Evaluate the linearity, precision, and limits of quantification of the Alliance HPLC, ACQUITY Arc Bio, and ACQUITY UPLC H-Class Bio liquid chromatography (LC) systems using a Protein A chromatography method for the concentration (titer) determination of the monoclonal antibody (mAb) trastuzumab (Herceptin).
The performance characteristics of the Waters Alliance HPLC System, ACQUITY UPLC H-Class Bio System, and the ACQUITY Arc Bio System for the Protein A column based determination of mAb concentration are compared.
HPLC Protein A affinity chromatography is a common and effective method for the determination of titer in mAb and Fc fusion protein cell culture processes. This method can also be used for the concentration determination of these proteins throughout their purification and formulation steps when sample matrix components or low concentrations prohibit the use of UV absorbance measurements. In this tech brief, a column packed with immobilized Protein A is used to bind the target protein. Protein A binds to the Fc domain of immunoglobulin G (IgG) 1, 2, 4, and fusion proteins with high specificity and avidity under favorable conditions. Once bound to the Protein A, the target protein can then be dissociated by altering the pH, ionic strength, and amount of denaturant in the mobile phase. This technique is typically performed on a high-performance liquid chromatographic (HPLC) system due to pressure constraints on the chromatography column (maximum of ~2600 psi).
Waters has developed a biocompatible version of the ACQUITY Arc System that was originally launched in 2016. That system was designed as a stepping stone in performance between the Alliance HPLC System and the ACQUITY UPLC H-Class System. The introduction of a corrosion resistant flow path where the wetted surfaces of the system are comprised of titanium and MP35N alloy components makes this system ideal for biochemical separations under salty or extreme pH conditions, such as Protein A affinity chromatography. Additionally, the ACQUITY Arc Bio System provides the ability to alter flow path pre-injector gradient mixing volume by easily switching between two mixing pathways. Here we compare the performance of an ACQUITY Arc Bio System to an Alliance HPLC System and ACQUITY UPLC H-Class Bio System for the separation of trastuzumab via Protein A chromatography.
A Poros Protein A (2.1 mm x 30 mm) affinity column from ThermoScientific was used to separate a dilution series of trastuzumab starting at 4 mg/mL and halving the concentration nine times down to 7.8 (μg)/mL. Manufacturer recommended chromatography conditions were followed and included a 0.5 minute column equilibration (50 mM phosphate, 150 mM NaCl at pH 7.0), a 1.0 minute step elution (100 mM phosphate at pH 2.5), and a 1.5 minute re-equilibration. An injection volume of 10 μL was used and UV detection was performed at 280 nm with a 3 mL/min flow rate on the Alliance HPLC and ACQUITY Arc Bio systems. The ACQUITY UPLC H-Class Bio System has a max flow rate of 2.2 mL/min and was run at 2 mL/min, therefore method times were increased to keep mobile phase volume the same.
