HPLC Analysis of Phosphorylated Peptides on the Alliance™ iS Bio System with MaxPeak™ High Performance Surface (HPS) Technology - A System Designed for Bioseparations
Andrew Steere, Norris Wong, Paula Hong
Waters Corporation, United States
Published on March 30, 2026
Abstract
HPLC analysis of peptides is often performed in regulated laboratories to ensure drug quality, verifying Critical Quality Attributes (CQA). However, HPLC peptide mapping analysis can be challenging given the method conditions, which typically require small changes in the gradient over an extended period to provide adequate separation. In addition, one type of peptide, phosphorylated peptides, can exhibit non-specific adsorption (NSA) with metal surfaces via Lewis acid-base interactions. The Alliance iS Bio HPLC System with MaxPeak HPS technology is designed to minimize NSA by applying a barrier layer to mitigate surface to sample interaction, while its quaternary pump delivers accurate and precise gradients under typical HPLC conditions.
In this study, the performance of the Alliance iS Bio HPLC System is compared to both legacy stainless steel and bio-inert systems to assess the suitability of the systems for HPLC peptide mapping applications. To assess the performance, a series of singly and a doubly phosphorylated peptides will be analyzed on the Alliance iS Bio System, focusing on retention time, area precision and peak tailing, system suitability attributes that can be impacted by NSA or pump delivery. To assess the impact of MaxPeak HPS technology and the Alliance iS HPLC gradient delivery on the analysis, the method performance will be compared to both a legacy stainless steel system and an alternative bio-inert system.
Benefits
- The Alliance iS Bio HPLC System with MaxPeak HPS technology reduces NSA of phosphorylated peptides
- The Alliance iS Bio HPLC System is suitable for HPLC peptide mapping applications, delivering precise gradients with quaternary delivery over extended periods
Introduction
HPLC analysis of peptides is often performed in regulated laboratories to monitor CQAs as part of process control strategies to ensure drug quality. However, HPLC peptide mapping analysis can be challenging given the method conditions, which typically require small changes in the gradient over an extended period to provide adequate separation. Additionally, one type of peptide, phosphorylated peptides, can exhibit NSA with metal surfaces via Lewis acid-base interactions. Phosphorylated peptides are produced through post transitional modifications. Under common reversed-phase HPLC conditions, the phosphorylated moiety can result in NSA with metal surfaces, including stainless steel. While these unwanted interactions can occur with any biomolecule that contains an electron-rich functional group, the phosphate group has a strong negative charge and as such molecules containing them tend to exhibit strong NSA.2
NSA can have a significant impact on chromatographic results, including but not limited to peak tailing, poor area precision, and decreased recovery. While there are a variety of mitigation techniques available, they are often time-consuming and may need to be repeated frequently, leading to inconsistent results.
A more modern approach to address NSA is to provide instrumentation that is not prone to this interaction. Bio-inert instrumentation has been developed to specifically address these issues. One example of this is MaxPeak HPS technology, which provides a protective barrier over the metal oxide layer minimizing interactions between the metal surfaces and metal-sensitive compounds such as phosphopeptides. The Alliance iS Bio HPLC System uses this technology, along with bio-compatible materials, in a single system solution, to reduce NSA of sensitive biomolecules, such as phosphopeptides. In addition, the design of the Alliance iS Bio HPLC quaternary pump provides precise gradient delivery under HPLC peptide mapping conditions.
Experimental
Sample Description
Waters MassPREP™ Phosphopeptide Standard Enolase (p/n: 186003285) was reconstituted using 100 µL of Mobile Phase A. Each vial contains 1 nmol each of four yeast enolase derived from phosphorylated peptides. Multiple samples were pooled as necessary to yield sufficient volumes for all runs on all systems then distributed to minimize variability. Reconstituted samples were transferred to QuanRecovery™ MaxPeak HPS polypropylene vials (p/n: 186009186), to reduce nonspecific binding.
