Extraction and Analysis of Orthocetamol, Metacetamol and Paracetamol in Human Plasma Using Oasis™ PRiME HLB and Waters ACQUITY™ Biphenyl RP Column with MaxPeak™ Technology
Kenneth Berthelette, Chris Collins, Thomas H. Walter
Waters Corporation, United States
Published on April 13, 2026
Abstract
The positional isomers orthocetamol, metacetamol and paracetamol were analyzed using a Waters ACQUITY Biphenyl RP Column with MaxPeak Technology. Subsequently, a suitable solid-phase extraction (SPE) method for extracting these compounds from pooled human plasma was created using the Oasis PRiME HLB 2-step protocol. Baseline separation of the three structurally similar compounds combined with high recovery indicates that this workflow would be suitable for monitoring these compounds in a bioanalytical setting.
Benefits
- Baseline separation of acetaminophen and its ortho- and meta- positional isomers
- >90% recovery of all three analytes from spiked human plasma
Introduction
Structurally similar components can be challenging to separate in reversed-phase liquid chromatography (RPLC) due to their comparable chemical properties. Positional isomers present an even tougher challenge as not only may they have similar chemical properties affecting their retention and elution, but they also have the same mass to charge ratio (m/z), making them difficult to distinguish by mass spectrometry (MS). To identify isobaric compounds, individual standards are often analyzed to establish identifications by retention time as well as m/z. This methodology requires not only a robust method which achieves baseline separation of the isomers, but also a stationary phase that produces reproducible results from batch to batch. In this application note, three positional isomers were analyzed. Orthocetamol, metacetamol and paracetamol were analyzed as neat standards and subsequently spiked into pooled human plasma. The plasma was then subjected to SPE to remove phospholipids and other matrix components which may affect the analysis or the lifetime of the column. SPE was performed using an Oasis PRiME HLB 96-well plate on a semi-automated positive pressure manifold using a 2-step protocol to minimize sample preparation time.1
ACQUITY BEH™ Biphenyl Column was used for this application for important reasons. First, these columns employ the highly reproducible ethylene-bridged hybrid (BEH) particle, which not only is stable across a wide range of mobile phase pHs, but also produces excellent batch to batch results as seen in the literature.2,3 Second, this column uses MaxPeak Premier High-Performance Surface (HPS) technology, which mitigates interactions between analytes and the column hardware.4 This technology also reduces variability caused by differences in the metal hardware.5 Lastly, the biphenyl bonded ligand has been shown to improve separations of isomers in biological assays.6,7 Using this column should provide the best separation possible for these compounds, while ensuring that the method is developed on a robust and reproducible stationary phase that can be used for the lifetime of the assay.
Experimental
Sample Description
A single stock solution was created with 1 mg/mL of each of the three compounds:, paracetamol metacetamol and orthocetamol. Pre-spike plasma was spiked with stock solution to a concentration of 80 µg/mL. Pre-spike plasma, post-spike plasma, and non-matrix post spike were diluted 1:1 with 4% phosphoric acid in water and vortexed to mix. Samples were then passed through an Oasis PRiME HLB 96-well plate (p/n: 186008054) using a 2-step protocol on a semi-automated positive pressure manifold. Post-spike plasma and non-matrix post-spike sample were then spiked to a concentration of 40 µg/mL. Lastly, all samples were diluted with water for analysis. The final analyte concentration was 20 µg/mL for each isomer.
LC Conditions
|
LC system: |
ACQUITY UPLC™ H-Class Plus with CM and CM Aux, PDA Detector and ACQUITY QDa™ Mass Detector |
|
Detection: |
SIR (single ion recording) 152 m/z |
|
Column: |
Waters Acquity Biphenyl RP Column with MaxPeak Technology, 2.1 x 50 mm, 1.7 µm (p/n: 186011744) |
|
Column temperature: |
30 °C |
|
Sample temperature: |
10 °C |
|
Injection volume: |
1.0 µL |
|
Flow rate: |
0.4 mL/min |
|
Mobile phase A: |
Water |
|
Mobile phase B: |
Methanol |
|
Mobile phase D: |
2% formic acid in water |
|
Gradient conditions: |
Constant 5% D to maintain mobile phase additive concentration; initial mobile phase conditions 5% B; linear ramp to 95% B in 5.00 minutes; hold for 1 minute; return to initial conditions and re-equilibrate for 2.00 minutes; total run time 8.00 minutes. |
MS Conditions
|
MS system: |
ACQUITY QDa Mass Detector |
|
Function: |
SIR 152 m/z |
|
ESI polarity: |
Positive |
|
Capillary voltage (kV): |
1.5 |
|
Cone voltage (V): |
15 |
|
Scan rate (pts/sec): |
5 |
Data Management
|
Chromatography software: |
Empower™ Chromatography Data System (CDS) |
Results and Discussion
The three positional isomers whose structures are shown in Figure 1 were analyzed using an ACQUITY BEH Biphenyl Column. Acetaminophen (paracetamol), and its ortho and meta positional isomers were selected as model compounds for this work. Once a suitable separation was achieved, the compounds were spiked into pooled human plasma.

