Analysis of Amino Acids in a Supplement Using the Alliance™ iS HPLC System and AccQ•Tag™ Ultra Derivatization
Kimberly Martin, Paula Hong
Waters Corporation, United States
Published on April 17 2026
Abstract
Amino acid analysis (AAA) is fundamental to nutritional research and dietary supplement quality assessment, yet it remains analytically challenging. Many amino acids lack strong chromophores, requiring derivatization to enable sensitive detection. AccQ•Tag Ultra Derivatization Chemistry is a widely adopted and highly robust solution to these challenges. The pre-column derivatization method employs 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate (AQC), a reagent that rapidly and quantitatively labels both primary and secondary amines to form stable derivatives. When paired with reversed-phase chromatography, this chemistry delivers high sensitivity, excellent stability, and reproducible retention times, making it particularly well-suited for complex dietary supplement matrices.
In this study, the performance of the Alliance iS HPLC System in combination with AccQ•Tag Ultra Amino Acid Analysis Consumables will be assessed for quantitation of dietary supplements. The system supports optimized AccQ•Tag Chemistry workflows with 2.5 µm columns, providing precise quaternary gradient delivery, controlled column temperature, and sensitive UV detection. This approach will show the benefits of combining Alliance iS HPLC System and AccQ•Tag Column chemistry with advanced system intelligence, making it a strong choice for laboratories performing routine amino acid analysis in dietary supplement testing.
Benefits
- Alliance iS HPLC System in combination with AccQ•Tag Ultra 2.5 µm Columns, Reagents, and Consumables provides a reliable, robust method for HPLC analysis of both hydrolysates and cell culture amino acids, designed for regulated laboratories.
- The combination of system and consumables with Empower™ 3 Chromatography Data System (CDS) provides a turnkey approach for amino acid analysis of dietary supplements, allowing regulated laboratories to benefit from a modern, compliance-ready system, designed to reduce common setup mistakes.
Introduction
AAA is a foundational technique in analytical chemistry, supporting essential workflows in biopharmaceutical development, nutritional science, and cell culture optimization. In nutritional research, precise amino acid profiling informs dietary formulation, quality control, and food composition studies, making accurate and reproducible analytical methods critical for both clinical and industrial applications. Among available approaches, the AccQ•Tag Ultra Chemistry—combining the AQC derivatization reagent with dedicated columns and eluents—has become a well‑established methodology due to its exceptional sensitivity, robustness, stability, and reproducibility. The method uses pre‑column derivatization with 6‑aminoquinolyl‑N‑hydroxysuccinimidyl carbamate (AQC), which reacts quantitatively with both primary and secondary amines to produce highly stable fluorescent derivatives.
The reversed‑phase separation of AQC‑labeled amino acids with AccQ•Tag Ultra Columns and Reagents has demonstrated excellent reproducibility on the ACQUITY Arc™ System and Arc HPLC System.1 Achieving consistent performance requires an LC platform capable of delivering precise quaternary gradients, maintaining tight temperature control in the column compartment, and providing highly sensitive UV detection. In the following study, the method was transferred to the Alliance iS HPLC System with TUV detection for the analysis of amino acids in dietary supplements, demonstrating its suitability for regulated laboratories that require quantitation of amino acids.
Experimental
Sample Description
All calibration standards were prepared from Waters Amino Acid Cell Culture Standard Kit (p/n: 186009300) using norvaline (p/n: 186009301) as the internal standard and 0.1 N hydrochloric acid HCl) in water as the diluent.3 The internal standard stock was prepared at 2500 µM in 0.1 N HCl. The final concentration of the calibrants varied to match the content range of each supplement, and a concentration of 250 µM was used for norvaline (internal standard).