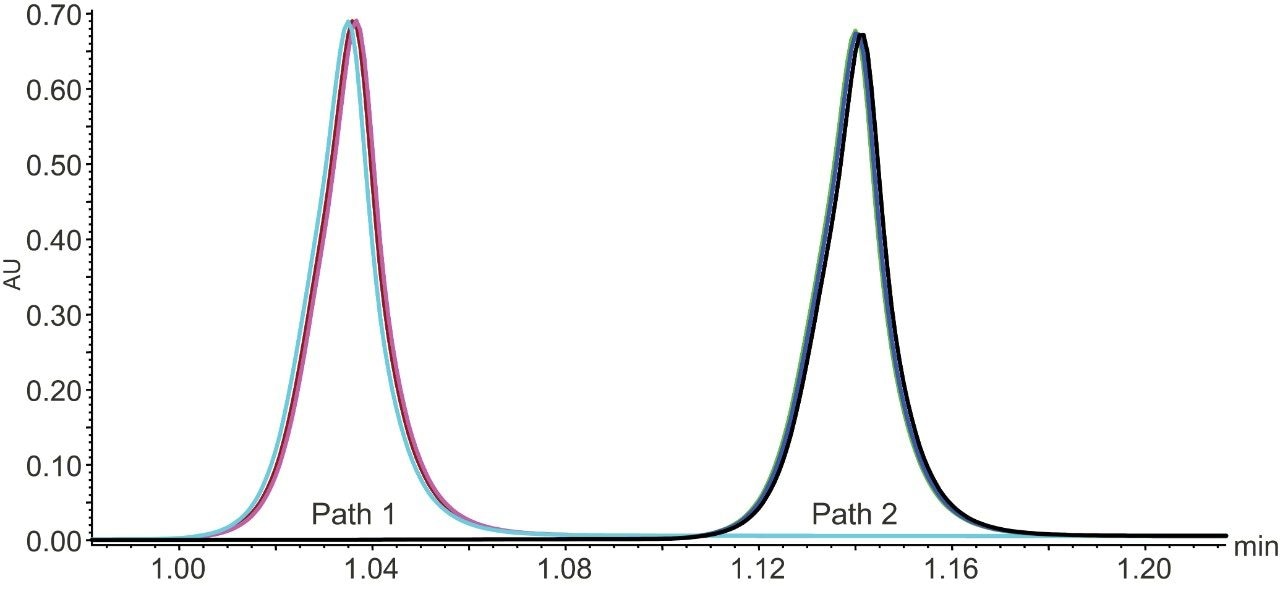
First, the two mixing pathways of the ACQUITY Arc Bio System were tested for differences in performance in the determination of the titer of trastuzumab (Figure 1). Mixing Path One has a larger dwell volume by ~0.3 mL. As Figure 1 shows, the larger dwell volume of this path increases the retention time by 0.104 minutes but does not alter retention time reproducibility as both pathways show relative standard deviations over the three injections of 0.1%. The greater mixing volume of Path One effectively decreases the steepness of the step gradient employed for the elution step and as a result the peak height is visibly decreased for this path. The average peak area for the two mixing pathways showed negligible differences (<1%) in peak area which are likely due to integration.
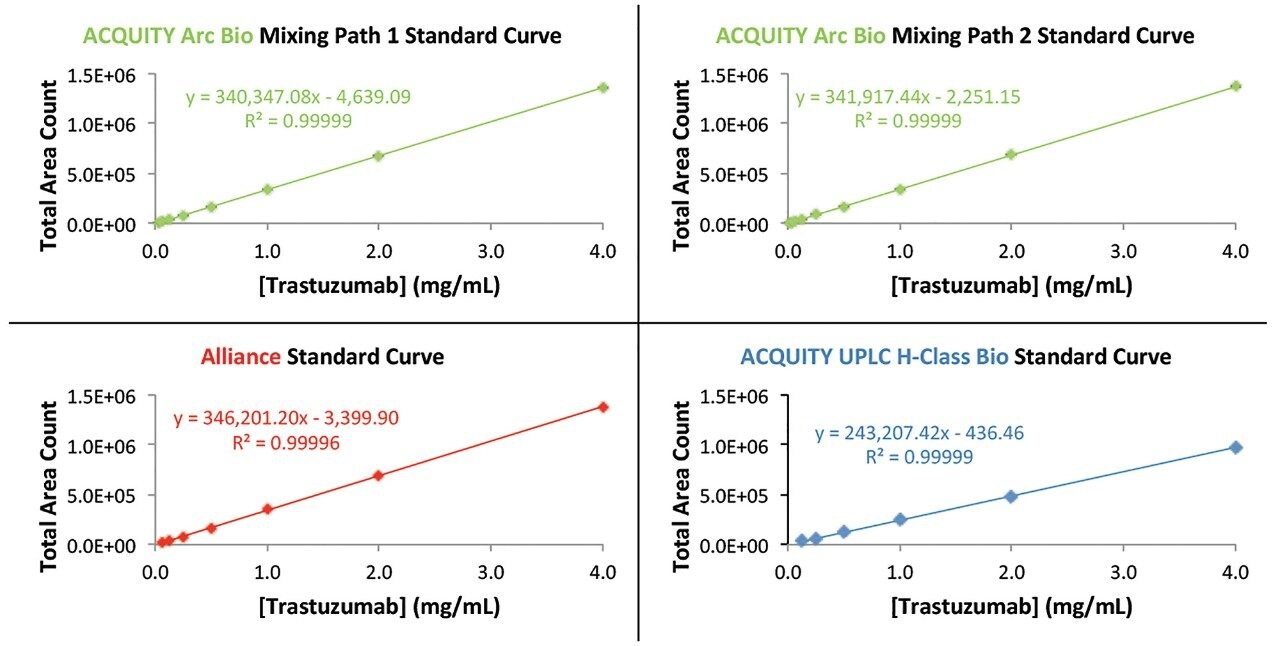
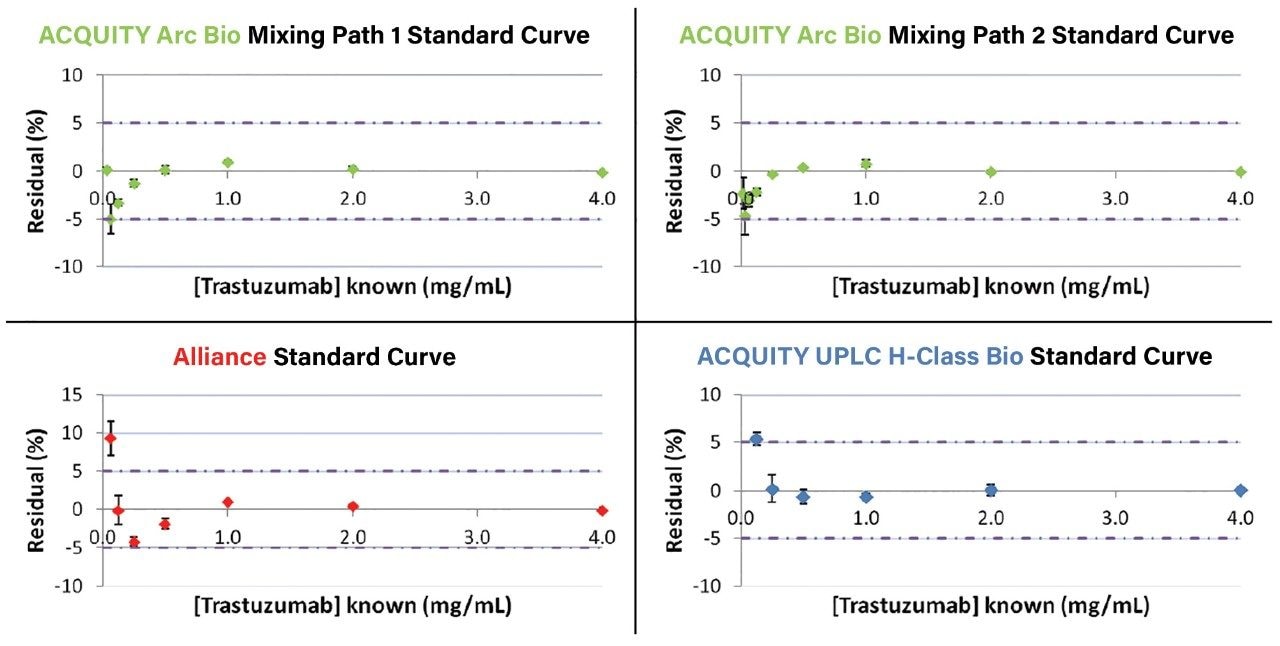
Next, calibration curves of trastuzumab were generated on all three systems (Figure 2). All three systems produced a correlation coefficient of ≥0.9999 with positive x-intercept values of less than or equal to 0.0136 mg/mL. From these linear response curves the percent relative residual values were determined. Both mixing pathways on the ACQUITY Arc Bio System provided a linear fit to lower concentration levels than the ACQUITY UPLC H-Class Bio and Alliance systems as defined by a relative residual value of less than or equal to 5% (Figure 3). The improved performance of the ACQUITY Arc Bio System with respect to the Alliance HPLC System is mostly attributable to lower system dispersion of the ACQUITY Arc Bio System, while the improved performance relative to the ACQUITY UPLC H-Class Bio System is due to the increased path length of the UV flow cell (10 mm vs. 5 mm).
