Many are familiar with electron ionization (EI). (Sometimes the earlier phrase "electron impact" is used-although, technically, it is incorrect). EI, often performed by exposing a sample to 70 eV electrons, is referred to as a "hard" technique. The energy of the electrons interacting with the molecule of interest is generally much greater than that contained in its bonds, so ionization occurs. The excess energy breaks bonds in a well-characterized way. The result is predictable, identifiable fragments from which we can deduce the molecule's identity. Abstraction of only an electron from the outer shell yields a radical cation in the positive mode (M.+) and a rich spectrum of fragments. Unlike "softer" atmospheric ionization techniques which produce a spectral response sometimes characteristic of the manufacturer's particular source design the EI technique is fairly independent of the source design. A spectrum produced by one EI instrument looks much like a spectrum of the same compound from another EI instrument, a fact that lends itself to creating spectral libraries to match unknowns to reference spectra.
Molecules that fragment excessively call for "soft" techniques. Chemical ionization (CI) produces ions by a gentler proton transfer process that preserves and promotes the appearance of the molecular ion itself. The sample is exposed to an excess of reagent gas such as that which evolves when methane forms the protonated molecular ion (M+H). The reverse process can produce negative ions. Transferring the proton to the gas molecule can, in some cases, produce the negative ion (M-H).
Chemical Ionization (CI) is sometimes used for compounds with chemistry similar to those analyzed by EI to enhance the abundance or appearance of the molecular ion in favor of significant fragmentation. Similar to EI, samples must be thermally stable since heating in the source causes vaporization. The ionization mechanism of CI relies on EI for the initial ionization step but within the source is a chemical reagent gas, such as methane, isobutane or ammonia, at high pressure. The reagent gas, which is present at a much higher concentration than the analyte (R), is ionized by electron ionization to give primary R+. reagent ions. The collision of the R+. ions with neutral R molecules lead to the formation of stable secondary ions which are the reactant species which then ionize analyte molecules (A) by ion-molecule reactions.
For example the ion-molecule reaction between a methane ion and a methane molecule gives rise to the fairly stable CH5+ species.
CH4+. + CH4 --> CH5+ + CH3.
The reactant ion CH5+ can ionize neutral analyte molecules (A) by proton transfer, hydride abstraction or charge exchange.
RH+ + A --> R + AH+ (proton transfer)
(R-H)+ + A --> R + (A-H)+ (hydride abstraction)
R+. + A --> R + A+. (charge exchange)
The most common ionization reactions are protonation, which is favored for molecules with proton affinities higher than the reagent. Hydride abstraction is common for lower proton affinity molecules and charge exchange occurs with reagents of high ionization energy.
The substance to be analyzed is at a much lower pressure than the reagent gas. If we consider methane as the reagent gas the electron impact causes mainly ionization of the methane. This fragments in part to CH3+. These species then undergo ion molecule reactions under the high source pressures employed.
CH4+. + CH4 --> CH5+ + CH3.
CH3+ + CH4 --> C2H5+ + H2
CH5+ can act as a Bronsted acid and C2H5+ as a Lewis acid to produce ions from the analyte.
Careful choice of the CI reagent gas can improve charge transfer to an analyte molecule as the gas phase acidity of the chemical ionization gas influences the efficiency of the charge transfer. In CI the analyte is more likely to result in a molecular ion with the reduced fragmentation conserving the energy normally internalized in EI to break bonds.
A variation, negative chemical ionization (NCI), can be performed with an analyte that contains electron-capturing moieties (e.g., fluorine atoms or nitrobenzyl groups). Sensitivity can be increased many-fold (reported to be 100 to 1000 times greater in some case) than that of EI. NCI is applicable to a wide variety of small molecules that are (or can be) chemically modified to promote electron capture.
In negative ion there are two primary mechanisms whereby negative ions are produced: electron capture and reactant ion chemical ionization. Under CI conditions electronegative molecules can capture thermal electrons to generate negative ions. True negative ion chemical ionization occurs by reaction of an analyte compound (AH) with negatively charged reactant ions (R-. or R-). Several types of ion-molecule reactions can occur, the most common being proton abstraction.
AH + R- --> A- + RH
As the proton affinity (basicity) of the reactant ion increases the more likely proton abstraction is to occur.
Perhaps the first encounter with a mass spectrometer for many is as the detector for a gas chromatograph. The range of GC-MS instrument types has expanded to transcend the limitations of earlier instrument designs or to meet increasingly stringent legislation in applications like environmental analysis, food safety screening, metabolomics, and clinical applications like forensics, toxicology, and drug screening.
