
This application note will highlight a process for selectively fractionating monoterpenes in oregano from the bulk extract using Waters Bio-Botanical Extraction System (BBES). Monoterpene relative purity and terpene ratios are compared for the BBES terpene fraction and common distillation techniques, using Waters ACQUITY UPLC H-Class System.
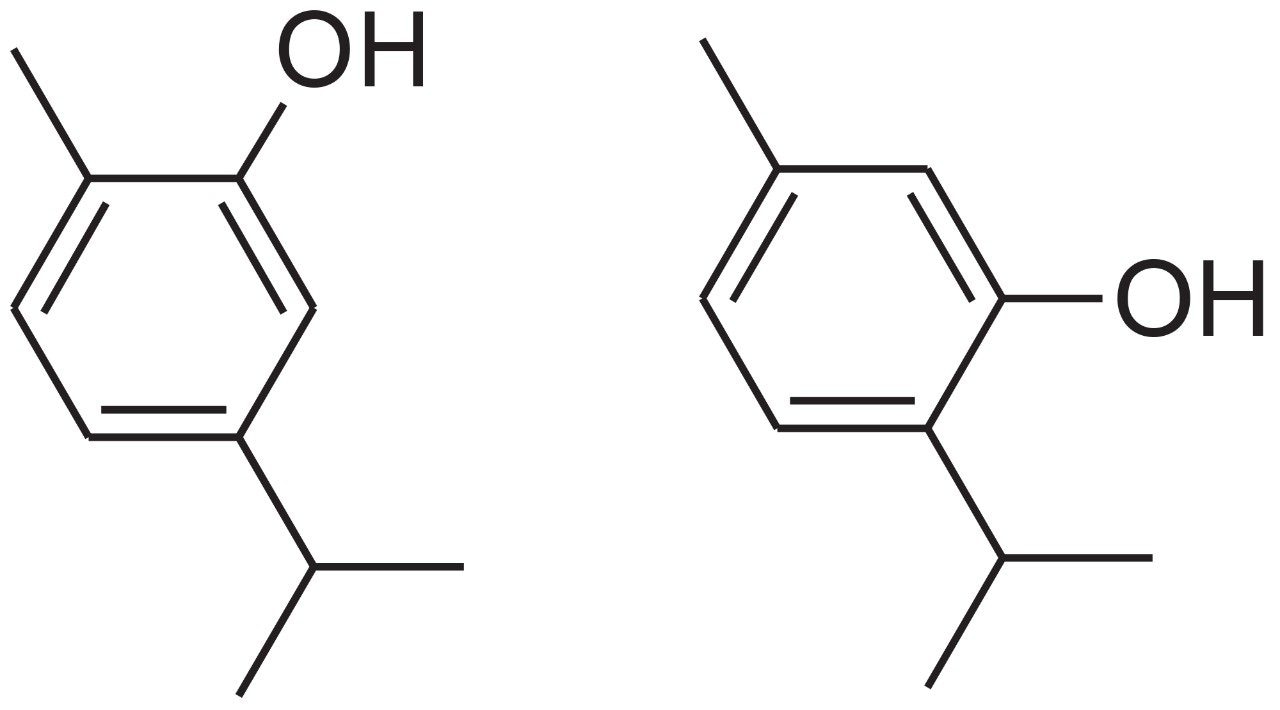
Oregano (origanum vulgare) is an herb typically used as flavoring in food preparations. While research is still ongoing with regard to use of raw dried oregano as a natural therapeutic, tinctures and essential oils from oregano leaves have been used as an antioxidant and as a holistic treatment to sooth a number of ailments such as asthma, coughing, indigestion, and arthritis.1 Carvacrol (Figure 1), the major monoterpene found in oregano, along with extracts of oregano containing carvacrol, has also been shown to be an effective anti-parasitic in humans and animals.2 Monoterpenes like carvacrol are light volatile oils ubiquitous in many natural products, and their role in the overall therapeutic properties of natural products is becoming increasingly important.3 Therefore, due to their potential therapeutic uses and unique aroma and flavor, techniques for isolating and concentrating terpenes from natural products are desired across a wide range of applications.1,2,3,6