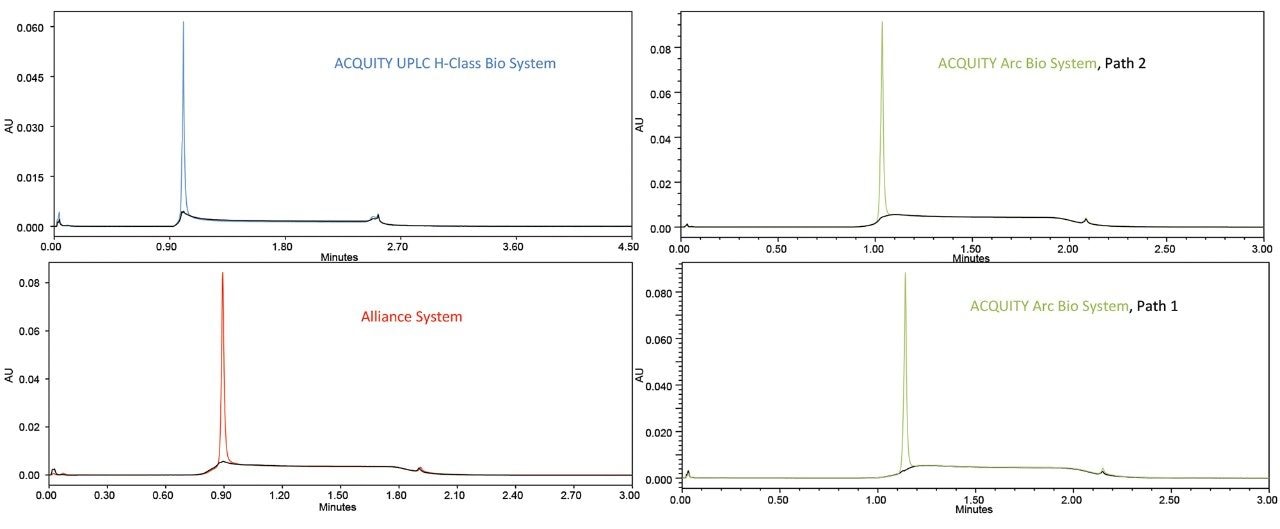
While both the ACQUITY UPLC H-Class Bio and Alliance HPLC systems are fully capable of performing Protein A based mAb concentration determinations, the ACQUITY Arc Bio System provides a broader linear range for the method evaluated in this study. In addition, the sample throughput for the ACQUITY Arc Bio System is 1.5X greater in comparison to the ACQUITY UPLC H-Class Bio System due to the higher maximum flow rate of the ACQUITY Arc Bio System (3 vs. 2 mL/min). As demonstrated here, the ACQUITY Arc Bio System offers two mixing pathways of differing volumes, both of which produce highly reproducible chromatograms in terms of retention time and total area of trastuzumab. However, the smaller volume Path 2 mixer provides slightly improved peak heights due to the step elution format of this method (Figure 4). In addition, the iron-free flow path of the ACQUITY Arc Bio System provides increased resistance to corrosion that can be induced by the buffers used for this analysis.

720006297, June 2018