Optimizing UHPLC-to-HPLC Method Translation for the USP Assay Method for Salicylamide
Norris Wong, Paula Hong
Waters Corporation, United States
Published on April 17, 2026
Abstract
In this study, a United States Pharmacopeia (USP) ultra-high-performance liquid chromatography (UHPLC) gradient method was translated to the Alliance™ iS HPLC System, demonstrating how the system’s 12,000 psi pressure capability and Empower™ Software tools support both UHPLC and conventional HPLC workflows. Many modern high-performance liquid chromatography (HPLC) systems can perform UHPLC separations, but dwell volume differences may complicate the migration, preventing the chromatography from being “just right”. Using Gradient Start and systematic method scaling in accordance with USP <621> chromatography guidance, method parameters were efficiently adjusted to account for system dwell volume differences, enabling successful migration of the separation. These tools facilitated better alignment of retention times, peak resolution, and system suitability results. These results illustrate how the combination of extended pressure range and integrated software tools enable flexible method translation, support efficient deployment of UHPLC methods on HPLC instrumentation.
Benefits
- Alliance iS HPLC System with 12,000 psi pressure range is compatible with a range of UHPLC separations
- Gradient Start provides a tool to investigate and mitigate the effect of dwell volume
- Waters Column Calculator can provide assistance in method scaling following USP General Chapter <621> chromatography guidance
Introduction
With the desire to modernize regulated methods, UHPLC methods are becoming increasingly common as an option. Furthermore, to accommodate laboratories that have a wide range of instruments, these methods may be developed at lower pressure ranges, making them compatible with some HPLC systems. Differences in dwell volume between systems remain a significant challenge, especially when translating UHPLC gradient methods to HPLC systems. In regulated laboratories, methods are frequently developed on UHPLC systems and later migrated to HPLC systems for routine analysis.¹ UHPLC separations are characterized by sub-2-µm particle size columns, higher system operating pressures, shorter run times, lower flow rates, and lower injection volumes compared to traditional HPLC separations.¹,² When these UHPLC methods are directly migrated to HPLC systems, the dwell volume differences may impact resolution and retention times of target analytes and cause system suitability failure, preventing the chromatography from being “just right”.¹,² There are several strategies to ensure that a UHPLC method can be successfully translated to an HPLC system. Using the Gradient Start feature, gradient timing can be adjusted to compensate for dwell volume differences to optimize the chromatography. Alternatively, the method can be scaled following USP General Chapter <621> guidance, which allows method adjustments for gradient methods.³ In this study, the USP UHPLC assay method for salicylamide, which uses a sub-2-µm column,⁴ is directly migrated to an HPLC system. Both strategies and their success in resolving dwell volume issues are examined.
Experimental
The USP UHPLC assay method for salicylamide was run on the Alliance iS HPLC System. Chromatography on the HPLC system was optimized using the gradient start adjustment feature and method scaling.
USP Assay Method for Salicylamide
Sample Description: A standard solution of USP Salicylamide RS (USP, p/n: 1608000) was prepared at 0.2 mg/mL in 50/50 acetonitrile/water. A system suitability solution of 1.0 mg/mL USP Salicylamide RS and 1 µg/mL USP Salicylic Acid Related Compound B RS (USP, p/n: 1609024) was prepared in 50/50 acetonitrile/water.