Amino Acid Supplement Capsule Sample Preparation
The amino acid supplement capsule sample (past expiry) was collected and weighed. Three capsules represent one serving; therefore, providing an adequate sample size for uniformity, nine capsules and three servings were prepared. The final weight of the nine capsules was 7255.76 mg. The sample was then diluted in 200 mL of 0.1 N HCl in water. The sample was then sonicated for 1.5 hours followed by stirring for 1 additional hour. The sample was filtered using a 0.45 um HA filter.
An additional dilution of 1:50 was performed on the sample extraction, making the final dilution of the sample 1:10,000. The sample was then derivatized using Waters AccQ•Tag Ultra Derivatization Protocol.4
LC Conditions
|
LC system: |
Alliance iS HPLC System |
|
Detection: |
TUV Detector |
|
Wavelength: |
260 nm |
|
Sampling rate: |
10 Hz |
|
Vials: |
LCGC Certified Clear Glass 12 x 32 mm Screw Neck Vial, total recovery with cap and PTFE/Silicone septum (not pre-slit) (p/n: 186000384C) |
|
Column(s): |
AccQ•Tag Ultra C18, 2.5 µm 4.6 x 150 mm Column (p/n: 186010407) |
|
Column temperature for hydrolysate and cell culture standard: |
43 °C |
|
Sample temperature: |
20 °C |
|
Injection volume: |
2 µL |
|
Flow rate: |
1.5 mL/min |
|
Mobile phase A: |
AccQ•Tag Ultra Eluent A (p/n: 186003838) |
|
Mobile phase B: |
90:10 (v/v) Water:AccQ•Tag Ultra Eluent B |
|
Mobile phase C: |
Milli-Q Water |
|
Mobile phase D: |
AccQ•Tag Ultra Eluent B (p/n: 186003839) |
|
Sample manager wash: |
95:5 (v/v) Water:Acetonitrile |
|
Sample manager purge: |
95:5 (v/v) Water:Acetonitrile |
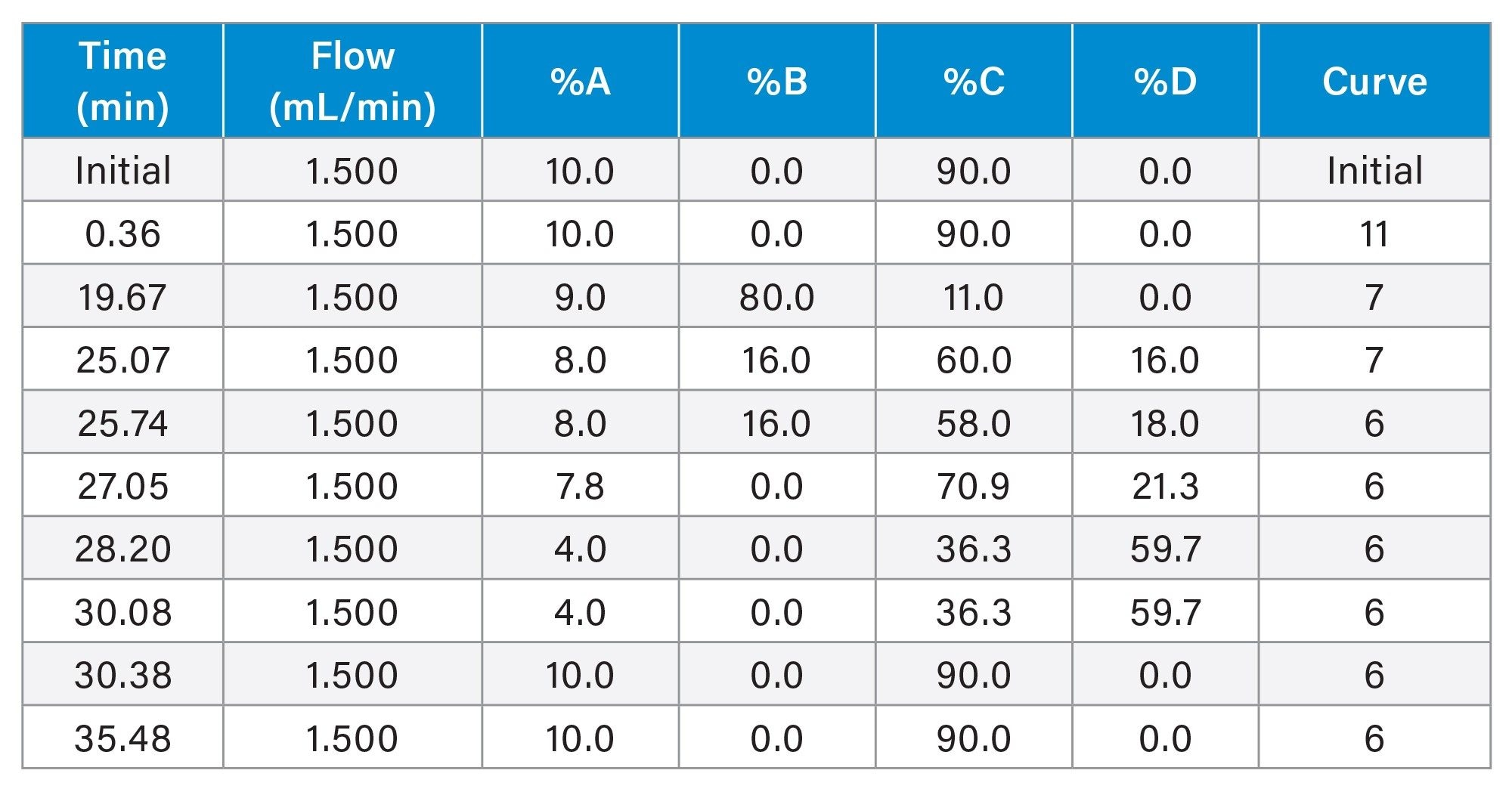
Gradient Table
Data Management
|
Chromatography data system: |
Empower 3.9.0 CDS |
Results and Discussion
Amino Acid Profile of a Dietary Capsule Supplement Sample
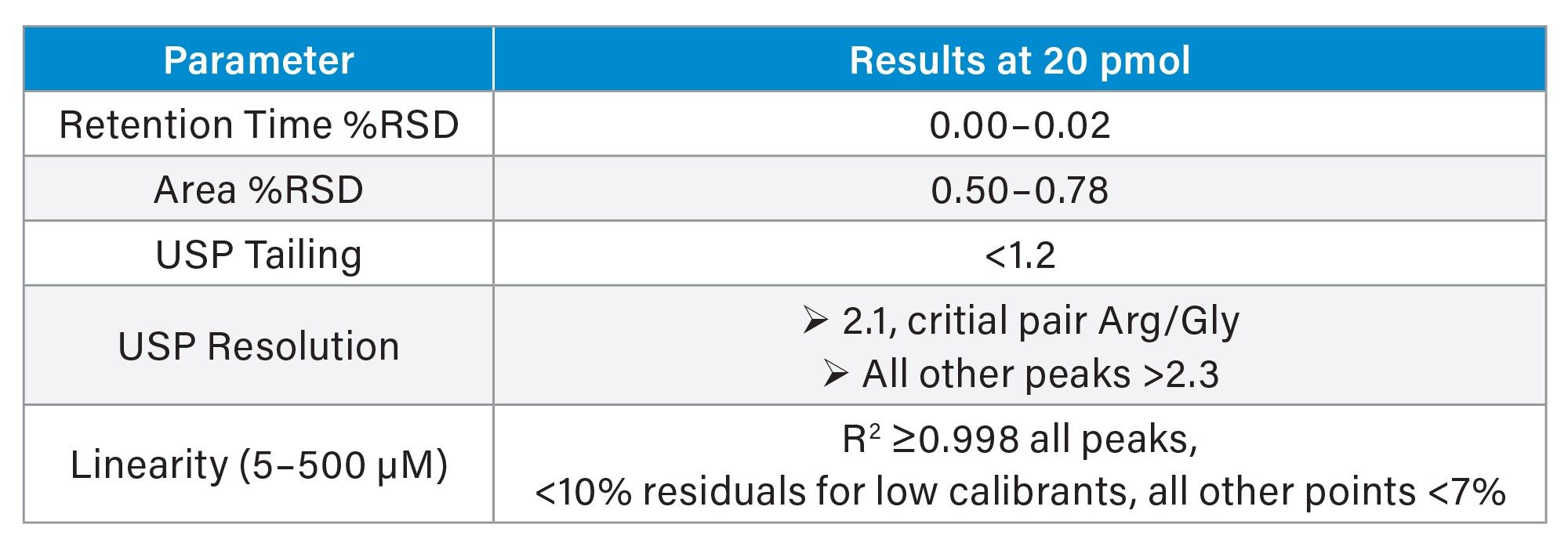
To assess the robustness of the method, the method that was previously verified on the ACQUITY Arc HPLC System1 was transferred to the Alliance iS HPLC TUV System. For this testing, the cell culture standard was evaluated on the Alliance iS HPLC System for repeatability, linearity, and sensitivity. Specific parameters tested included limits of detection and quantitation (LOD/LOQ), calibration linearity, and intraassay precision, producing comparable