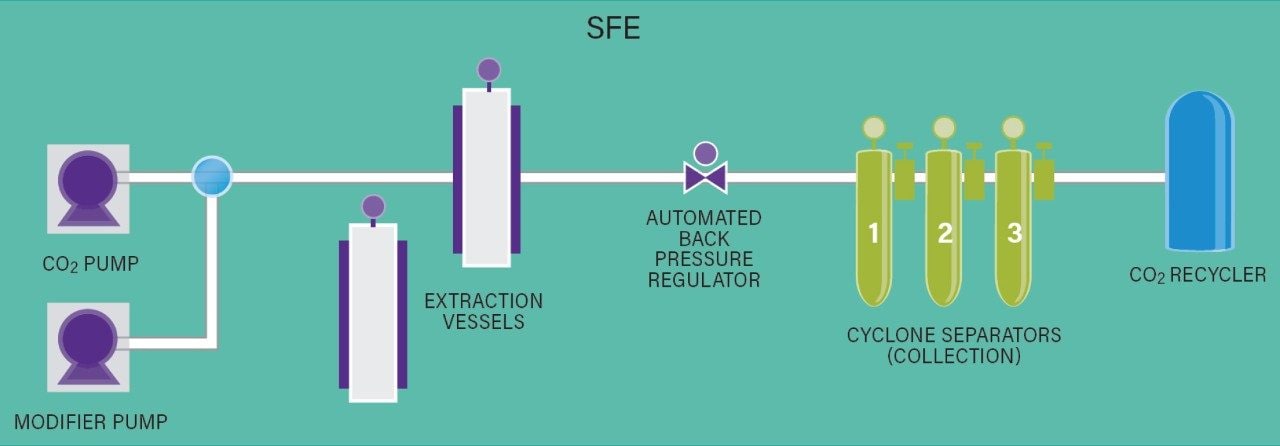
Supercritical Fluid Extraction (SFE) utilizes carbon dioxide (CO2) as the primary component of the extracting solvent. The unique properties of CO2 allow solvent strength to be altered during an extraction process by changing temperature and pressure.4 In the gas phase, the CO2 solvent has no solvating power. As pressure is increased the CO2 density increases and the solvent is capable of dissolving and extracting non-polar to moderately polar analytes, and the addition of polar modifiers can further extend the solvent polarity range.4 Supercritical Fluid Fractionation (SFF) is an efficient technique for generating chemically different extract fractions from a single supercritical fluid extraction, and it is accomplished by gradually decreasing pressure following extraction. By controlling temperature and pressure during extract collection, the consistency and contents of extract fractions can be optimized for a specific targeted outcome.1,4,5,6 This application note will highlight a process for selectively fractionating monoterpenes in oregano from the bulk extract using Waters Bio-Botanical Extraction System (BBES). Monoterpene relative purity and terpene ratios are compared for the BBES terpene fraction and common distillation techniques, using Waters ACQUITY UPLC H-Class System.