LC Conditions
|
LC system: |
Alliance iS Bio HPLC System, Bio-inert HPLC System X, Legacy Stainless-Steel HPLC System |
|
Column: |
XSelect™ Premier Peptide CSH™ C18 Column, 130A, 2.5 µm, 4.6 x 100 mm (p/n: 186009908) |
|
Mobile phase A: |
0.1% trifluoroacetic acid in water |
|
Mobile phase B: |
0.1% trifluoroacetic acid in acetonitrile |
|
Needle wash: |
90/10 water/acetonitrile |
|
Seal wash: |
90/10 water/acetonitrile |
|
Flow rate: |
0.500 mL/min |
|
Injection volume: |
25 µL |
|
Sample temperature: |
5 °C |
|
Column temperature: |
60 °C |
|
Wavelength: |
214 nm |
|
Sampling rate: |
5 Hz |
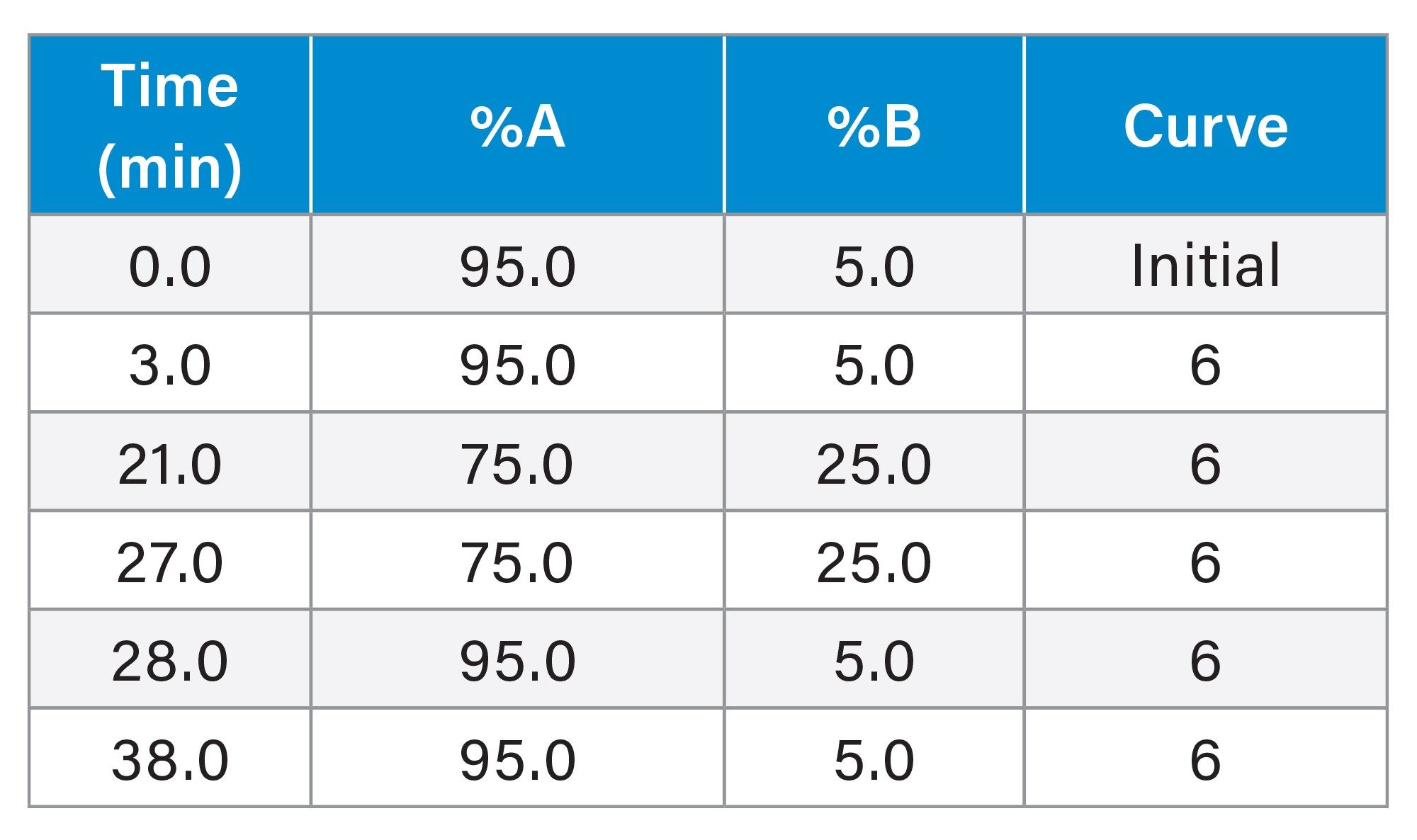
Gradient Table
Data Management
|
Chromatography software: |
Empower™ 3.8.0.1 Chromatography Data Software |
Results and Discussion
To assess the impact of MaxPeak HPS technology and gradient delivery, a set of both singly and doubly phosphorylated peptides were selected, eluting over a range of organic concentrations from approximately 13-18% organic. The standards included four peptides: three singly phosphorylated- t18p, t19p, t43p, and one doubly phosphorylated- t43pp. To assess repeatability, six replicate injections of the phosphopeptide standards were performed.