results as previously observed.1 These findings in Table 1 indicate that the Alliance iS HPLC System provides consistent performance to support robust amino acid analyses, meeting method criteria to support quantitation. The chromatographic separation for amino acid hydrolysate and cell culture standards are shown in the stacked chromatogram overlay in Figure 1.
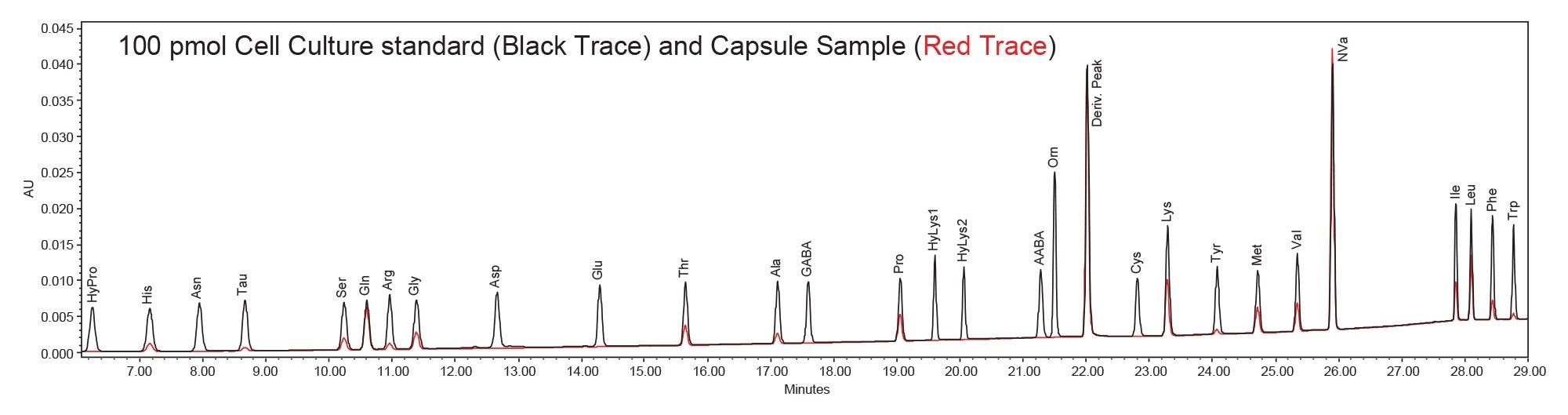
Following evaluation of the method performance, commercial amino acid capsule supplements were analyzed to quantitatively determine their amino acid content relative to manufacturer label claims. Given the complex sample matrix, the supplements were extracted prior to derivatization. Due to the complexity of the sample components, some optimization of sample preparation was conducted. The amino acid derivatives in the matrix were effectively resolved using reversed-phase liquid chromatography under optimized gradient conditions, providing consistent retention and peak shape across the analyte panel.1 No interference peaks were observed. The amino acid capsule supplement sample and amino acid cell culture standard is shown in Figure 2.
