Quantitation of Cortisone-21-Acetate Impurities using HPLC/UV with a Waters XBridge Biphenyl RP Column with MaxPeak Premier Technology
Melissa Aiello, Kenneth Berthelette, Christopher Collins, Thomas H. Walter
Waters Corporation, United States
Published on April 13, 2026
Abstract
The creation of precise HPLC methods for assessing purity is crucial in the development of pharmaceuticals. These methods help characterize many process-related and storage-related impurities and allow for strict quality control of such compounds in the final drug product. For a method to be precise, the High-Performance Liquid Chromatography (HPLC) Column must be chosen carefully. While octadecyl silica (C18) columns are the gold standard for reversed-phase chromatographic separations, there are a number of alternatives when C18 columns don’t resolve all the analytes of interest. An example is biphenyl columns, which offer alternative selectivity for compounds containing π electrons through π-π interactions with the stationary phase. However, biphenyl columns often show UV-detected column bleed in gradient separations when using acidic or basic mobile phases. Often observed as a baseline rise or hump, column bleed makes it difficult to develop methods that deliver consistent results. In this work, a Waters XBridge Biphenyl RP Column with MaxPeak Premier Technology was compared to two biphenyl columns from other vendors to analyze the forced degradation products of cortisone-21-acetate. Unlike the comparison columns, the XBridge Biphenyl Column showed no bleed, enabling the precise quantification of low-level impurities using UV detection.
Benefits
- Waters XBridge Biphenyl Columns produce little to no UV-detected column bleed compared to biphenyl columns from other vendors, enabling reliable quantitation of low-level impurities with UV detection
Introduction
Corticosteroids are a group of steroid hormones that are secreted by the adrenal cortex. They play a fundamental role in the body’s response to stress, such as how sugar and fat are metabolized by cells, and in controlling inflammation by suppressing certain cellular protein signals.1 Cortisone is a corticosteroid with mostly glucocorticoid activity, such as suppressing inflammation in the body.2 The man-made prodrug is the acetate ester at C21, cortisone-21-acetate (C21A; Figure 1). It is widely used in medicine, from topical applications to treat eczema (atopic dermatitis), to enteric applications such as replacement hormones in adrenocortical insufficiency (Addison’s disease).2 As a widely used drug available both over and behind the counter, strict quality control of drug products containing cortisone-21-acetate is crucial.

C21A drug substances are often analyzed in the pharmaceutical laboratory using HPLC assays with both UV and mass detection. While it is common to use a C18 column in these analyses, recent studies demonstrate that a biphenyl column offers improved selectivity for corticosteroids.3 The biphenyl group contains two phenyl rings that can undergo π-π interactions with analytes, providing an additional mechanism for chromatographic retention.4
However, there are analytical challenges with currently available biphenyl columns. One example is the problem of UV-detected column bleed, where under high and low pH conditions, a significant baseline rise or hump is observed in gradient separations.5 Waters recently introduced a trifunctionally-bonded biphenyl stationary phase based on BEH (ethylene-bridged hybrid) particles that not only shows superior pH stability, but also excellent batch-to-batch reproducibility and little to no UV-detected column bleed.5
Quantitation of low-level impurities of drug substances is crucial to drug development, and a column that provides reliable peak resolution and flat baselines is fundamental. In this work, a Waters XBridge Biphenyl RP Column with MaxPeak Premier Technology was evaluated along with biphenyl columns from two other vendors for the analysis of the forced degradation products of cortisone-21-acetate. Specifically, the level of UV-detected column bleed and its impact on the precision of quantitation of low-level impurities was evaluated.
Experimental
Sample Preparation
A 1 mg/mL solution of cortisone-21-acetate in 50:50 (v/v) water/acetonitrile was prepared, and 0.9 mL was aliquoted into a separate vial. A 0.1 mL volume of 1 N HCl was added and the vial was heated to 70 °C for 3 hours. The contents of the vial were quenched with 0.1 mL of 1 N NaOH and then aliquoted into an LC vial for analysis.