The samples were analyzed on three HPLC systems including an Alliance iS Bio HPLC System, a second bio-inert HPLC system (Bio-inert HPLC System X), and a legacy stainless-steel HPLC system. All three systems used in this study are quaternary systems, with slight differences in dwell volume and extra-column dispersion. The XSelect Premier Peptide CSH C18 Column was used on all systems in this study. Figure 1 shows the chromatographic results on all three systems.
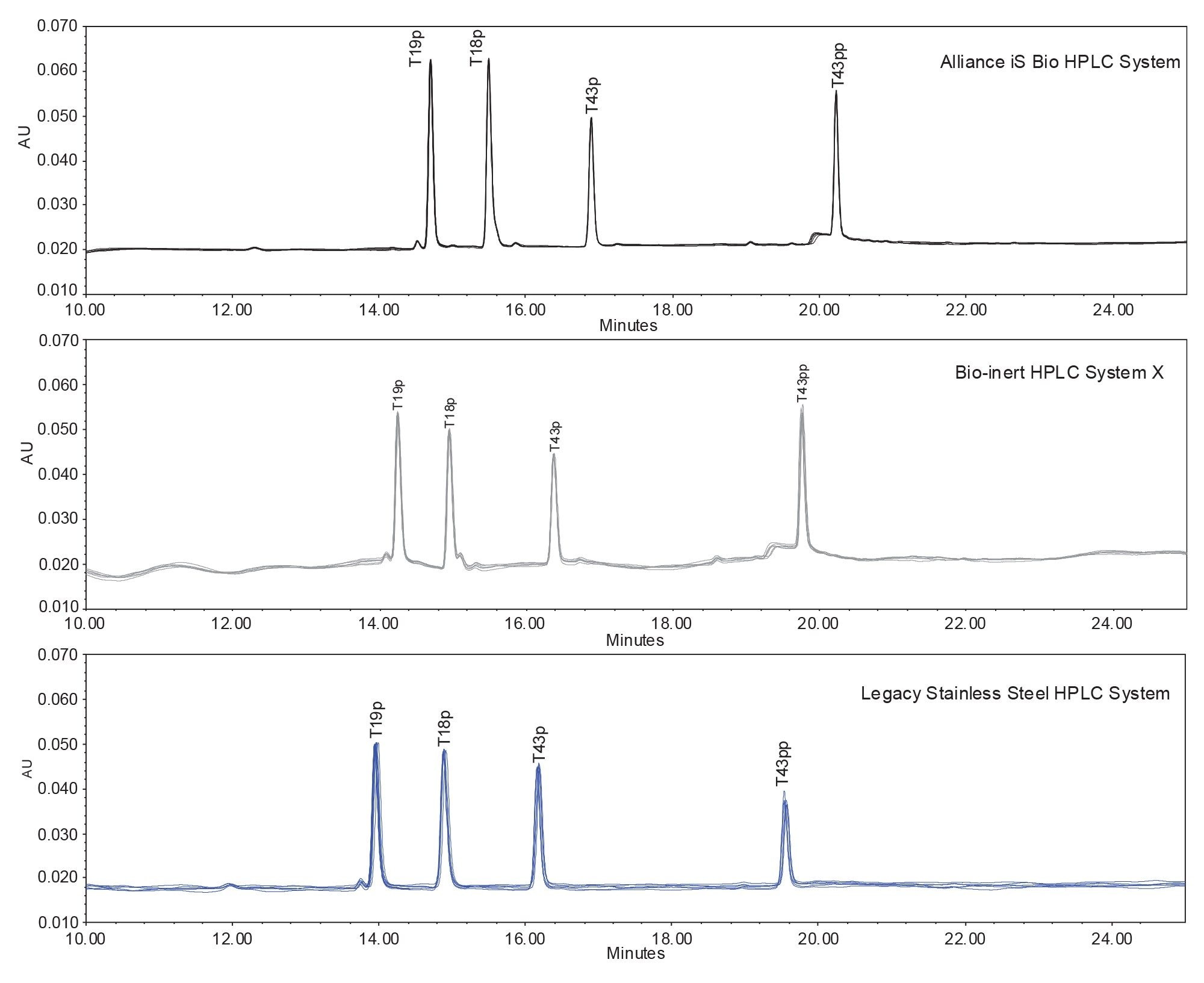
Chromatograms are shown on the same scale for visual comparison purposes. Retention time differences were observed due to differences in dwell volumes among systems. All systems produced acceptable results for the analysis, albeit with some notable differences in terms of performance. For the six repeatable injections, the Alliance iS Bio System produced the lowest noise, with USP noise averaging 3.9 x10-5 µAU. In