Global regulators increasingly expect detailed analytical and biological evidence to demonstrate comparability between biosimilars and reference products. This requires developers to thoroughly assess multiple protein batches, emphasizing precise biosimilar protein characterization and deeper characterization of biotherapeutic proteins.
Waters’ integrated solutions provide a comprehensive platform for streamlined, reliable analysis of biosimilar and reference product attributes, including:
Establish biosimilar comparability with confidence—partner with Waters for safer, more effective medicines.
Regulatory Insight: FDA Draft Guidance (2025)
Recent FDA draft guidance enables developers to bypass comparative clinical efficacy studies if advanced analytical data provides sufficient evidence of biosimilarity—potentially eliminating one of the costliest development steps and accelerating timelines dramatically.
This shifts an even greater responsibility onto biologics and biosimilars protein analysis and robust analytical testing.

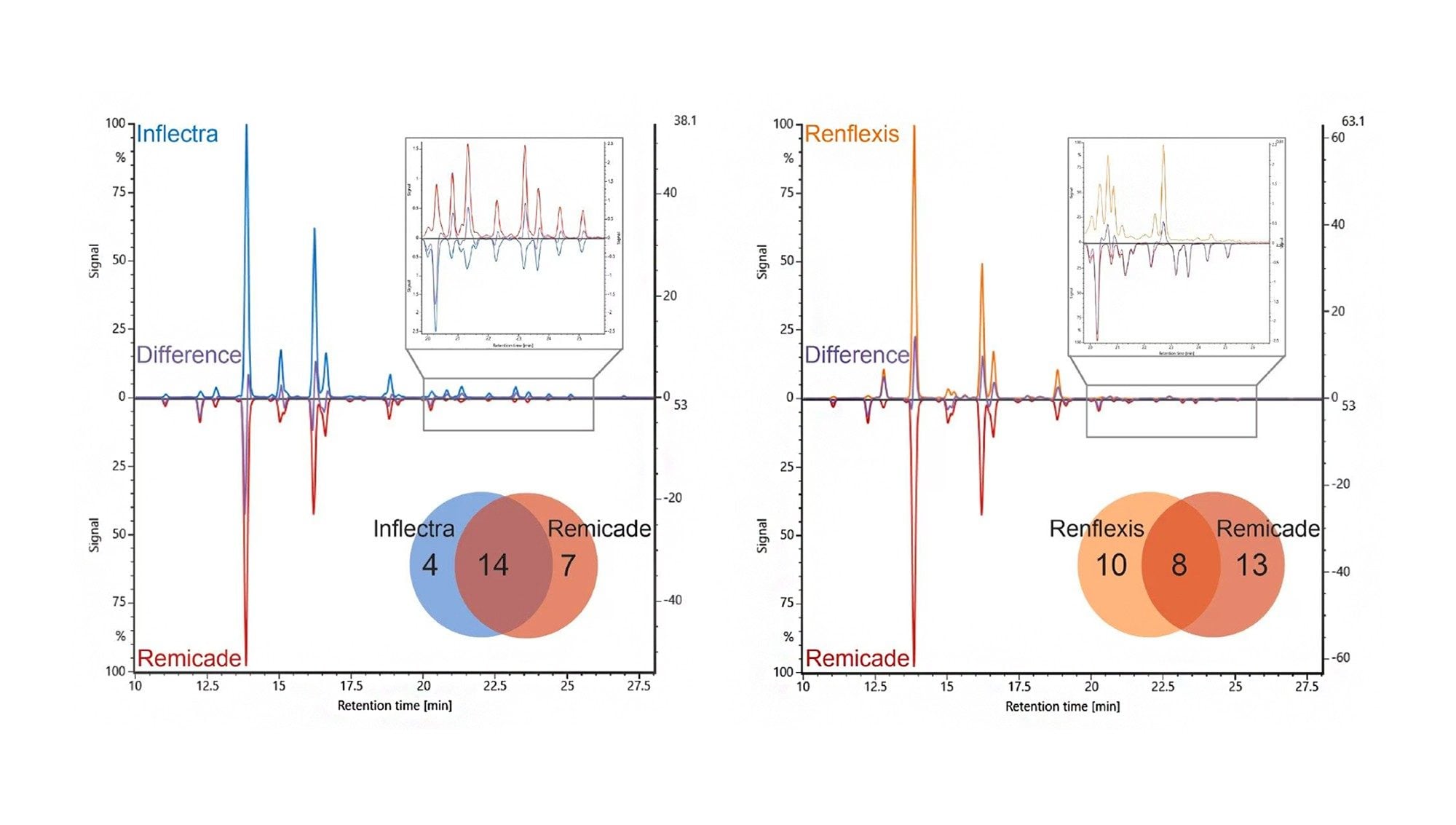
Peptide mapping is essential for establishing similarity through high sequence coverage and the detection of clinically relevant modifications at the peptide level. Non‑reducing digests further support characterization of biosimilars by enabling disulfide linkage confirmation and equivalence verification to innovator molecules.
Waters solutions deliver high‑precision data to support faster decision‑making across development and manufacturing.
Peptide mapping is essential for establishing similarity through high sequence coverage and the detection of clinically relevant modifications at the peptide level. Non‑reducing digests further support characterization of biosimilars by enabling disulfide linkage confirmation and equivalence verification to innovator molecules.
Waters solutions deliver high‑precision data to support faster decision‑making across development and manufacturing.
Protein higher‑order structure is critical in determining functional equivalence.
Comparing structural profiles to the reference product reveals potential functional differences or confirms acceptable similarity.