Analytical Method Conditions
|
LC system: |
Arc™ HPLC with 2998 PDA Detector |
|
Detection: |
UV @ 254 nm |
|
Column(s): |
Waters XBridge Biphenyl RP Column with MaxPeak Premier Technology 2.1 x 50 mm, 2.5 µm Vendor R Biphenyl Column 2.1 x 50 mm, 2.7 µm Vendor A Biphenyl Column 2.1 x 50 mm, 2.7 µm |
|
Column temperature: |
30 °C |
|
Injection volume: |
0.1 µL |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Washes and diluent: |
50:50 (v/v) water/acetonitrile |
|
Flow rate: |
1.0 mL/min |
|
Gradient: |
Initial condition of 5% B was followed by a linear gradient to 95% B over 6.86 minutes. The composition was held at 95% B for 1.14 minutes then returned to the initial condition and held for 2.28 minutes to re-equilibrate. The total run time was 10.30 minutes. |
Data Management
|
Chromatography software: |
Empower™ 3 Chromatography Data System (CDS) |
Results and Discussion
Baseline drift is a common occurrence during gradient separations when using UV detection. This is generally due to the change in UV absorptivity of the mobile phase as the gradient progresses.6 However, most biphenyl columns exhibit an additional baseline distortion due to the elution of bonded phase hydrolysis products with the biphenyl group attached (column bleed),5 which can result in a hump in the chromatogram. Figure 2 compares the chromatograms of blank diluent injections on different biphenyl columns. Biphenyl columns from Vendor A and Vendor R demonstrate significant column bleed up to about 4.3 minutes, while the Waters XBridge Biphenyl Column shows no detectable bleed. This attests to the ability of the trifunctionally-bonded stationary phase to resist acid-catalyzed hydrolytic degradation.5
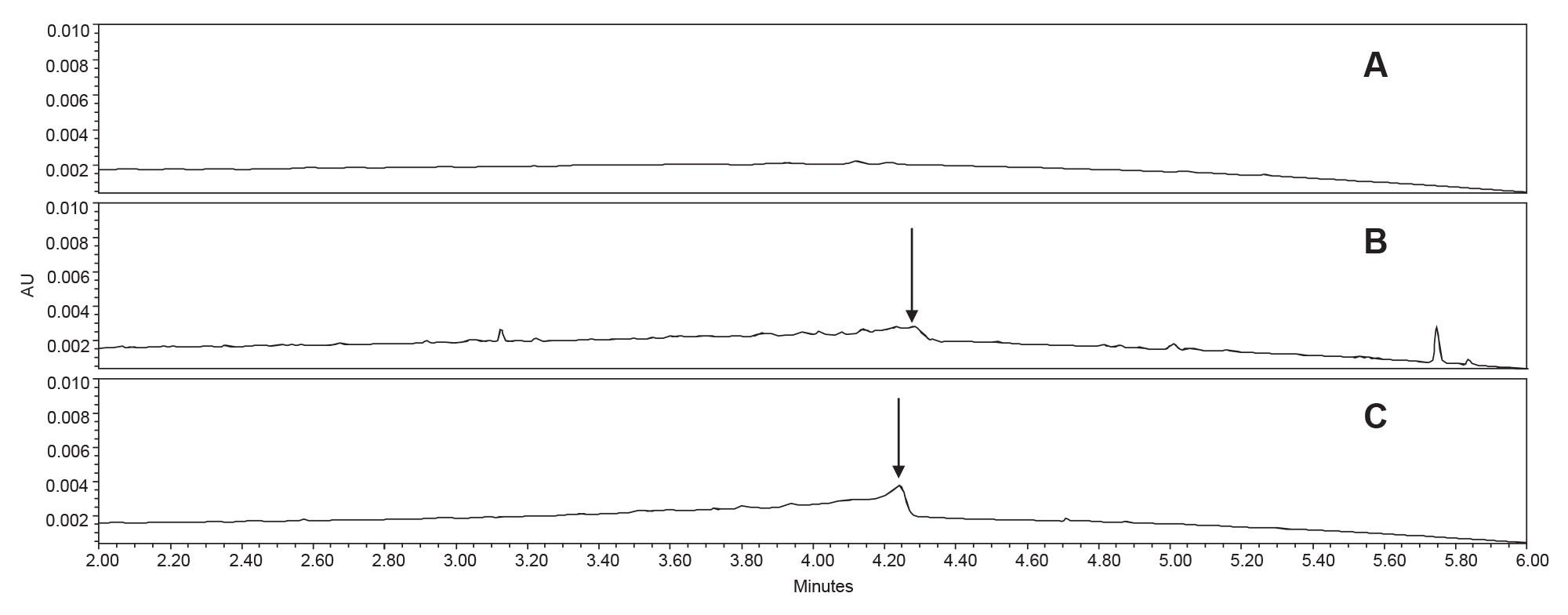
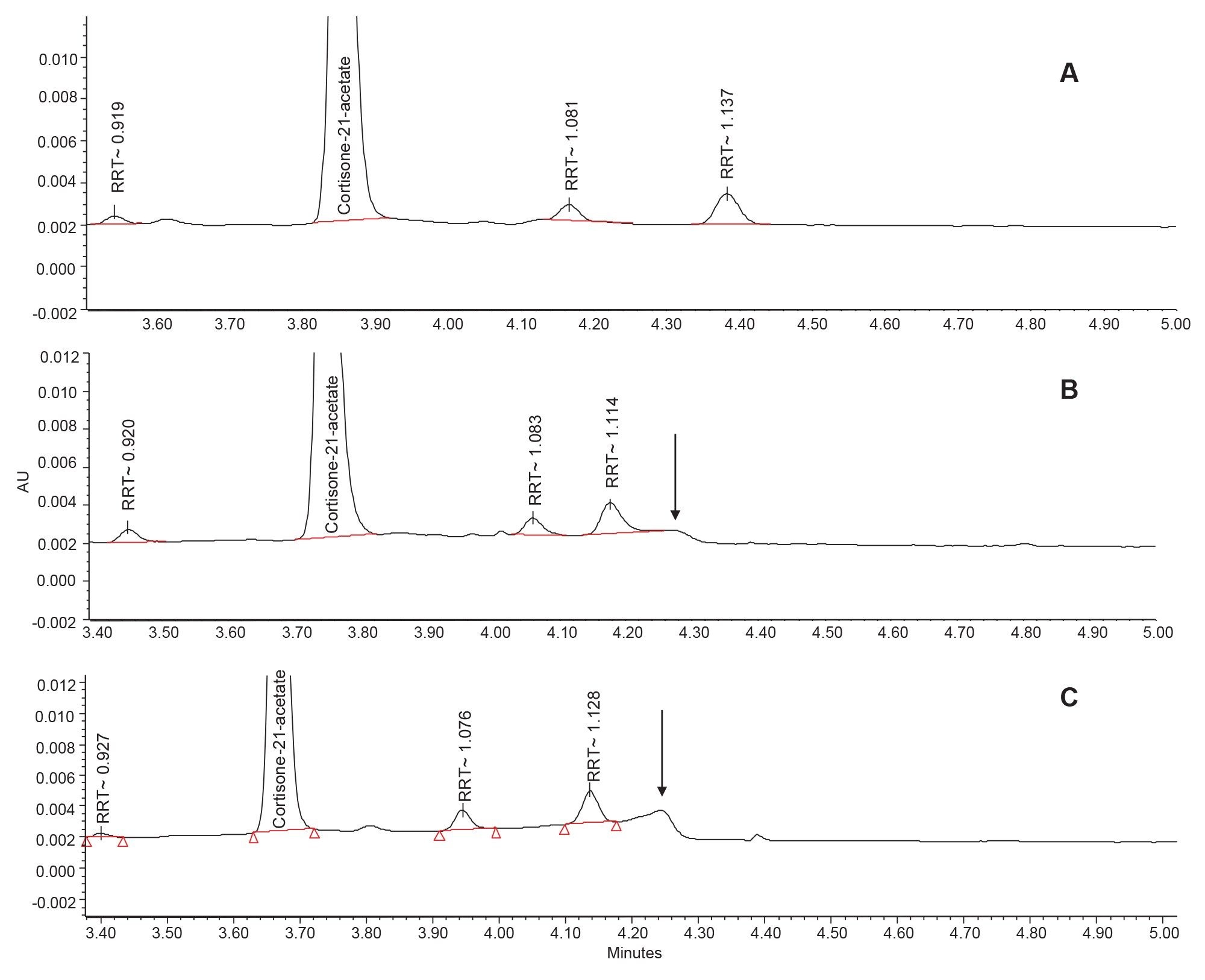
Column bleed can have an impact on the identification and quantitation of chromatographic peaks. This is especially apparent in pharmaceutical assays that require the precise integration of small impurity peaks, often with peak areas less than 1% of the total area. In the forced degradation of cortisone-21-acetate, there are several degradant peaks that elute in the region of the column bleed. One such peak is relative retention time (RRT) 1.137 using the XBridge Biphenyl Column (1.114 and 1.128 RRT for the Vendor R and Vendor A columns, respectively).