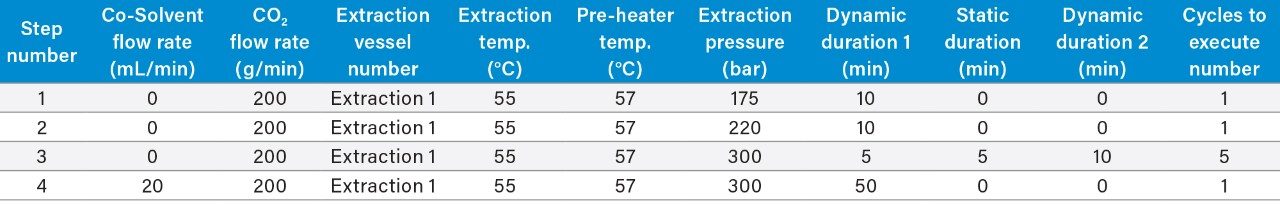
A preparative scale supercritical fluid extraction was performed using Waters Bio-Botanical Extraction System (Figure 2) equipped with a 5L extraction vessel basket and P-50 co-solvent pump. Detailed extraction and fractionation conditions can be found in Table 1 and Table 2, respectively. In summary, the pressure was increased using three step gradients of 150 bar, 220 bar, and 300 bar over the course of 20 minutes. At 300 bar, five static-dynamic cycles were performed with 5 minute static and 10 minute dynamic steps. Three CO2 only fractions were generated from cyclonic separators placed in series. One fraction was collected from high pressure cyclone separator 1 (CS1), one from cyclone separator 2 (CS2) and one from low pressure cyclone separator 3 (CS3). The CO2 extract fractions were drained at the end of step 3 in Table 1. Following the static dynamic cycles, 10% isopropanol in CO2 was used to generate a co-solvent extract fraction, step 4 in Table 1. One co-solvent fraction was generated by combining the co-solvent containing extract from all 3 cyclone separators (data not shown). A total of 1 L of isopropanol was used. Using the SFE method described, the extract mass yield was 3.1% (20 g). The SFE fractions were then compared to an essential oil produced via a method of laboratory scale hydrodistillation7 of the same oregano feedstock.
Semi-quantitative analyses were performed using the ACQUITY UPLC H-Class System, in order to compare the relative purity and ratios of monoterpenes in various extract fractions. Extracts were diluted in ethanol, filtered using Acrodisc 1 μm glass fiber syringe filters (Waters pn: WAT200819) and analyzed directly. Analysis of the dried oregano leaves was made prior to extraction with the BBES, and the raffinate was analyzed post-extraction (data not shown). Approximately 0.5 g of oregano was placed in 15 mL homogenizing vial containing a mixture of 2.8 mm and 5.0 mm ceramic grinding medium. Six milliliters of isopropanol were added and the samples homogenized for 1 minute using Precellys Evolution laboratory homogenizer (Bertin Corporation). The resulting liquid extract was filtered and analyzed. Relative UV purity for the monoterpenes thymol and carvacrol was calculated from the UV chromatogram at 275 nm, similar to the qualitative analysis of oregano extracts used elsewhere.5 Note: UV purity was used here in order to quickly compare the quality of an array of extracts. This type of comparative analysis is qualitative andis not meant to replace a true quantitative assay. UV purity will give an overestimate of the actual purity determined gravimetrically against standards.
Dried oregano leaves were purchased online from StarWest Botanicals pn: 209885-31) and commercial oregano essential oil was purchased online from The Vitamin Shoppe (pn: IF-0017). Thymol and Carvacrol standards were purchased from Sigma-Aldrich.