In the past, two types of mass spectrometers dominated GC-MS analysis: magnetic sector and the single quadrupole instruments. The former, which offered high resolution and accurate-mass analyses, was used in applications that required extreme sensitivity. The latter performed routine analysis of target compounds.
The most challenging GC-MS analyses were reserved for magnetic sector instruments: dioxins in environmental/industrial samples or screening for the illegal use of performance-enhancing drugs in competitive sports. Femtogram detection levels, at high resolution/selectivity, are easily achieved on magnetic sector instruments.
Shortly after their introduction, quadrupole GC-MS systems gained acceptance in target analysis applications. USEPA methods dictated the use of quadrupole GC-MS instruments to analyze samples for numerous environmental contaminants. Because those applications require only picogram-to-nanogram levels of detection, the poorer sensitivity of the quad relative to the sector was not a limitation. On the contrary, the greatly reduced cost, ease-of-use and portability proved a godsend.
The revolutionary technology that gave us analytical access to about 80% of the chemical universe unreachable by GC is also responsible for the phenomenal growth and interest in mass spectrometry in recent decades. A few individuals are singled out (see the section on ‘A Brief History') for coupling LC with MS. Beginning arguably in the 1970s LCMS as we know it today reached maturation in the early 1990s. Many of the devices and techniques we use today in practice are drawn directly from that time.
Liquid chromatography was defined in the early 1900's by the work of the Russian botanist, Mikhail S. Tswett. His studies focused on separating leaf pigments extracted from plants using a solvent in a column packed with particles. In its simplest form liquid chromatography relies on the ability to predict and reproduce with great precision competing interactions between analytes in solution (the mobile or condensed phase) being passed over a bed of packed particles (the stationary phase). Development of columns packed with a variety of functional moieties in recent years and the solvent delivery systems able to precisely deliver the mobile phase has enabled LC to become the analytical backbone for many industries.
The acronym HPLC was coined by Csaba Horváth in 1970 to indicate that high pressure was used to generate the flow required for liquid chromatography in packed columns. Continued advances in performance since then including development of smaller particles and greater selectivity also saw the meaning of the acronym change to high-performance liquid chromatography.
In 2004, further advances in instrumentation and column technology achieved significant increases in resolution, speed, and sensitivity in liquid chromatography. Columns with smaller particles [1.7 micron] and instrumentation with specialized capabilities designed to deliver mobile phase at 15,000 psi [1,000 bar] came to be known as UPLC technology representing the differentiated term ultra-performance liquid chromatography. Much of what is embodied in this current technology was predicted by investigators such as John Knox in the 1970s. Knox predicted the optimum particle diameters would be 1–2μm and chromatography would be thermally sensitive to frictional heat. Technology capable of developing robust, uniform small particles was necessarily encountered and resolved on the path to developing UPLC for widespread use. View a good basic primer on HPLC and UPLC.
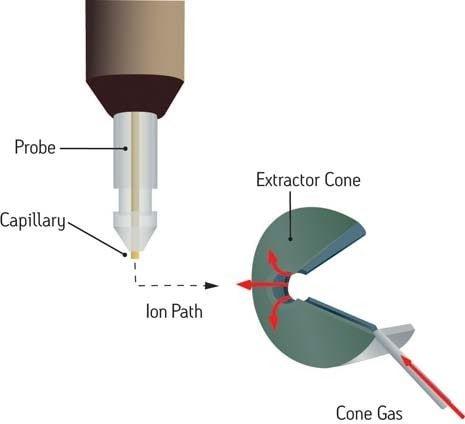
The general term "atmospheric pressure ionization" (API) includes the most notable technique, electrospray ionization (ESI), which itself provides the basis for various, related techniques capable of creating ions at atmospheric pressure rather than in a vacuum. The sample is dissolved in a polar solvent (typically less volatile than that used with GC) and pumped through a stainless steel capillary which carries between 2000 and 4000 V. The liquid aerosolizes as it exits the capillary at atmospheric pressure, the desolvating droplets shedding ions that flow into the mass spectrometer, induced by the combined effects of electrostatic attraction and vacuum.