In the bottom and middle chromatograms in Figure 3, the impurity peak elutes close to the maxima of the column bleed, making accurate and precise area quantitation difficult. In contrast, the top chromatogram, which shows the separation obtained using the XBridge Biphenyl Column, shows a flat baseline with no column bleed. Additionally, the XBridge Biphenyl Column has greater retention of these analytes compared to the Vendor A column and better resolution of adjacent impurities than the Vendor R column.
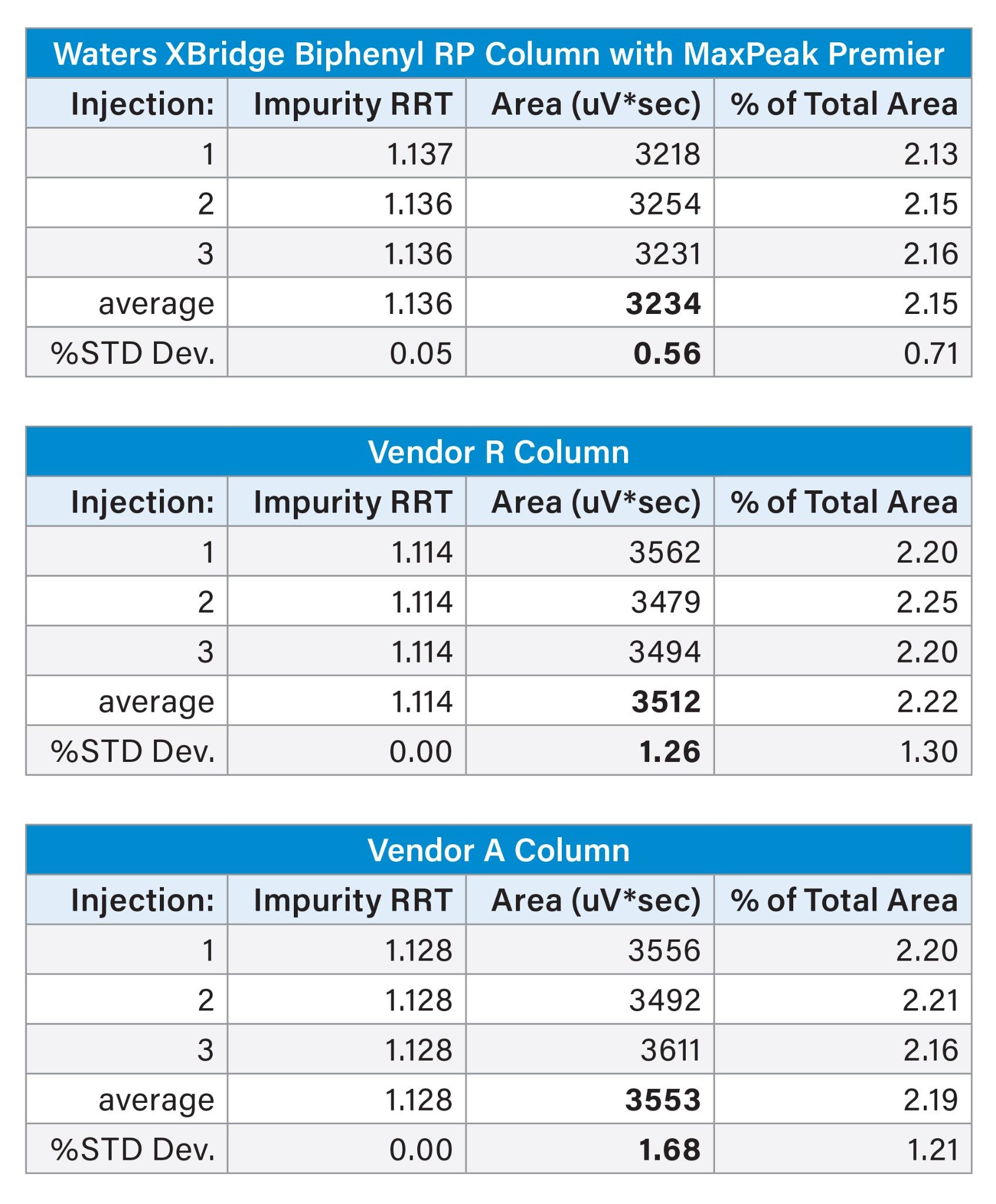
Triplicate injections of the forced degradation sample were made on an XBridge Biphenyl Column and Vendor A and Vendor R biphenyl columns. The peaks for impurity RRT 1.137 on the XBridge Biphenyl Column (1.114 and 1.128 RRT for the Vendor R and Vendor A columns, respectively) were integrated and the results averaged for three injections. The integrated area data is summarized in Table 1. The XBridge Biphenyl Column gave the most consistent peak area, with a relative standard deviation (RSD) of 0.56% compared to 1.26% and 1.68% for the Vendor R and Vendor A columns. Additionally, the peak areas are slightly overestimated with the Vendor R and A columns by about 10%, making the impurity percent of total area for the