Waters’ structural biology solutions integrate precise chromatography, robust sample management, and advanced mass spectrometry for efficient acquisition and interpretation of complex HOS datasets.
HDX‑MS and CIU enable detailed assessment of conformational dynamics and stability—key elements in advanced biosimilar characterization workflows.
Protein higher‑order structure is critical in determining functional equivalence.
Comparing structural profiles to the reference product reveals potential functional differences or confirms acceptable similarity.
Waters’ structural biology solutions integrate precise chromatography, robust sample management, and advanced mass spectrometry for efficient acquisition and interpretation of complex HOS datasets.
HDX‑MS and CIU enable detailed assessment of conformational dynamics and stability—key elements in advanced biosimilar characterization workflows.
Glycosylation affects efficacy, safety, and immunogenicity.
Understanding glycan patterns is essential for demonstrating equivalent quality, consistency, and manufacturing robustness. Waters offers leading solutions for analysis of glycopeptides and released N‑glycans, providing deep insight into major and minor glycoforms to support comparability and process control.
Glycosylation affects efficacy, safety, and immunogenicity.
Understanding glycan patterns is essential for demonstrating equivalent quality, consistency, and manufacturing robustness. Waters offers leading solutions for analysis of glycopeptides and released N‑glycans, providing deep insight into major and minor glycoforms to support comparability and process control.
Multi‑Attribute Biosimilarity Assessment (MALS)
Absolute molecular weight determination and high‑resolution aggregation and fragmentation profiling are critical for establishing biosimilarity. The DAWN detector enables confident structural comparability across development, supporting sensitive detection of clinically relevant differences without reliance on reference standards.
Comprehensive Particle Characterization for Biosimilars
Robust visible and subvisible particle characterization supports biosimilar comparability by enabling early risk identification, consistent control strategies, and alignment with regulatory expectations. High‑throughput particle data across development and QC from Aura Particle Analyzers, helps demonstrate equivalence in product quality throughout the biosimilar lifecycle.
Multi‑Attribute Biosimilarity Assessment (MALS)
Absolute molecular weight determination and high‑resolution aggregation and fragmentation profiling are critical for establishing biosimilarity. The DAWN detector enables confident structural comparability across development, supporting sensitive detection of clinically relevant differences without reliance on reference standards.
Comprehensive Particle Characterization for Biosimilars
Robust visible and subvisible particle characterization supports biosimilar comparability by enabling early risk identification, consistent control strategies, and alignment with regulatory expectations. High‑throughput particle data across development and QC from Aura Particle Analyzers, helps demonstrate equivalence in product quality throughout the biosimilar lifecycle.
Webinar: Deploying Waters Solutions to Enhance Biosimilar Development

Streamline biosimilar analysis workflows and unify LC‑MS systems across organizations with this compliant‑ready, networked informatics platform.
BioResolve RP mAb Polyphenyl Columns provide improved peak capacity and minimal carry-over for intact and subunit analysis of proteins and mAbs specifically QC-tested with a NIST-based, mAb subunit standard. BioResolve SCX mAb Columns and Buffers reliably separate charge variants for a wide range of mAbs using optical or LC-MS compatible mobile phases.
Minimize secondary ionic or hydrophobic interactions in size-based separations with ACQUITY and XBridge Premier Protein SEC 250 Å Columns with MaxPeak High Performance Surface (HPS) Technology for reliable protein aggregate, monomer, and fragment analyses using a “platform” type method with 30% increase in resolution.
Gain enhanced recovery and sensitivity for acidic peptides with Waters ACQUITY, XSelect, and XBridge Premier Peptide Columns that are QC batch-tested with Waters Cytochrome c Digest Standard for increased column-to-column performance consistency.
Achieve quick, clean, and complete mAb and biosimilar digestions in just 30 minutes with RapiZyme Trypsin, Mass Spectrometry (MS) Grade, a highly activie autolysis-resistant enzyme that reduces the likelihood of artificial modifications occurring during sample preparation prior to LC-MS analysis.
Achieve high efficiency, reproducible peptide maps in under 2.5 hours with PeptideWorks Tryptic Protein Digestion Kits for manual or automated workflows that are 4x faster than average home-brew methods and provide a 78% reduction in missed cleavages.
Obtain fast, high resolution glycan identification for your biosimilars with Waters complete line of glycan columns to analyze glycoproteins, glycopeptides and released N-glycans with exceptional reproducibility.
Achieve unprecedented fluorescent and mass spectrometric performance for glycan detection with GlycoWorks N-glycan Kits that improve the throughput of N-glycan sample preparation so you don’t have to compromise between speed and sensitivity.
Fast, efficient, and reproducible online pepsin digestion is critical in experiments that require more peptide coverage. With the Waters Enzymate BEH Pepsin Column, you can easily incorporate online protein digestion into existing HDX- MS workflows. The Enzymate BEH Pepsin Column efficiently digests intact proteins into peptides in an online HDX system, typically in about 30 seconds.
Accelerate antibody titer measurements with BioResolve Protein A Affinity Columns featuring MaxPeak Premier Technology. Achieve up to 7× greater sensitivity, faster decision‑making, and improved process control.
These columns also enable advanced workflows, including Direct Connect 2D ProA‑SEC and ProA‑MS methods.
Streamline sample preparation for LC-MS analyses with Waters Laboratory Automation and Equipment that deliver verified workflows to help minimize variability, improve traceability, and simplify method transfer.
Optimize your laboratory's productivity and success with Waters Global Services to maintain peak system performance, minimize downtime, address application challenges, and support stringent compliance requirements.
Maximize resources and minimize risk with payment options from Waters Capital, including upgrading aging equipment, getting customized support, and bundling services into one monthly payment.