|
LC system: |
ACQUITY UPLC H-Class |
|
Detection: |
PDA, 275 nm, Resolution 4.8 nm, 450 nm reference wavelength |
|
Column: |
Waters CSH C18 1.7 μm × 2.1 mm I.D. × 50 mm |
|
Column temp.: |
50 °C |
|
Sample temp.: |
Ambient |
|
Injection volume: |
1 μL |
|
Flow rate: |
0.4 mL/min |
|
Mobile phase A: |
Water 0.1% formic acid |
|
Mobile phase B: |
Acetonitrile 0.1% formic acid |
|
Gradient: |
Hold 5% B for 0.5 min, 5%–95% B linear gradient from 0.5 min to 8.0 min, 10 min re-equilibration |
Empower 3 Chromatography Data Software
ChromScope v2.0 Software
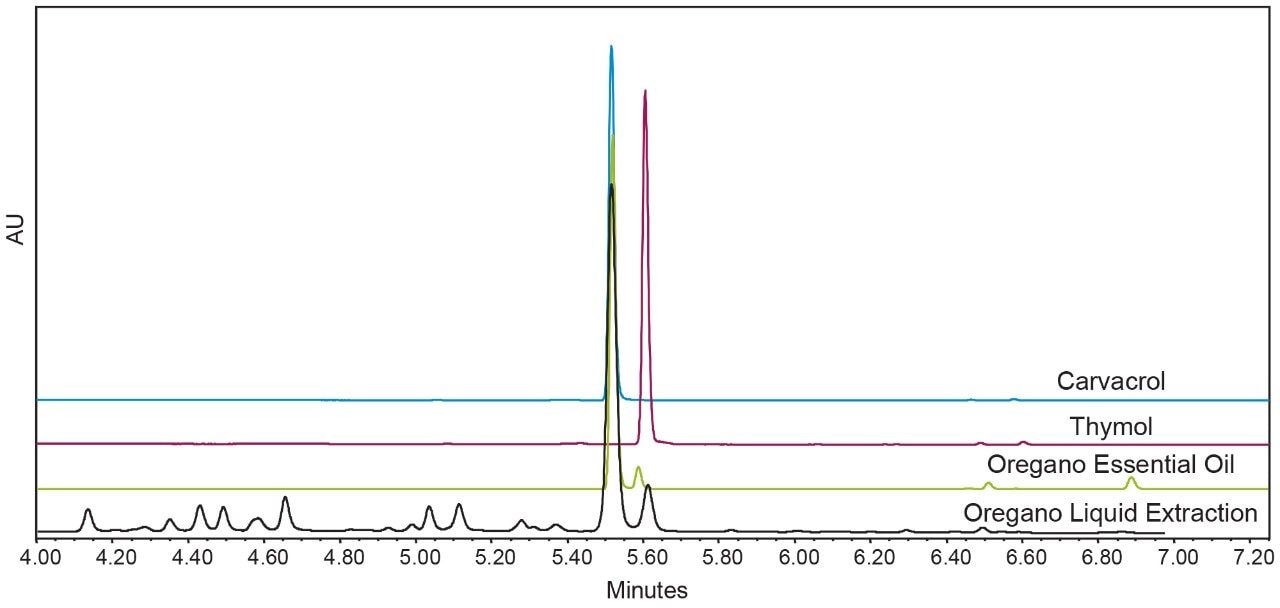
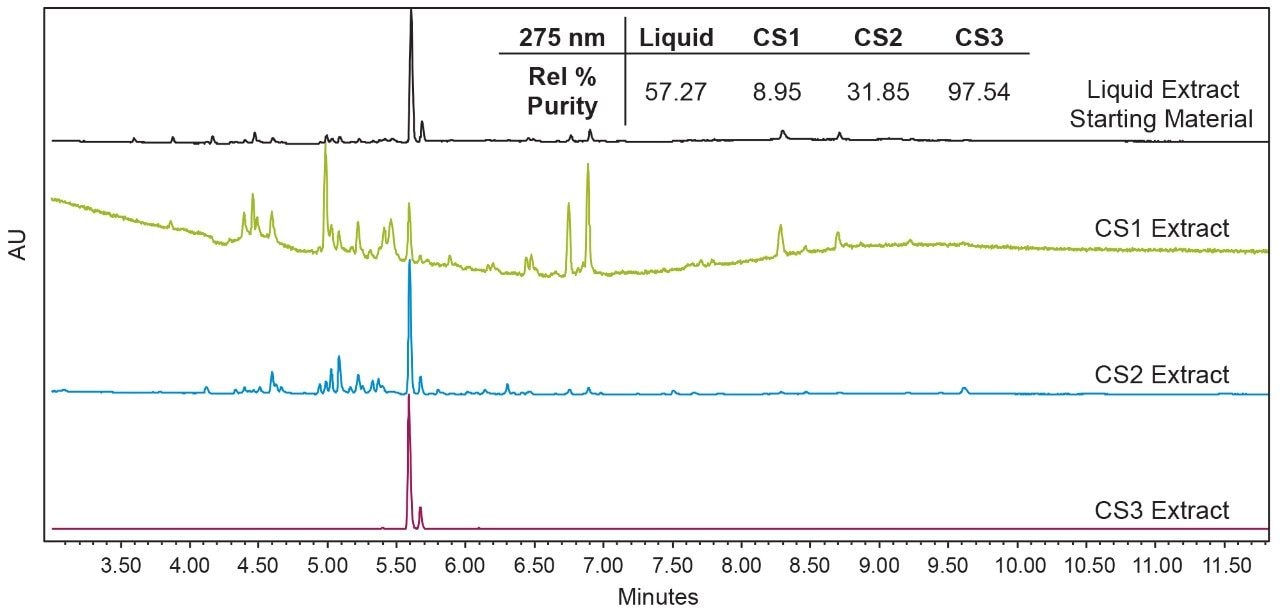
Initial work was done to develop an analytical chromatographic method and analytical scale liquid extraction for dried oregano, in order to characterize the starting material and compare subsequent extract fractions. Figure 3 shows an overlay of the oregano liquid extract with standards for the major monoterpenes in oregano, carvacrol and thymol, and a commercially produced essential oil of oregano for comparison. In the liquid extraction of the dried oregano feedstock used for SFE and hydrodistillation, the main peak of interest, carvacrol, is the most prominent component, followed by the structural isomer thymol. Other minor chromatographic peaks were not characterized, and are considered excipient material for the purposes of this study. However, many of the minor constituents found in oregano extract have been studied elsewhere.1,2,5 The ability to selectively enrich compound classes using SFE with fractionation is shown in Figure 4. The three CO2 extracts are each compared to the liquid extract, and the relative UV purity at 275 nm for the monoterpenes carvacrol and thymol in each extract is shown in the inset table. Using