Plasma samples and non-matrix samples (neat solutions) were processed using an Oasis PRiME HLB 30 mg plate and the 2-step protocol. Oasis PRiME HLB \ SPE devices are designed to remove phospholipids from biological fluids to reduce matrix effects and extend column lifetime.8 The 2-step protocol is shown in Figure 2.
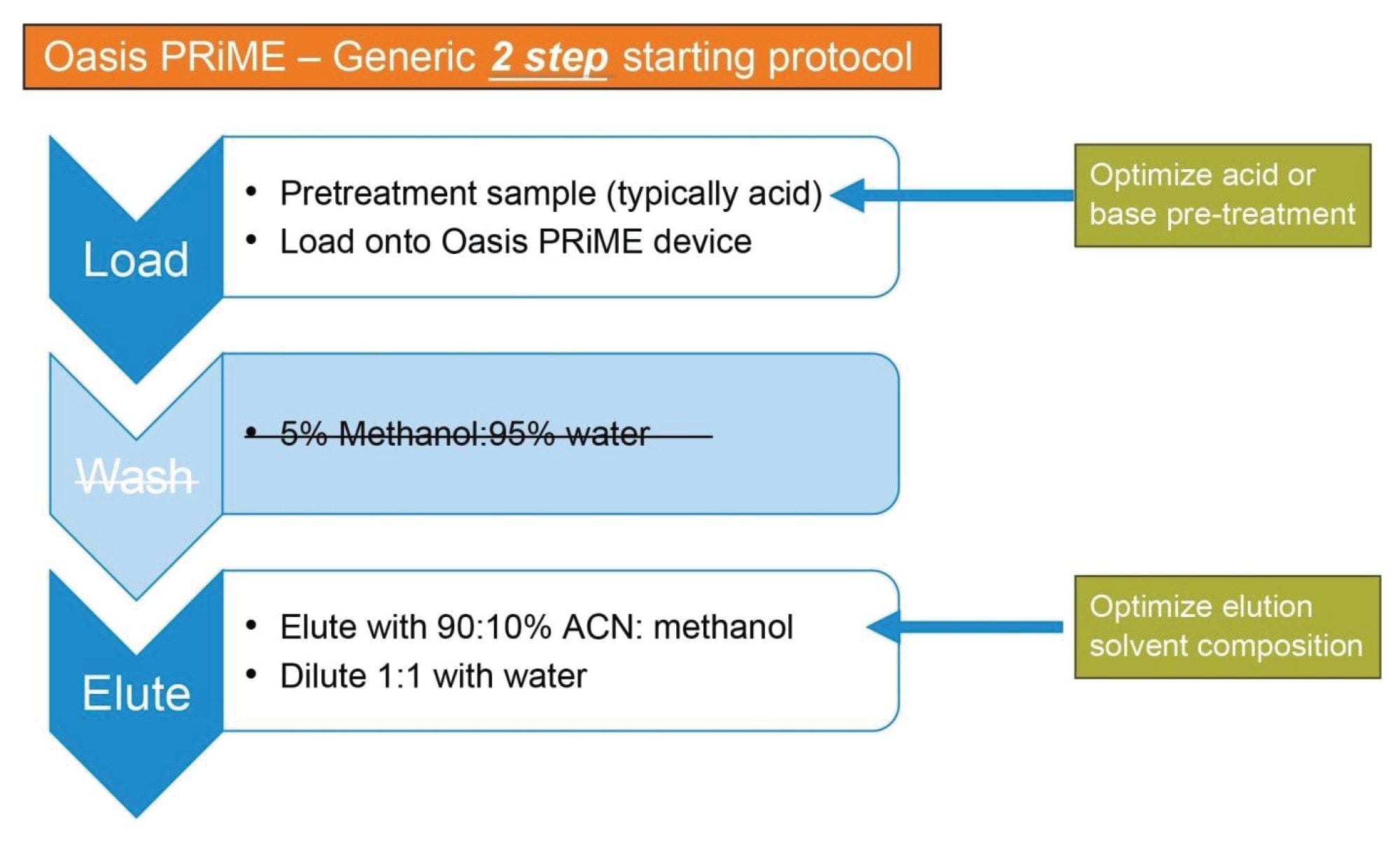
The 2-step protocol, which includes load and elute steps, was specifically developed to reduce sample preparation time without sacrificing recovery and is suitable as a first step in most bioanalytical workflows to extract analytes of interest by hydrophobic interactions. Prior to loading the samples onto the SPE device, a pre-treatment step is employed to ensure the analytes are in a neutral charge state. This improves retention on the SPE sorbent while allowing polar interferences, like salts, to pass through during the load step. Depending on the nature of the samples, a wash step may be needed to improve the SPE results. However, for initial scouting, the 2-step protocol gives adequate removal of matrix interferences, providing a good basis for further development work. The elute step uses a high