contrast, the average USP noise on Bio-Inert HPLC System X was 1.8 x10-4 µAU, and the stainless steel system average was 1.7 x10-4 µAU. All noise values were obtained between the third and fourth quantitated peaks. The increased noise observed in the latter systems may in part be explained by configuration differences. The lack of an in-line mixer of the Bio-inert system may have contributed to the visible baseline ripple whereas a larger pump stroke volume may have played a role in the legacy stainless-steel HPLC system’s variable baseline. While there may be a variety of system contributors that impact these results, the Alliance iS Bio HPLC pump has the lowest stroke volume of the three systems tested as well as an in-line mixer, both of which can impact baseline ripple due to TFA gradients at lower wavelengths and thus may be a contributor to the improved performance of the Alliance iS Bio HPLC System.4
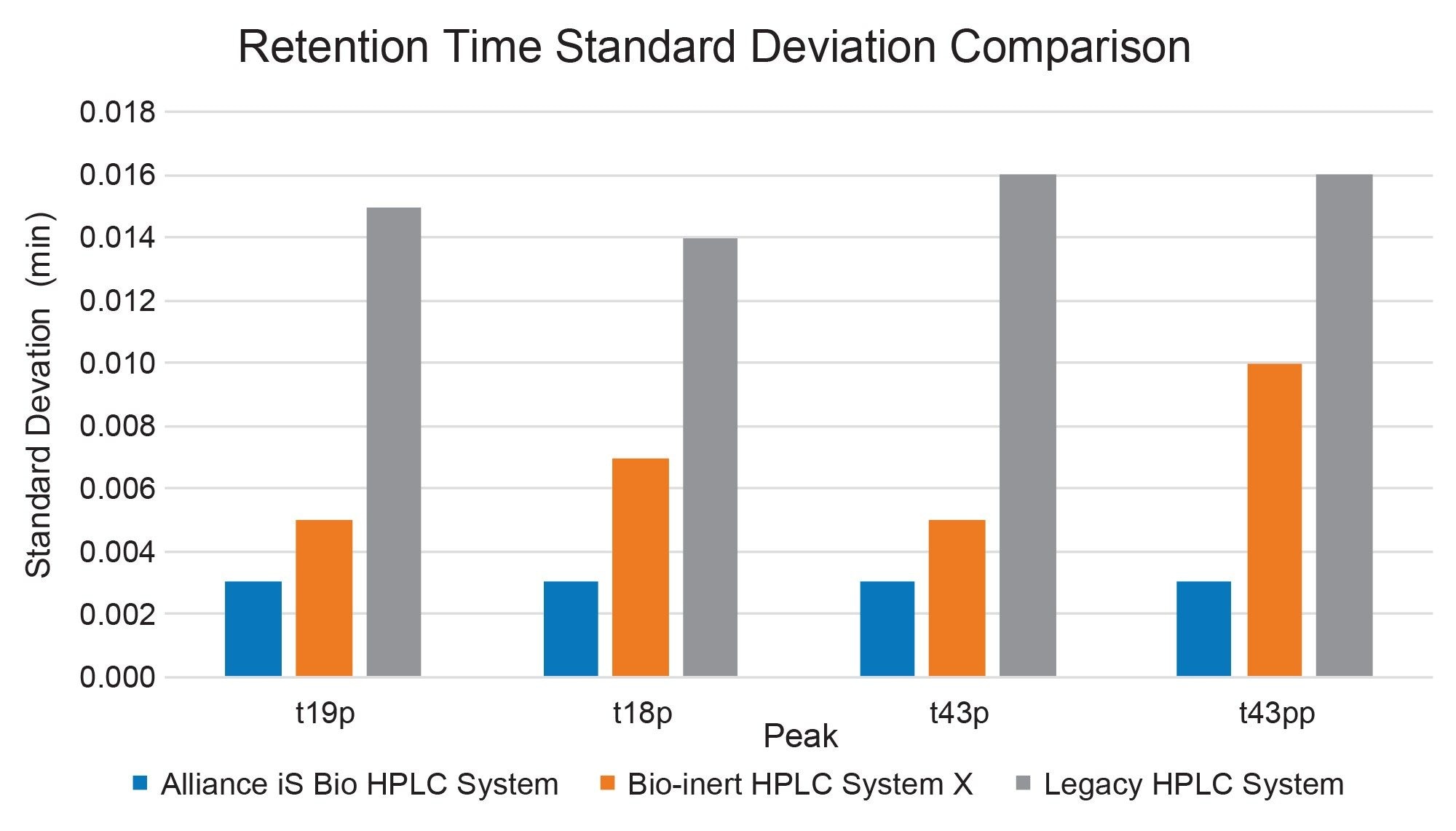
To assess the precision of the pump over the extended gradient, the retention time precision was evaluated. Given the longer elution times, retention time precision was measured by retention time standard deviation (Figure 2). All systems produced standard deviations of less than 0.02 minutes, well within expected composition precision of the systems.5 The highest level of repeatability was observed on the Alliance iS Bio HPLC System with a maximum retention time standard deviation (RT SD) of 0.003 minutes. Slightly higher values were observed on the Bio-inert System X and the legacy stainless-steel system, with maximum values of 0.010 and 0.016 minutes standard deviation. While all results are acceptable and would not negatively impact peak confirmation, the RT SD also remained most consistent across the four peaks on the Alliance iS Bio HPLC System.
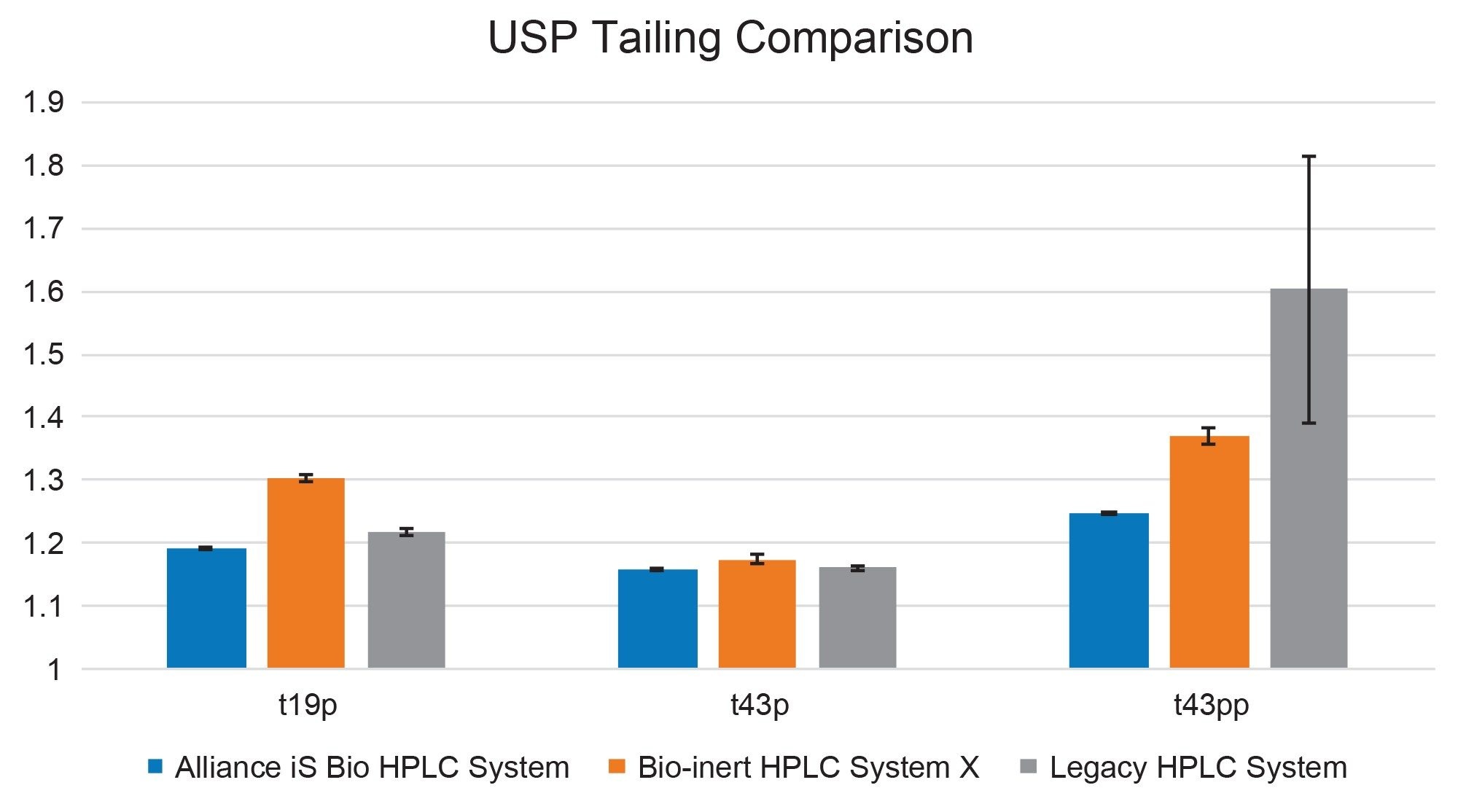
To assess the impact of system performance, USP tailing results are shown in Figure 3. General system characteristics such as poor connections would impact all peaks equally, while NSA vary by peptide moieties, with the doubly phosphorylated peptide being most impacted. The results indicate differences most attributable to NSA. Similar performance was observed for both the t19p and t43p singly phosphorylated peptides on all systems. When comparing the doubly phosphorylated t43pp peptide, the analyte most likely to exhibit NSA, there is a significant performance difference between the stainless-steel and bio-inert systems. The standard deviation of the USP tailing for the six injections was also significantly larger for the t43pp peptide on the stainless-steel system as shown by the error bars on each result. These results indicate that NSA is mitigated by the use of bio-inert surfaces, including MaxPeak HPS technology.