the SFE method summarized in Table 1 and Table 2, three distinct, chemically different extracts were generated. The CS3 fraction is the primary terpene fraction, enriched with terpenes and free of other excipient material. The CS2 fraction contains most of the excipient material and a portion of the terpenes, and the CS1 fraction is almost completely free of terpenes.
There are multiple processes available for producing terpene rich fractions from natural products;7 the most common commercial processes being hydrodistillation and steam distillation. Using distillation it is possible to produce high quality, concentrated essential oils however, this process is energy intensive6 and exposes the natural product to temperatures in excess of 100 °C for extended periods of time which can degrade other compounds of interest present in the plant.1,5 Figure 5 compares the SFE terpene fraction to a hydrodistillation of the same dried oregano used in this study and a commercially available essential oil of oregano produced via steam distillation. Each process was able to effectively separate carvacrol and thymol from excipient plant material, with nearly identical relative UV purities of 92.5%, 97.0%, and 97.5% for commercial steam distillation, hydrodistillation, and SFE respectively. The major difference between the essential oils produced are distillation processes used high temperatures, and generated a single terpene-rich fraction; an additional extraction would need to be performed to extract additional non-volatile plant components. SFE exposed the natural product to a max temperature of 55 °C, and generated a high quality fraction rich in terpenes and additional fractions rich in excipient material, which are of interest in whole-plant value-added applications.