organic content solvent to elute the analytes from the device. Due to the proprietary design of the Oasis PRiME HLB sorbent, phospholipids are retained even under high organic conditions, eliminating them from the final eluate, producing a cleaner sample. A final dilution with water is recommended to ensure that the elution solvent does not affect the chromatographic separation.
The three analytes of interest were easily detected by mass spectrometry using SIR at 152 m/z in pre-spike plasma after cleanup, with good MS signal using an ACQUITY QDa Mass Detector. A blank plasma sample was also processed using the 2-step protocol to determine if any of the analytes are endogenous in the plasma. Figure 3 shows representative chromatograms of the pre-spike and blank plasma samples.
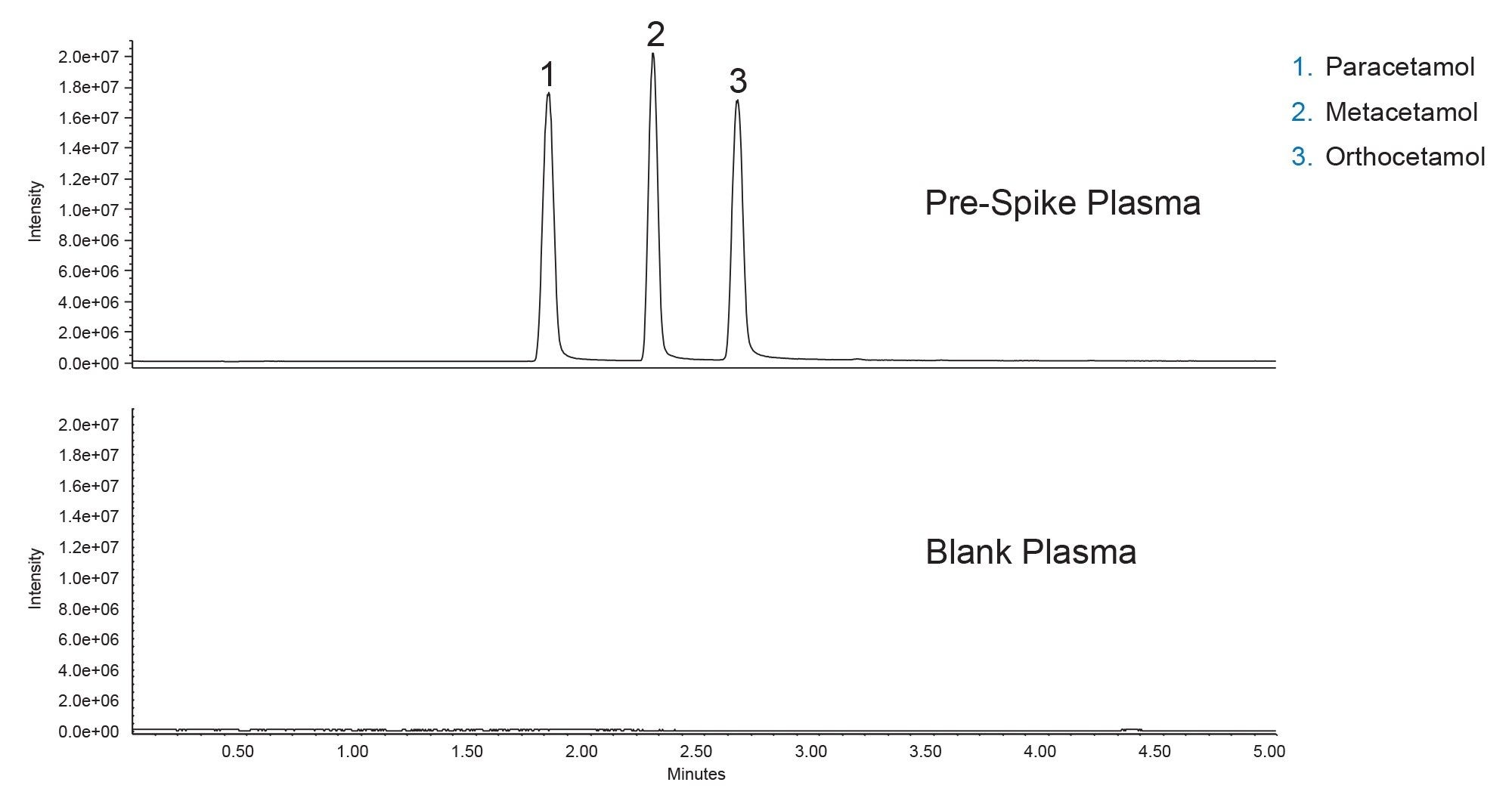
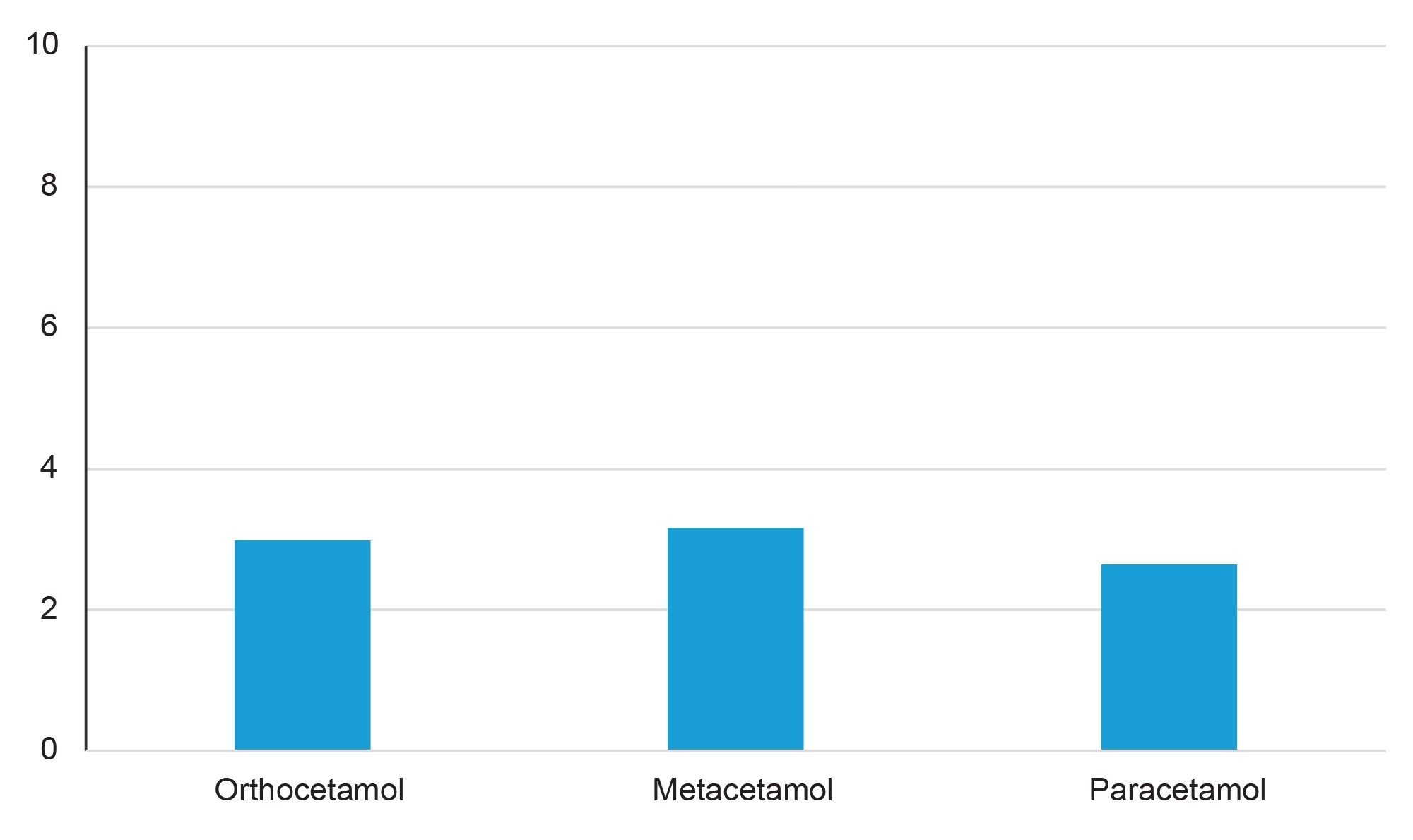
The BEH Biphenyl Column provided baseline resolution of the positional isomers, with sharp symmetrical peaks. The blank plasma sample shows none of the analytes of interest, indicating that the spiked amounts should be accurate. The selective nature of SIR eliminates background noise and improves overall