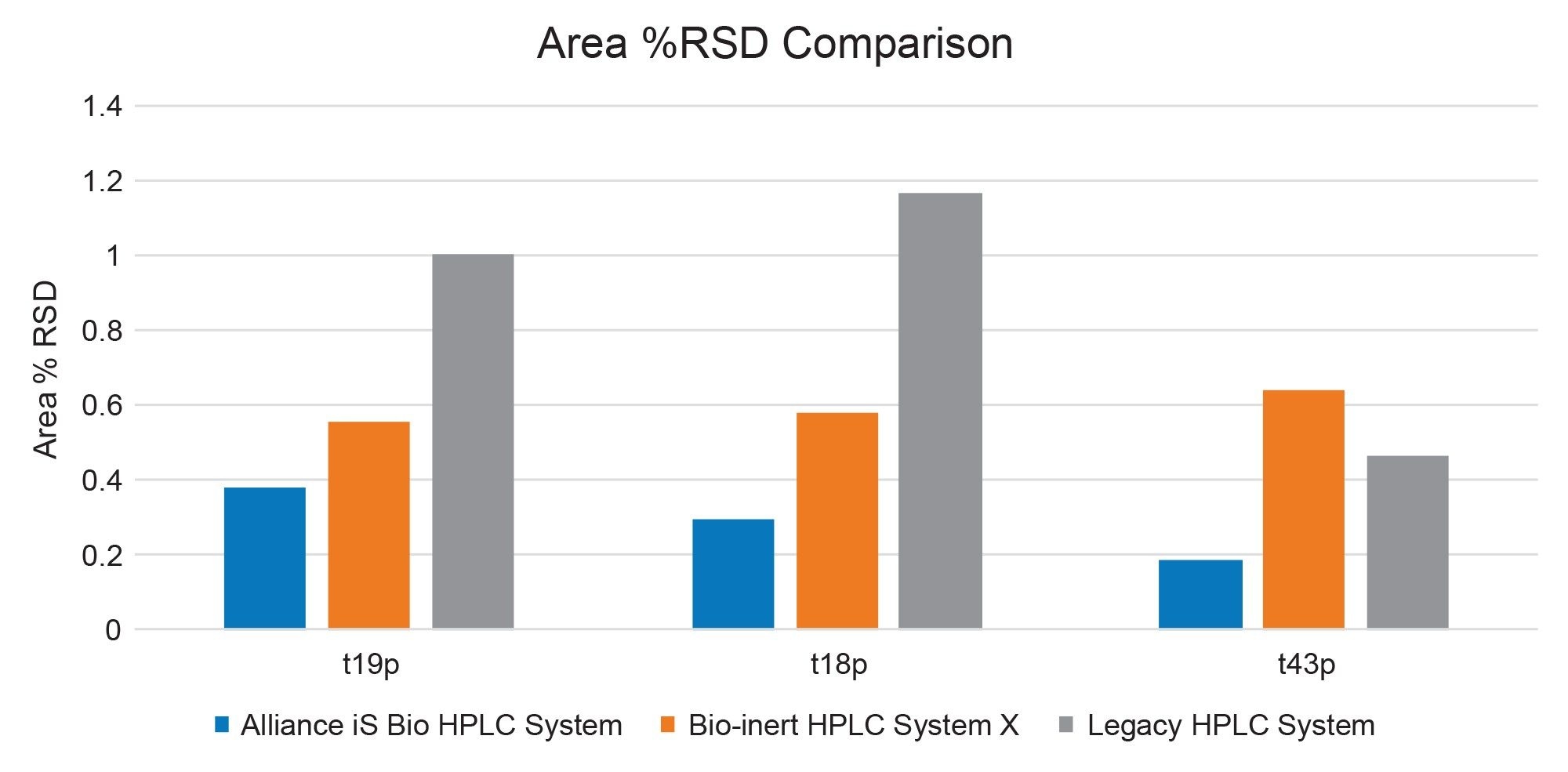
Another chromatographic attribute, area precision, can be impacted by a variety of factors, including noise, NSA, and injection precision. The area % RSD results for the three singly phosphorylated peaks (Figure 4) show high precision on all three systems for selected peaks, with the highest RSD being under 1.2%. The Alliance iS Bio HPLC System showed the highest precision with values of less than 0.4% RSD for all three singly phosphorylated peptides. The legacy stainless-steel system produced larger variability, suggesting that NSA may impact the area precision of the peptides.