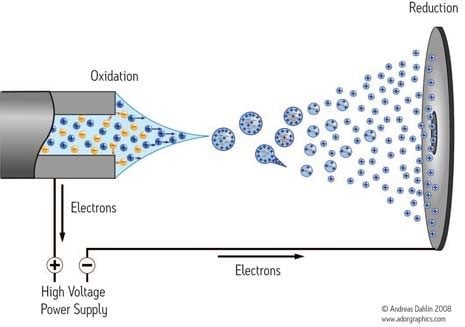
The mechanism by which potential transfers from the liquid to the analyte, creating ions, remains a topic of controversy. In 1968, Malcolm Dole first proposed the charge residue mechanism in which he hypothesized that as a droplet evaporates, its charge remains unchanged. The droplet's surface tension, ultimately unable to oppose the repulsive forces from the imposed charge, explodes into many smaller droplets. These Coulombic fissions occur until droplets containing a single analyte ion remain. When the solvent evaporates from the last droplet, a gas-phase ion forms.
In 1976, Iribarne and Thomson proposed a different model, the ion evaporation mechanism, in which small droplets form by Coulombic fission, similar to the way they form in Dole's model. However, according to ion evaporation theory, the electric field strength at the surface of the droplet is high enough to make leaving the droplet surface and transferring directly into the gas phase energetically favorable for solvated ions.
It is possible that the two mechanisms may actually work in concert: the charge residue mechanism dominant for masses higher than 3000 Da while ion evaporation dominant for lower masses (see R Cole, Some tenets pertaining to Electrospray ionization mass spectrometry, J of Mass Spec, 35, 763–772 (2000).
The liquid from the liquid chromatograph enters the ESI probe in a state of charge balance. So when the solvent leaves the ESI probe it carries a net ionic charge. To ensure that ESI is a continuous technique, the solution must be charged by electrochemical reactions whereby electrons transfer to a conductive surface acting as an electrode. Among other effects, this process can lead to pH changes. It is assumed that, in positive mode, positive-charged droplets leave the spray, and electrons are accepted by the electrode (oxidation). (The reverse would be true in negative mode.) The surface area of the electro-active electrode, the magnitude of the current, and the nature of the chemical species and their electrode potentials all exert an effect.
Over all, ESI is an efficient process. However, the activation energy and energy difference for the reaction, in total, for individual species varies. The flow rate of the solution and the applied current define limits for each droplet. Competition between molecules occurs, and suppression of analytes of interest is not uncommon.
Extensions of basic ESI theory, such as reducing the liquid to extremely low volumes - for example to 30 nL/min in the case of nanospray-have proved effective, especially in sample-limited studies of proteins and amino acids.
Although work demonstrating APCI was published in parallel with that demonstrating ESI, APCI was not widely adopted until ESI was commercialized, which occurred in the wake of Fenn's work in 1985.
Horning first introduced APCI in 1973 to analyze volatile compounds using various introduction techniques, one of which was HPLC. The adjunctive capability of APCI permits analytes that resist conversion to gas-phase ions by ESI, the less polar and more volatile ones introduced into a mass spectrometer from a condensed phase, or liquid, stream. Unlike ESI, APCI transfers neutral analytes into the gas phase by vaporizing the introduced liquid in a heated gas stream. Chemical ionization relies on the transfer of charged species between a reagent ion and a target molecule to produce a target ion that can be mass analyzed. Most commonly, in positive ion mode, an adduct forms between the target molecule and the small H+ ion, although adducts with salts are common as well. For example, the ammonium adduct can form (M+NH4)+ when the weak-acid-weak-base salt, ammonium acetate, is present in the mobile phase, a modifier often used in place of the less volatile and highly ionic phosphate buffer. At higher salt concentrations, competition between the protonated and ammoniated forms can produce a decreased response for both. The maximum number of ions capable of forming by APCI is much greater than it is in ESI because reagent ions form redundantly. The liquid is pushed through a nonconductive tube, usually of fused-silica glass, around which a nebulizing gas flows. The resultant fine droplets collide with the inner, heated wall of a tube or probe that extends beyond the end of the nonconductive tube, and are thus converted to the gas phase. This ionization type is often performed at much greater linear velocities than those of the HPLC or ultra performance liquid chromatography (UPLC) flow rates normally associated with electrospray. Contemporary instruments however provide much greater desolvation capacities enhancing performance for all aerosol dependent techniques.
The desolvated analyte molecules are then ionized via chemical ionization. The ionizing potential is applied, not through the liquid as in ESI, but at the tip of a needle as a plasma, or corona, through which the droplets pass. In effect, the mobile phase acts as an intermediary transferring the charge to the analyte. Hence the early name given APCI: "solvent-mediated electrospray."