LC Conditions
|
LC system: |
Alliance iS HPLC System |
|
Detection: |
UV @ 300 nm |
|
Data rate: |
10 points/sec |
|
Vials: |
TruView™ pH Control LCMS Certified Clear Glass, 23 x 32 mm, Screw Neck Vial, with Cap and pre-slit PTFE/Silicone Septum, 2 mL Volume, 100/pk (p/n: 186005666CV) |
|
Column(s): |
ACQUITY™ Premier HSS T3 Column, 2.1 mm x 100 mm; 1.8 µm (p/n: 186009468) |
|
Column temperature: |
35 °C |
|
Sample temperature: |
15 °C |
|
Injection volume: |
1 µL |
|
Flow rate: |
0.3 mL/min |
|
Mobile phase A: |
0.1% Formic acid in water |
|
Mobile phase B: |
Acetonitrile |
Gradient Table
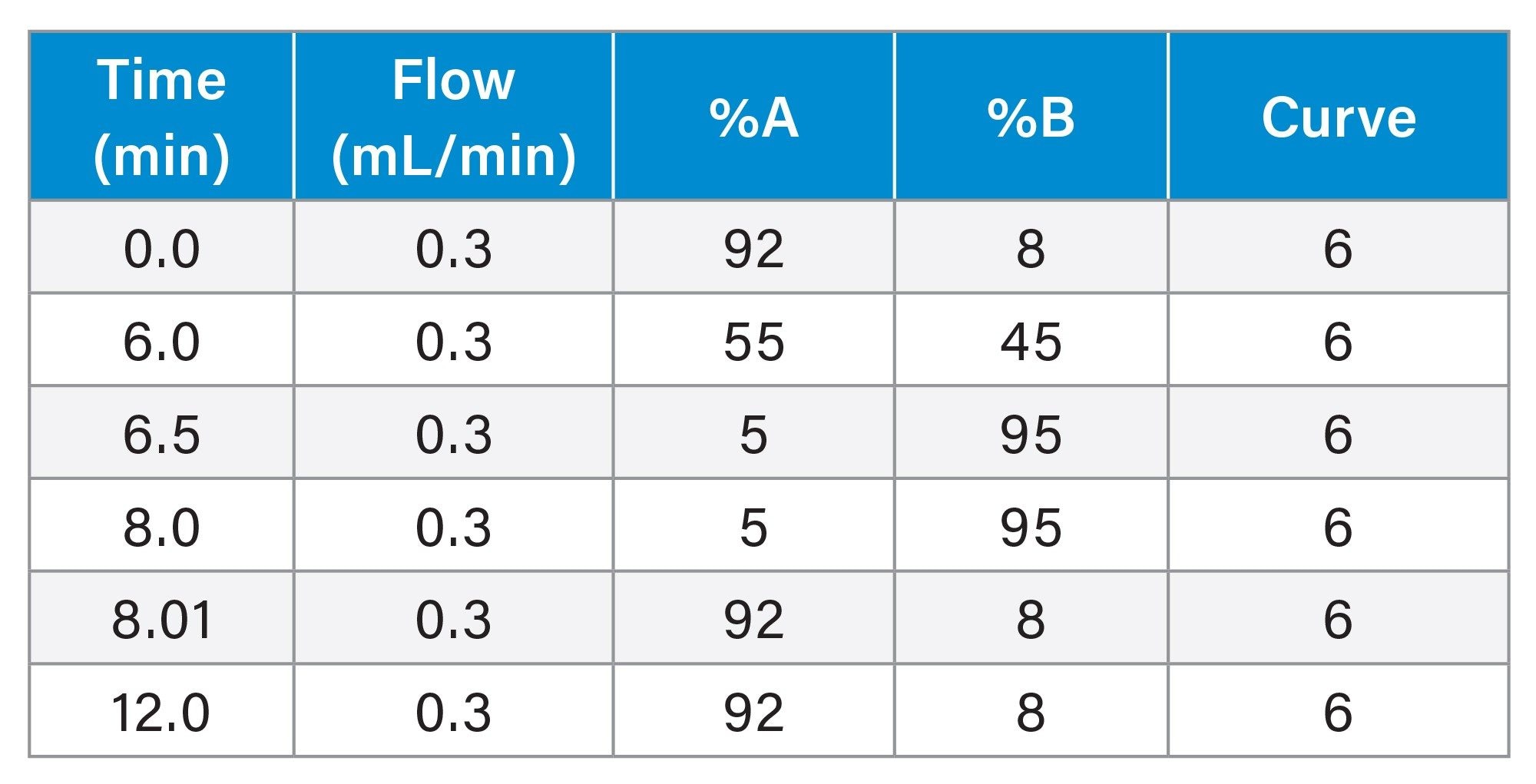
Scaled LC Conditions
|
LC system: |
Alliance iS HPLC System |
|
Detection: |
UV @ 300 nm |
|
Data rate: |
10 points/sec |
|
Vials: |
TruView™ pH Control LCMS Certified Clear Glass, 23 x 32 mm, Screw Neck Vial, with Cap and pre-slit PTFE/Silicone Septum, 2 mL Volume, 100/pk (p/n: 186005666CV) |
|
Column(s): |
XBridge™ BEH C18 Column 4.6 mm x 150 mm; 3.5 µm (p/n: 186003034) |
|
Column temperature: |
35 °C |
|
Sample temperature: |
15 °C |
|
Injection volume: |
7 µL |
|
Flow rate: |
0.74 mL/min |
|
Mobile phase A: |
0.1% Formic acid in water |
|
Mobile phase B: |
Acetonitrile |
Scaled Gradient Table
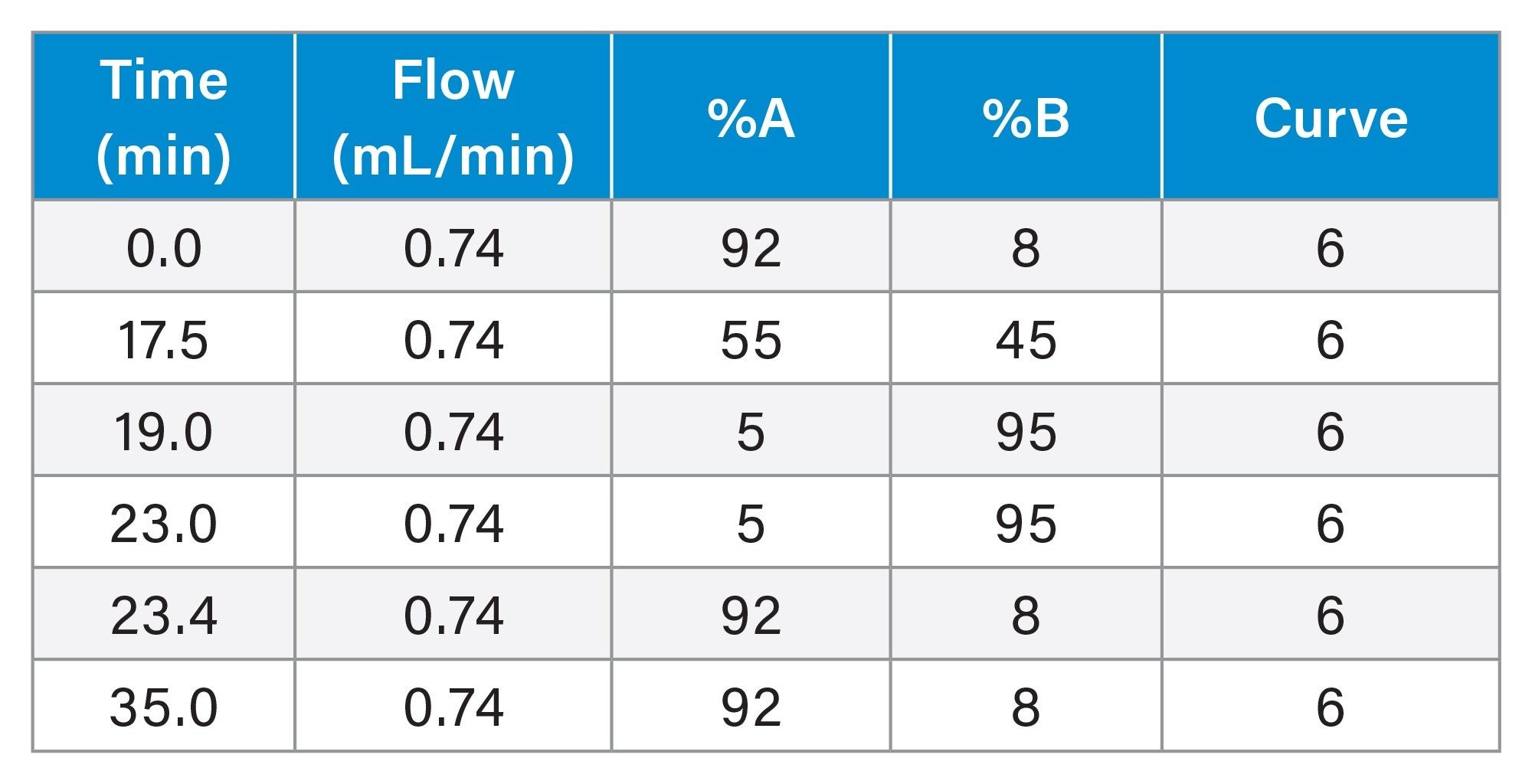
Data Management
|
Chromatography software: |
Empower 3 FR5 and 3.7 |
Results and Discussion
Assessment of Ability to Perform UHPLC Method on an HPLC System
Direct migration of a UHPLC gradient method to an HPLC system can be complicated by differences in instrument characteristics. For example, greater extra-column dispersion can lead to excessive band broadening, while larger dwell volumes can impact the gradient delivery and re-equilibration on column. Focusing on dwell volume (or gradient delay), any inline instrument components from the point of mixing contribute to the total dwell volume.⁵,⁶