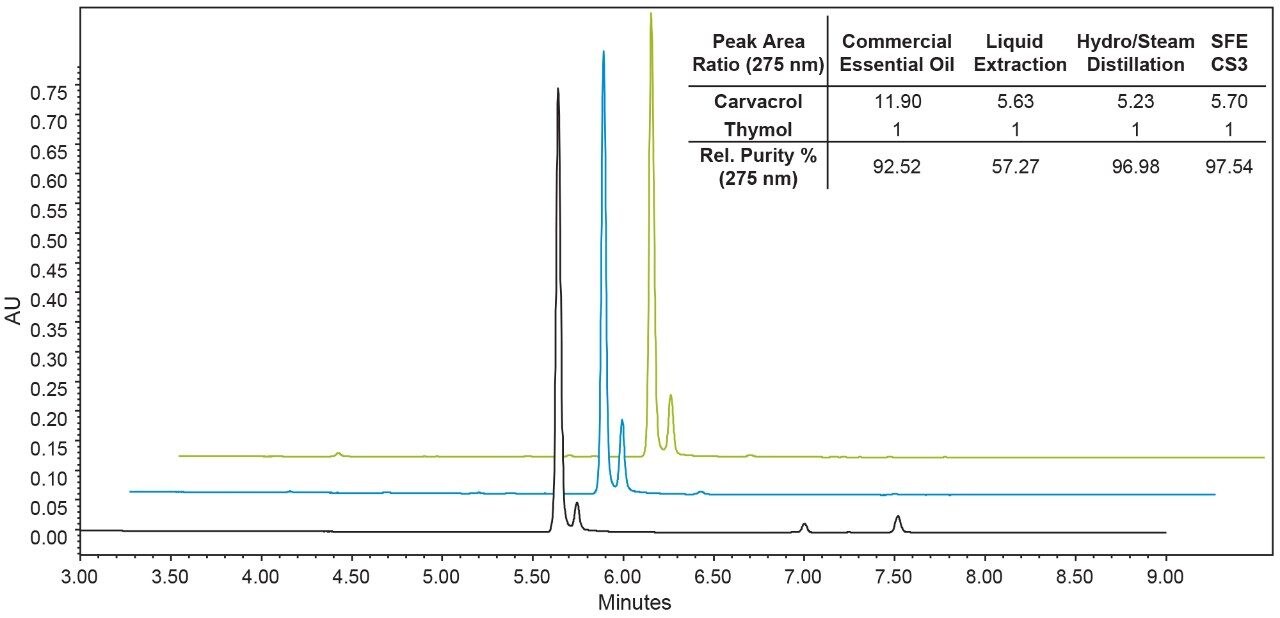
Recent natural products research suggests that the therapeutic effect observed using natural product therapies can be influenced by the ratios of the primary active components to other minor “synergistic” secondary components that enhance the therapeutic effect of the primary active components.3 As a result, it is often desirable for the extracts or essential oils of natural products to mimic the profiles found in the native plant. Specifically with regard to terpenes, producing a “plant similar” extract is important because the terpenes are the flavor and aroma compounds responsible for providing the smell and taste of the natural product,6 in addition to potentially modulating other active therapeutics.3 The inset table in Figure 5 compares the terpene ratios for each essential oil in this study. The commercially produced essential oil is included for reference. The liquid extraction of the raw oregano shows the relative amounts of carvacrol and thymol present in the starting oregano leaves. The SFE terpene fraction was the most similar to the original dried oregano; using SFE the ratio of carvacrol to thymol was 5.7:1 compared to 5.6:1 for the plant and 5.2:1 for hydrodistillation. The commercial essential oil of oregano (a product of commercial steam distillation) contained almost twice as much carvacrol compared to the dried oregano, but this should not be surprising since it was not produced from the same lot as the oregano used in this study.
SFE with fractionation allows for the flexibility to target and enrich specific compound classes in natural products. Waters Bio-Botanical Extraction System was used to generate terpene-rich and terpene-depleted extracts of oregano, and Waters H-Class UPLC system was used to quickly compare the quality of various extracts generated from an oregano feedstock. An SFE-SFF terpene fraction was compared to a distilled fraction of dried oregano feedstock; fractionation using the BBES provided the most similar terpene profile to the native plant and with the highest relative UV purity. These results show that comparable essential oils can be produced using both distillation and SFE-SFF. In cases where natural products contain thermally labile compounds and in whole-plant value-added applications, where essential oil production and extraction of additional non-volatile compounds are both desirable, extraction using the Bio-Botanical Extraction System with fractionation is a better alternative to distillation.
720006301, June 2018