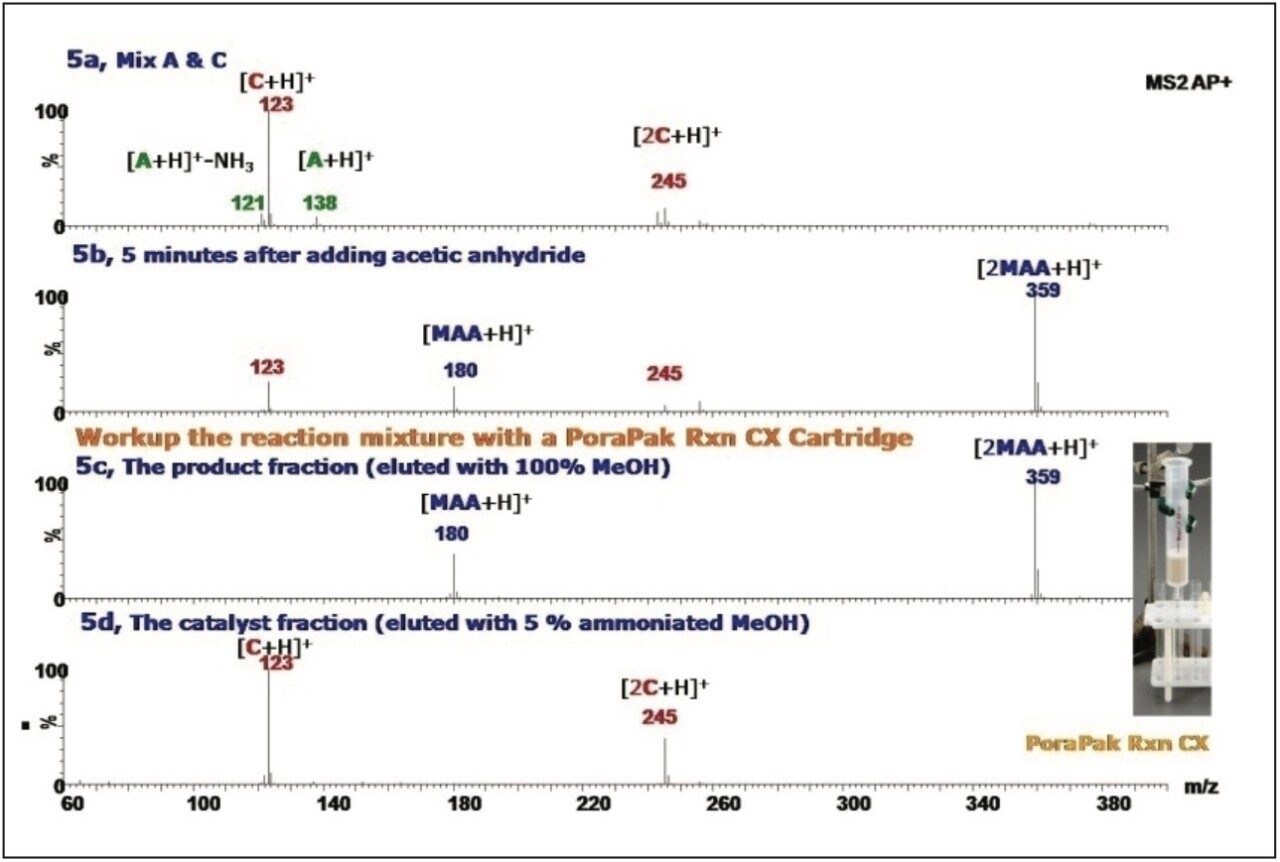
The reaction mixture was purified with a PoraPak Rxn CX Cartridge to separate the product N-[(4-methoxyphenyl) methyl]-acetamide (MAA) from the catalyst DMAP. The PoraPak Rxn CX Cartridge contains strong cationexchange sorbents designed to separate neutral and base compounds without conventional workup procedures.
The reaction mixture was first loaded onto a 20 mL PoraPak Rxn CX Cartridge. Then, 100% methanol was used to elute the product, MAA. Figure 5c shows the spectrum of the methanol eluted fraction and only the protonated N-[(4-methoxyphenyl) methyl]-acetamide ion (m/z 180) and its dimer ion (m/z 359) were observed. The trapped base compounds were eluted from the cartridge using a methanol solution containing 5% ammonium hydroxide. Only the protonated DMAP ion (m/z 123) and the dimer ion (m/z 245) were observed, as shown in Spectrum 5d. The data illustrate that the reaction product MAA was successfully separated from DMAP with the PoraPak Rxn CX Cartridge.
As demonstrated in this work, ASAP coupled to an ACQUITY TQ Detector can provide full scan MS, as well as MS/MS experiments. The system can rapidly examine the progress of reactions, confirm the formation of reaction products, identify product fractions from solid phase extraction, and obtain product ion spectra for structural confirmation. An alternative option for rapid testing of simple mixtures is the single-stage quadrupole mass spectrometer (ASAP/ACQUITY SQD), which offers ease-of-use and a lower cost. For comprehensive sample characterization, ASAP coupled to a quadrupole time-of-flight mass spectrometer, such as the Xevo G2 Q-Tof provides high sensitivity, full spectrum analyses, accurate mass data, and the ability to perform MS/MS experiments. Depending on the complexity of the reaction mixtures and the testing requirements, each of these systems offers different benefits.
This application note demonstrates the benefits of ASAP for organic synthesis labs. Without workup or separation, ASAP together with MS can be used to directly analyze reaction mixtures. Mass spectra can be obtained in less than one minute to:
In this way, informed decisions can be made without delay, greatly enhancing the workflow efficiency of organic synthesis. The ASAP solution reduces solvent usage and lessens the impact on the environment in line with the principles of green chemistry. As a result, daily lab operating costs can be reduced.
720003628, July 2010