To assess the performance of the USP salicylamide assay method, a UHPLC gradient method using a 2.1 × 100 mm column was executed on an HPLC, as described in the compendial method. The resulting chromatography did not produce expected results. A critical suitability criterion for the USP salicylamide assay is USP resolution (Rs) between salicylic acid related compound B and salicylamide (Rs ≥ 1.5). For this criterion, no value could be determined (as shown in Figure 1A), since the chromatography was poor. Peak identification was difficult to establish due to a significant retention time shift.
To assist in troubleshooting, the system diagnostic channels were investigated. Of the various available channels, system pressure trace is a common means of visualizing gradient delivery and is a good tool for troubleshooting gradient methods. In this investigation, during the injection, the HPLC system pressure dips to its lowest point or nadir at 12 minutes, at the end of the injection (Figure 1B). Thus, the pressure never returns to initial levels before the end of injection. This suggests the initial mobile phase conditions have not been delivered to the column, impacting both column equilibration before the next injection and the chromatographic data.

For gradient methods, the gradient delay acts as an unprogrammed isocratic hold. On systems with larger dwell volume, the duration of this unprogrammed isocratic hold can result in a shift in retention times and impact the re-equilibration or the ability of the column to return to initial method conditions prior to the next injection (Figure 2).⁶,⁷ This is particularly noticeable if the flow rate is very low (<0.5 mL/min) or the column is not sufficiently re-equilibrated (5-10 column volumes). For example, in the USP salicylamide assay method with a flow rate of 0.3 mL/min, the gradient delivery might shift by a few minutes (Figure 2), impacting re-equilibration.

Strategy 1: Adjustment of Dwell Volume with Gradient Start
The Alliance iS HPLC System has integrated tools within the instrument method to make adjustments for dwell volume following USP <621> guidelines.³ Gradient Start allows a user to increase or decrease the gradient delay, relative to the start of the injection. As shown in Figure 3, gradient start can be adjusted by volume (µL) or time (min). Thus, if expected retention time for a peak is known, as in method migration, the gradient start can be adjusted by the observed retention time difference.
For the USP method, the expected retention time was unknown. To determine a value for the gradient adjustment, the system pressure trace was used (Figure 4). Based on the required equilibration of 1.2 mL, the gradient start could be adjusted before the injection, ensuring the column would be sufficiently equilibrated. The adjustment was then shortened to see the minimum delay required. A final value of 1157 µL or 3.86 minutes was ascertained for the gradient adjustment, close to the 1.2 mL required for the column to be re-equilibrated. This ensured a return to initial pressure for approximately 1 minute or 0.3 mL column volumes before the end of injection.