assay about 0.05% higher than determined using the XBridge Biphenyl Column. This is most likely due to the baseline distortion caused by column bleed, resulting in inaccuracy when integrating the small impurity peak.
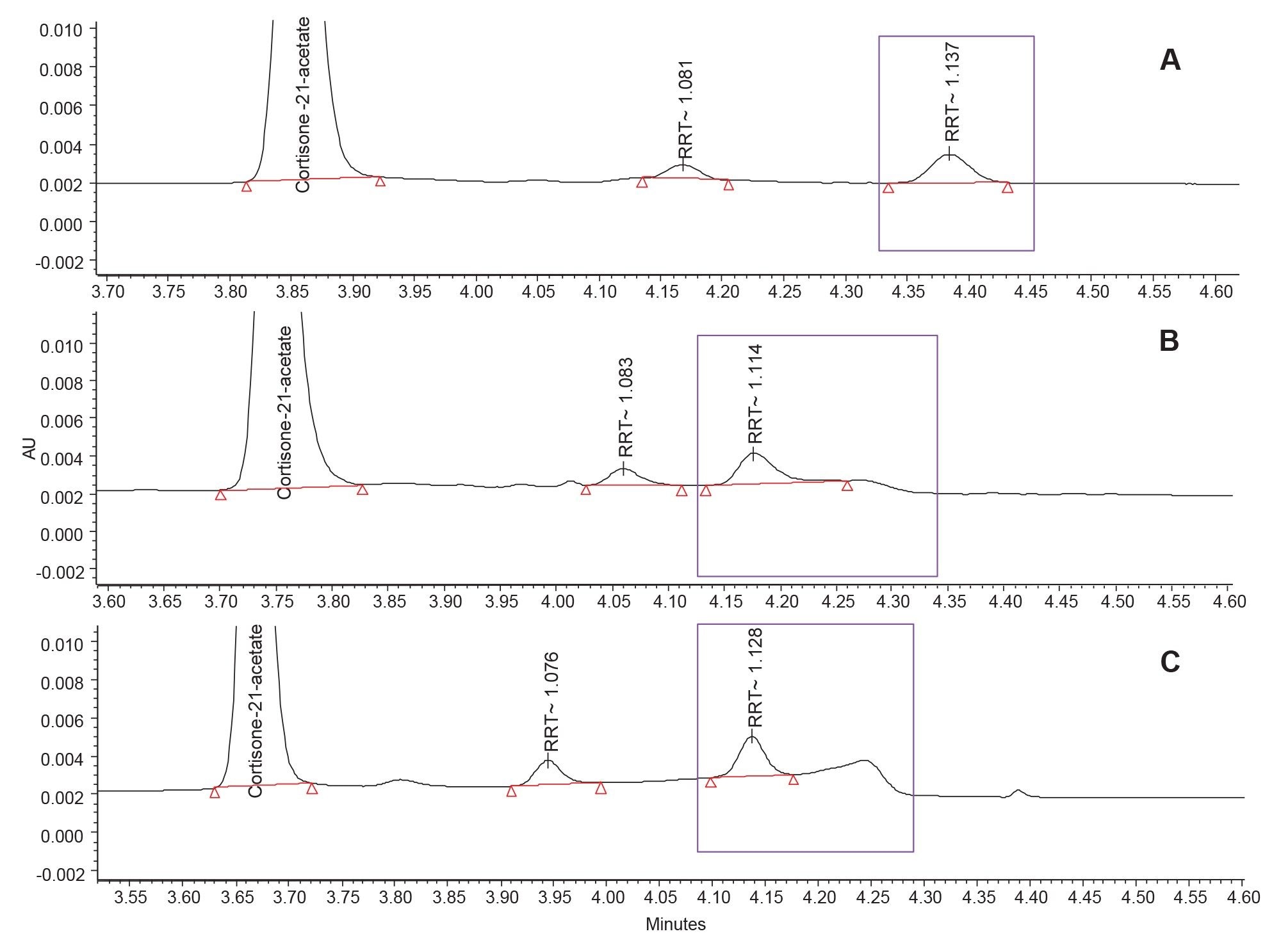
Enlarged chromatograms showing the integration of the impurity peak for each column are displayed in Figure 4. For the columns with significant baseline rise, it is difficult to quantitate the peak areas of the low-level impurities close to the ‘cliff’ of the bleed region.
Conclusion
Water XBridge Biphenyl RP Columns with MaxPeak Premier Technology not only provide alternative selectivity compared to C18 columns, but also address the shortcomings of other biphenyl columns. An important issue with other biphenyl columns is the propensity for baseline distortion in gradient separations when using UV detection due to elution of hydrolysis products of the bonded phase, which is called column bleed. The new XBridge Biphenyl Columns are stable over a wide pH range (pH 1.5–10), and show no column bleed under the acidic conditions used in this study. This allows for more accurate and precise quantitation of low-level impurities when using UV detection with gradient separations. This is crucial in a pharmaceutical QC setting, where government-regulated standards of allowed impurities direct batch testing requirements, and ultimately ensure the delivery of safe and effective drugs for patients.