Quantitative results were compared with declared label values to assess accuracy and compliance. This type of assessment is essential for verifying product integrity, supporting quality assurance processes, and aligning with regulatory expectations for dietary supplement labeling. The recovery data demonstrated that the analytical workflow—AQC derivatization combined with RPLC separation on the Alliance iS HPLC System—delivered reliable performance across all amino acids assessed. Recoveries ranged from 75% to 113%, with the majority falling within the preferred 88–99% range, indicating strong agreement with expected concentrations. These results are consistent with typical performance for amino acid analysis in complex supplement matrices, where extraction efficiency and matrix interactions often influence final recoveries.
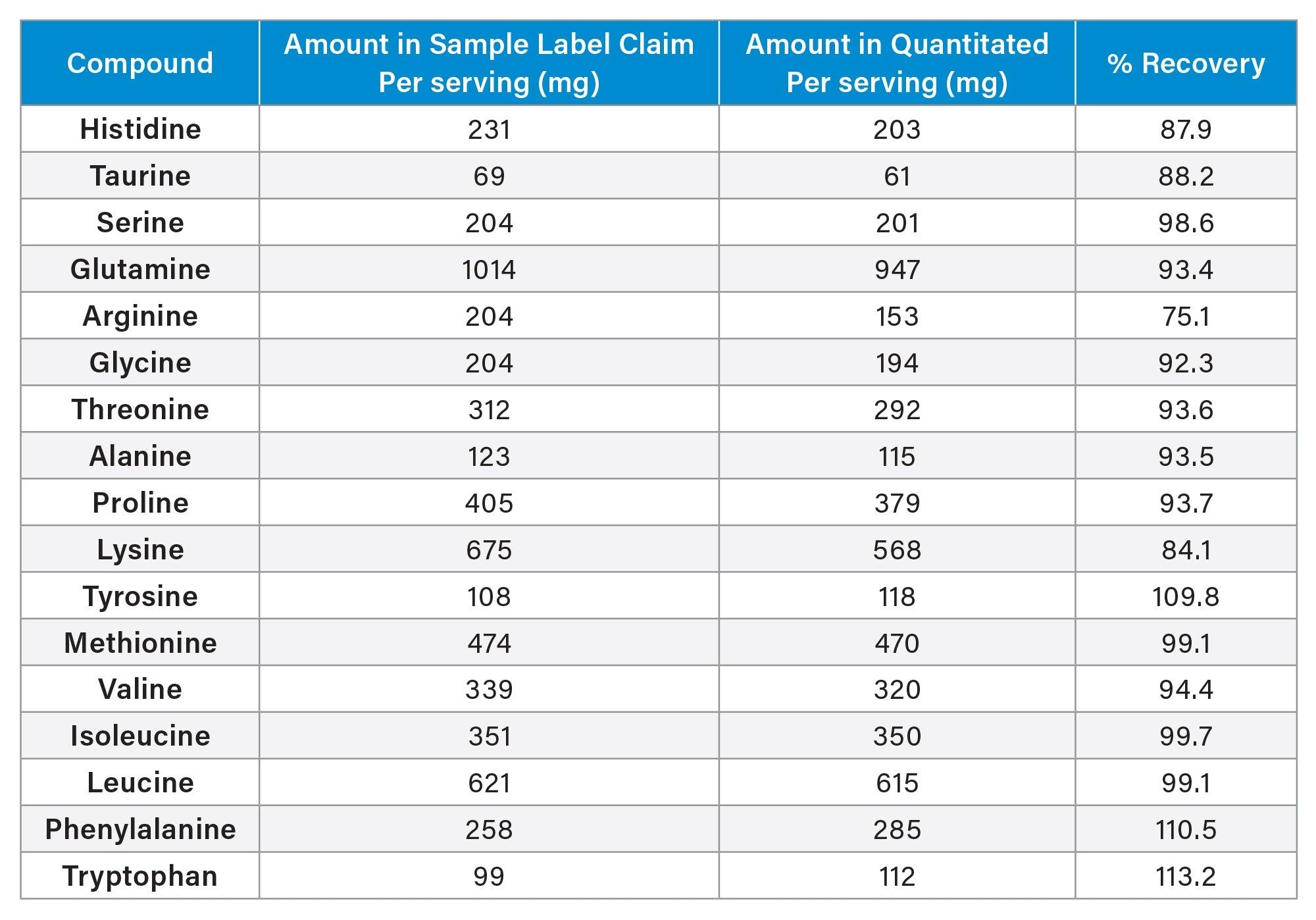
A few amino acids exhibited recoveries near the upper or lower bounds of the observed range. These deviations may arise from incomplete extraction, matrix suppression or enhancement, or inherent variability in derivatization efficiency among certain amino acids. Although these results remain within acceptable limits for dietary supplement analysis, further optimization of the extraction procedure—such as evaluating alternative solvent systems, adjusting extraction time, or incorporating additional mechanical agitation—may help improve recoveries.
Overall, the tight clustering of recoveries for most amino acids reflects the robustness of both the derivatization chemistry and the chromatographic method. AQC reagent chemistry is well characterized for its stability and efficient derivatization of diverse amino acids, enabling reproducible fluorescence based or UV based detection. The Alliance iS HPLC System further enhanced method reproducibility through intelligent instrument monitoring, stable flow control, and environmental consistency, reducing variability during routine operation.
Conclusion