With gradient adjustment, the chromatography improved with salicylic acid RCB and salicylamide eluting as separate, fully resolved peaks (as shown in Figure 5). The Rs of salicylic acid RCB and salicylamide was assessed. The gradient adjusted trace demonstrated a USP resolution of 3.6, which met method system suitability.

Strategy 2: Scaling UHPLC Method to HPLC Column
Method scaling is an alternative solution for migrating UHPLC methods to HPLC. This typically involves scaling up to columns with larger particle sizes while ensuring the system suitability continues to be met. Laboratories often may perform method scaling when modifying high separation efficiency methods developed on UHPLC systems for more routine analysis on HPLC systems.⁸ USP General Chapter <621> Chromatography PF 49 (6) lists allowable adjustments for isocratic and gradient methods, as well as related manual calculations required for scaling.³ For the salicylamide assay method, scaling to an HPLC column required additional adjustments to the gradient table, run time, flow rate, and injection volume.
Calculations for these parameters can be performed using the Waters Columns Calculator. The method was scaled from a 2.1 × 100 mm, 1.8 µm column to a 4.6 × 150 mm, 3.5 µm column, preserving the L/dp ratio within -25% of the original column (Figure 6). The calculator is a tool accessible from the Empower method editor or can be installed as a standalone app and performs all necessary calculations.⁸ With scaling a method up, there are impacts to throughput and solvent consumption. As shown in Figure 6, the longer runtime requires more time to complete an injection and, coupled with the greater flow rate, consumes a greater quantity of mobile phase. Sample consumption also increased due to the larger injection volume. The scaled salicylamide method was compatible with the Alliance iS HPLC System and met the system suitability criteria of the method (Figure 7).


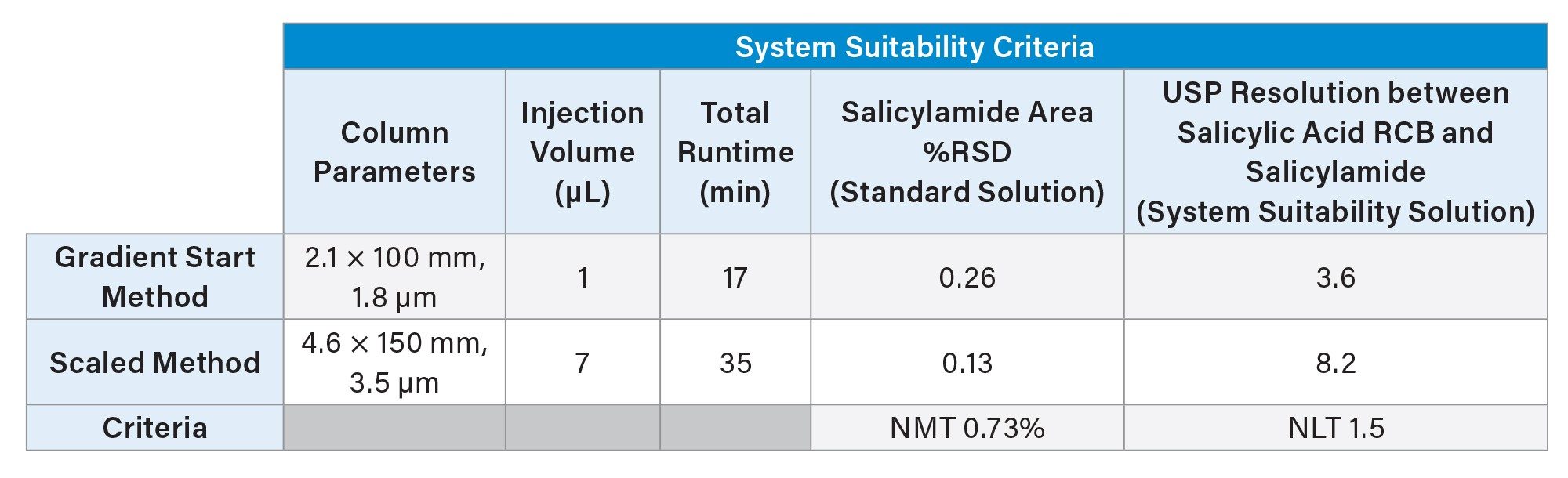
Comparison of Results
System suitability determines whether a method performed on an LC system is “just right”. In this example, both gradient start and method scaling strategies successfully met system suitability criteria (Table 1). However, there were significant differences in column volumes, with adjustment of gradient start employing a smaller volume UPLC column. The first criterion is the area %RSD of salicylamide in the standard solution, which must not exceed 0.73%. Both strategies met area %RSD, with the gradient start area %RSD of 0.26% and method scaling area %RSD of 0.13%. The lower area %RSD is likely due to the larger injection (7 µL) volume of the scaled method.