sensitivity. The presence of endogenous compounds would have altered the final concentrations and could have affected recovery or matrix effect calculations. However, as the blank plasma showed no compounds present, the spiked amount should be the correct concentration in the samples. Calculations of % recovery and matrix effects showed great recovery with minimal matrix effects (see Figure 4 and Figure 5).

Conclusion
With high recoveries, low matrix effects, baseline separation and good peak shape for all analytes, the use of an Oasis PRiME HLB SPE 96-well plate and an ACQUITY Premier BEH Biphenyl Column yielded a method for analyzing the positional isomers orthocetamol, metacetamol and paracetamol in human plasma.
References
- Tanna N, Plummer C. Oasis PRiME HLB – The Fastest Way to Clean Samples. Waters Application Note. 720008684, 2025.
- Alkhateeb F, Jenkins K, Rainville P. BEH C18 Batch-to-Batch Robustness for the Analysis of Rosuvastatin and Impurities. Waters Application Note. 720006739, 2020.
- Walter TH, Yang J. Assessment of the Reproducibility of a C18 Bonded Phase Based on Ethylene-Bridged Hybrid Organic/Inorganic Particles, J. Chromatogr. A, 1716 (2024) 464637. doi: 10.1016/j.chroma.2024.464637.
- DeLano M, Walter TH, Lauber MA, Gilar M, Jung MC, Nguyen JM, Boissel C, Patel AV, Bates-Harrison A, Wyndham KD, Using Hybrid Organic-Inorganic Surface Technology to Mitigate Analyte Interactions with Metal Surfaces in UHPLC, Anal. Chem. 93 (2021) 5773-5781. doi: 10.1021/acs.analchem.0c05203.
- Alkhateeb F, Rainville P. Batch-to-Batch Robustness of MaxPeak Premier Columns for the Analysis of Dexamethasone Phosphate and Related Compounds. Waters Application Note. 720007350, 2021.
- Masike K, Tugizimana F, Ndlovu N, Smit E, Preez L, Dubery I, Madala E. Deciphering the Influence of Column Chemistry and Mass Spectrometry Settings for the Analyses of Geometrical Isomers of L-Chicoric Acid. J Chrom B 1052, (2017), 73-81.
- Lindner J, Vogeser M, Grimm SH. Biphenyl Based Stationary Phase for Improved Selectivity in Complex Steroid Assays. J Pharm Bio Anal. 142 (2017) 66-73.
- Tanna N, Plummer C, Walter TH. Improved Bioanalytical Performance Using a Simplified 2-Step Oasis PRiME HLB Protocol with an Ultra-Short UPLC Column for UPLC-MS/MS. Waters Application Note. 720009018, 2025.
720009277, April 2026