Accurate quantification of amino acids in dietary supplements is essential for verifying label claims, ensuring product integrity, and meeting regulatory expectations for nutritional product labeling and quality control. Because amino acids serve as key functional ingredients, analytical methods must exhibit high precision, accuracy, and robustness to satisfy product quality.
In this study, the combination of AQC precolumn derivatization and reversed-phase HPLC separation on the Alliance iS HPLC System produced strong analytical performance across a diverse amino acid panel. Percent recoveries ranged from 75% to 113.2% relative to manufacturer label claims, with most amino acids falling within the preferred 88%–99% range. These results highlight the effectiveness of the AccQ•Tag Ultra Derivatization Chemistry as well as the chromatographic stability, reproducibility, and overall reliability of the Alliance iS HPLC System.
Overall, the precision and recovery results validate the robustness of the analytical workflow and confirm the suitability of the Alliance iS HPLC System for routine quality control applications in regulated laboratory environments. The strong alignment between measured concentrations and declared label values supports product integrity and reinforces confidence in the method’s quantitative accuracy. By integrating advanced intelligent instrumentation with rigorously validated chromatographic methodologies, the Alliance iS HPLC System enables laboratories performing amino acid analysis to maintain analytical robustness, achieve reproducible data quality, and uphold regulatory compliance without sacrificing operational efficiency or throughput.
References
- Martin, K.; Berthelette, K.D.; Hong, P. Instrument Considerations for Reliable Amino Acid Analysis Using AccQ•Tag Ultra C18 2.5 µm Column. Waters Application Note. 720007678. 2022.
- FDA, Guide for Industry Q6B Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products. Q6Bfnl.PDF. 1999.
- Amino Acids Standard Kits Care and Use Manual, Waters Corporation Care and Use Manual. 720006663. 2024.
- AccQ•Tag Ultra Derivatization Kit Care and Use Manual, Waters Care and Use Manual, 715001331. 2014.
720009316, April 2026