The second criterion is the USP resolution between salicylic acid RCB and salicylamide in the system suitability solution, which needed to be greater than 1.5. While both met the system suitability criterion, the USP resolution between salicylic acid RCB and salicylamide in the system suitability solution increased going from (Rs=3.6) with the gradient start adjusted method, to (Rs=8.2) in the scaled method (Table 1). The gradient start method, which uses a smaller column UHPLC column on an HPLC system, gives lower resolution due to the greater impact of extra-column band broadening on peak widths. Larger volume HPLC columns, with larger peak volumes, are less impacted by this attribute.
Conclusion
UHPLC methods are typically developed and optimized for UHPLC systems. However, there may be a need to migrate the method to HPLC systems with different instrument characteristics. In cases like these, adjustments of dwell volume and extra-column volume may be needed for the UHPLC method. For dwell volume adjustments, a variety of strategies can be used. Tools such as Gradient Start and systematic method scaling can help make method migration easier.
Using Gradient Start allows the Alliance iS HPLC System to compensate for dwell volume adjustments. However, this strategy does not mitigate extra-column band broadening effects that may be observed when running a UHPLC method on an HPLC System. Another way to accommodate UHPLC methods is to adjust the method by scaling to a column more compatible with the system, or an HPLC column. Method scaling may be more intensive due to the manual calculations outlined by USP <621>, but Waters Columns Calculator can automate these calculations for the user and output new gradient tables, run times, and flow rates.
Both the gradient adjusted and scaled methods successfully met all system suitability criteria and met the challenging resolution criteria between salicylic acid RCB and salicylamide. Executing a UHPLC method on a HPLC system is not an impossible task and the chromatography can be “just right”. With these tools, the dwell volume of any HPLC system can accommodate the method and meet system suitability.
References
- Dong M. More Myths in Ultrahigh-Pressure Liquid Chromatography. LCGC Europe. 2013 Nov;26(11).
- Dong M. UHPLC, Part I: Perspectives and Instrumental Features. LCGC North America. 2017 Jun;35(6):374-81.
- General Chapter: USP. Chromatography <621>. In: USP-NF. Rockville, MD: USP, 01 Dec 2024. DOI: https://doi.org/10.31003/USPNF_M99380_09_01.
- Monograph: USP. Salicylamide. In: USP-NF. Rockville, MD: USP; 13 Aug 2025. DOI: http://doi.org/10.31003/USPNF_M74280_04_01.
- Hong P and McConville P. Dwell Volume and Extra-Column Volume: What Are They and How Do They Impact Method Transfer? Waters White Paper. 720005723. 2018 April.
- Dolan J. Gradient Elution, Part IV: Dwell-Volume Problems. LCGC North America. 2013 Jun; 31(6).
- Dolan, J. Dwell Volume Revisited. LCGC North America. 2006 May;24(5).
- Berthelette K, Turner J, Kalwood J, Haynes K. Faster, Simpler Method Scaling Across Particle Sizes Using the Waters Columns Calculator Compared to Manual Calculation Workflow. Waters Application Note. 720007887. 2023 July.
Featured Products
720009288, April 2026