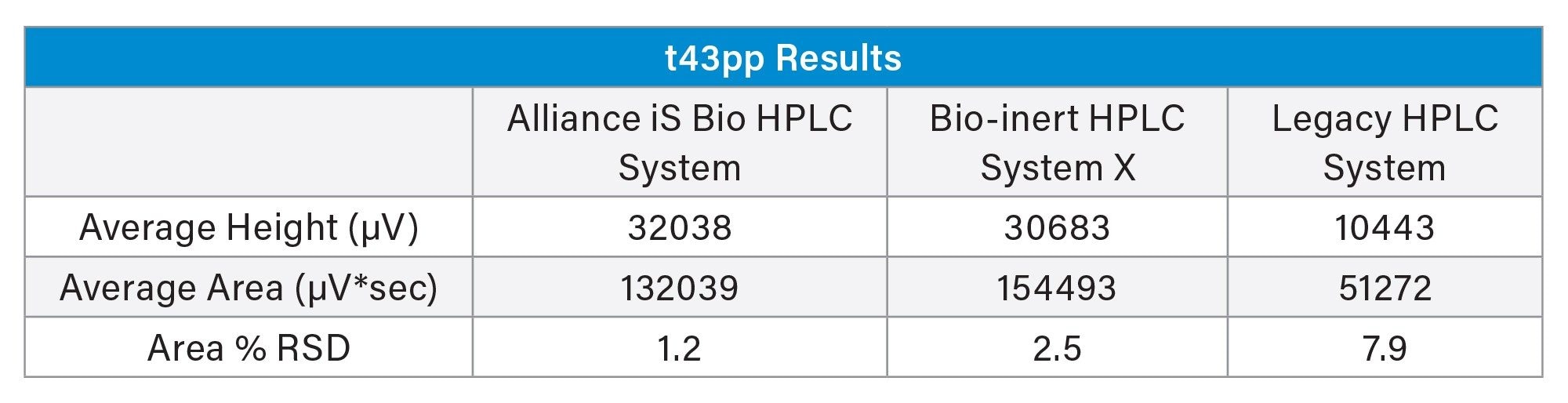
For the fourth peak, the doubly phosphorylated t43pp peak, higher variability was observed across all three systems (Table 1). Based on chemical structure, the doubly phosphorylated peptide is more prone to NSA. This is supported by the relative peak height of the peptide on the stainless-steel system (Figure 1), where the t43pp peak height is significantly reduced comparatively. Evaluation of area precision also indicated NSA was observed for t43pp (Figure 4). The two bio systems have similar height and area results, and both yield an area % RSD under 2.5%. The legacy stainless-steel system produced raw peak height and area results of about one-third of the bio-inert systems, with significantly higher area precision (% RSD of 7.9%).
Conclusion
HPLC analysis of phosphorylated peptides on the Alliance iS Bio HPLC System demonstrated high level of performance. For this challenging method with both a long shallow gradient and analytes prone to NSA, the Alliance iS Bio HPLC System met all method criteria. In addition, chromatographic analysis demonstrated improved repeatability as compared to legacy stainless-steel system. The Alliance iS Bio HPLC quaternary pump delivered highly repeatable retention times for all peaks, compared to another modern bio-inert solution and a legacy stainless-steel system. Due to key factors such as a robust flow path and MaxPeak HPS technology, the system yields the best performance for biomolecules prone to NSA. Continued improvements of instrumentation plays a key role in advancement of biomolecule analysis, and the mitigation of NSA demonstrated in this study shows one example of how this is possible.
References
- Walter TH, Trudeau M, Simeone J, Rainville P, Patel AV, Lauber M, Kellett J, DeLano M, Brennan K, Boissel C, Birdsall RE, Berthelette K. Low Adsorption UPLC Systems and Columns Based on MaxPeak High Performance Surfaces: The Acquity Premier Solution. Waters White Paper. 720007128. 2021.
- Koshel BM, Simeone J, Dao D, Nguyen JM, Rzewuski SC, Lauber M, Birdsall RE, Yu YQ. Bypassing LC System Passivation Requirements Using Acquity Premier with MaxPeak HPS Technology for the Recovery of a Phosphorylated Peptide. Waters Application Note. 720006921. 2020.
- Hughes C, Gethings LA, Plumb RS. Maximizing Phosphopeptide Recovery in LC-MS Studies with MaxPeak High Performance Surfaces Technology. Waters Application Note. 720007025. 2020.
- Martin K, Gauthier L, Hong P. Improved Sensitivity for Trifluoracetic Acid Gradients on the Alliance iS HPLC Systems. Waters Application Note. 720008550. 2024.
- Alliance iS HPLC System Specification Sheet. 720007867. 2025.
Featured Products
720009289, March 2026