References
- Case-Lo, C. (2018). Glucocorticoids: List, Uses, Side Effects, and More. Healthline. https://www.healthline.com/health/glucocorticoids.
- Muhtadi, F. J. Cortisone Acetate. Analytical Profiles of Drug Substances and Excipients. Academic Press, Volume 26,1999, Pages 167–245. ISBN 9780122608261.
- Lindner, J. M., Vogeser, M., Grimm, S. H. Biphenyl based stationary phases for improved selectivity in complex steroid assays. J. Pharm. Biomed. Anal., 142 (2017) 66–73. https://doi.org/10.1016/j.jpba.2017.04.020.
- Appulage, D. K., Wang, E. H., Carroll, F., Schug, K. A. Automated screening of reversed-phase stationary phases for small-molecule separations using liquid chromatography with mass spectrometry. J. Sep. Sci. 39 (2016) 1638–1647.
- Zabala, G., Alden, B., Boissel, C., Walter, T. H., Gu, W., Walsh, D. P., Cook, J. A Highly Stable Biphenyl HPLC Stationary Phase Based on Ethylene-Bridged Hybrid Particles. 720009261. Waters Application Note, 2026.
- Dolan, J. W. Gradient Elution, Part V: Baseline Drift Problems. LC GC North Am. 31(7) (2013) 538–543.
720009246, April 2026