
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates drug substance impurities can be extracted from formulated biotherapeutic drug-product such as antibody-drug conjugates using solid phase extraction chemistry.
Oasis SPE chemistries can be used to extract small hydrophobic impurities from biological matrices in an efficient on-line format.
The production of an antibody-drug conjugate (ADC) relies on the conjugation of potent drugs to a monoclonal antibody (mAb) to produce a therapeutic modality that enables the targeted delivery of toxic payloads to tumor surfaces with reduced systemic toxicity effects to healthy tissue. Trace levels of non-conjugated free drug impurities in formulated ADCs reduce the effective therapeutic window and pose a risk to patient safety. For these reasons, characterization of residual free drug impurities is required to ensure a safe and efficacious product. Reversed Phase Liquid Chromatography (RPLC) techniques are well established in the analysis of small molecules associated with pharmaceuticals, but direct injection of ADC samples onto a RP column without prior sample treatment can lead to column fouling and matrix effects. Protein precipitation techniques are commonly employed in the sample preparation phase to separate protein components from the hydrophobic free drug species. While effective in removing interfering protein matrices, these labor intensive approaches are frequently performed off-line requiring additional resources and may be subject to potential sample loss through excessive handling.
Solid Phase Extraction (SPE) chemistries offered in off-line and on-line formats such as the Waters Oasis SPE Products address these challenges through the targeted extraction of free hydrophobic drug species. Of the six chemistries offered, the Oasis MAX line is particularly well suited for ADCs as the sorbent material is engineered as a hydrophobic polymer chain interspersed with quaternary amine functional groups. Conceptually (Figure 1), molecules such as ADCs will not be adsorbed on the Oasis MAX Column because both the ADC molecules and sorbent surface bear the same net positive charge under acidic conditions. The small hydrophobic non-conjugated or free drug species are adsorbed to the hydrophobic polymer backbone of the SPE sorbent, and enriched for downstream analysis. This concept is demonstrated in Figure 2 where a sample of the mAb trastuzumab was spiked with a surrogate drug molecule (NAc-linker-DSEA, MW = 1167.5 Da). Using an optimized step-gradient, the non-adsorbed protein and sample matrix was eluted to waste in the first five minutes at 18%B prior to elution of the adsorbed drug surrogate at 13.5 min at 36%B. Peak composition was verified using an in-line mass spectrometer (inset). The unique selectivity of the SPE column toward free drug species facilitates a means for analysts to resolve drug impurities from biological matrices in an efficient on-line format.
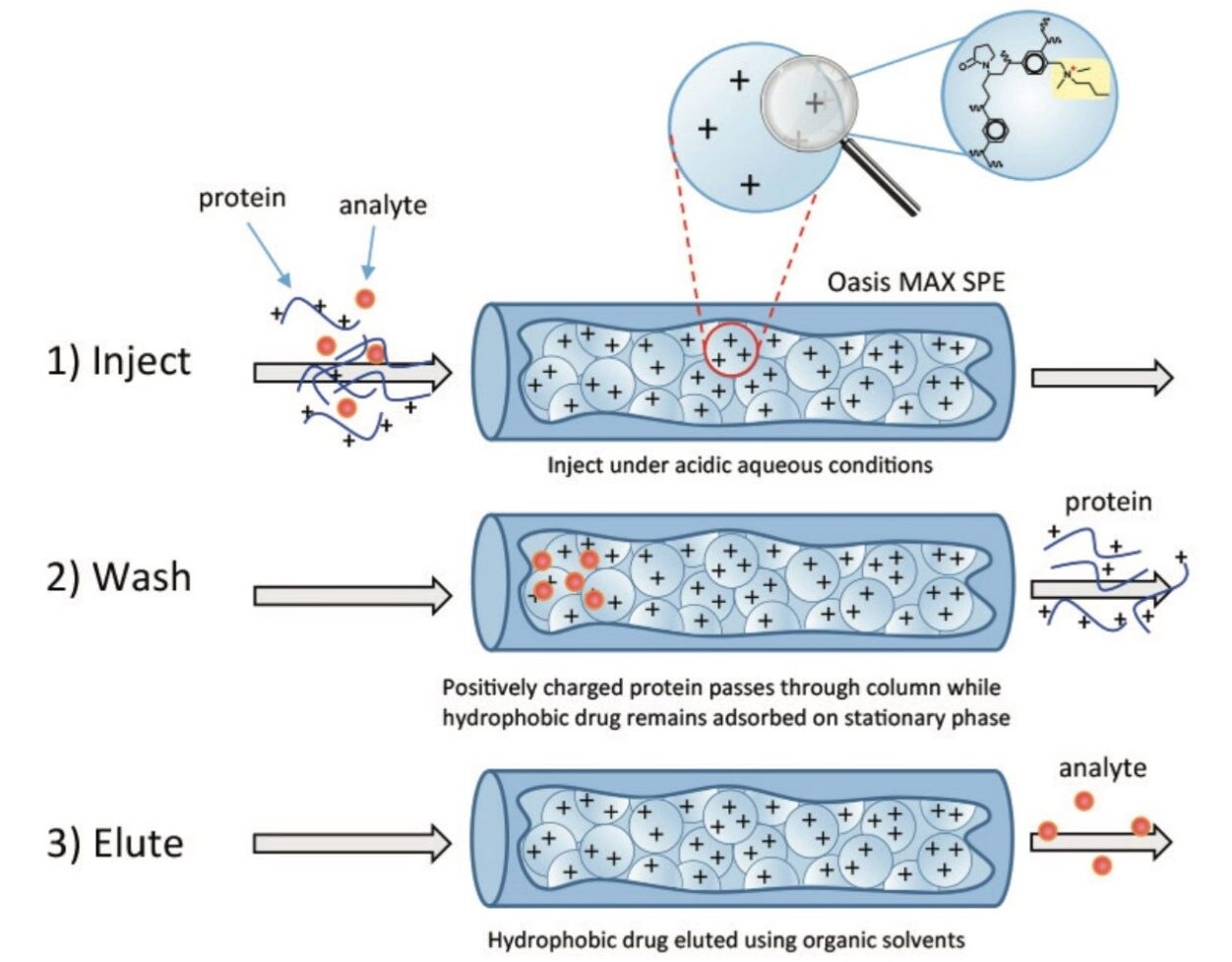
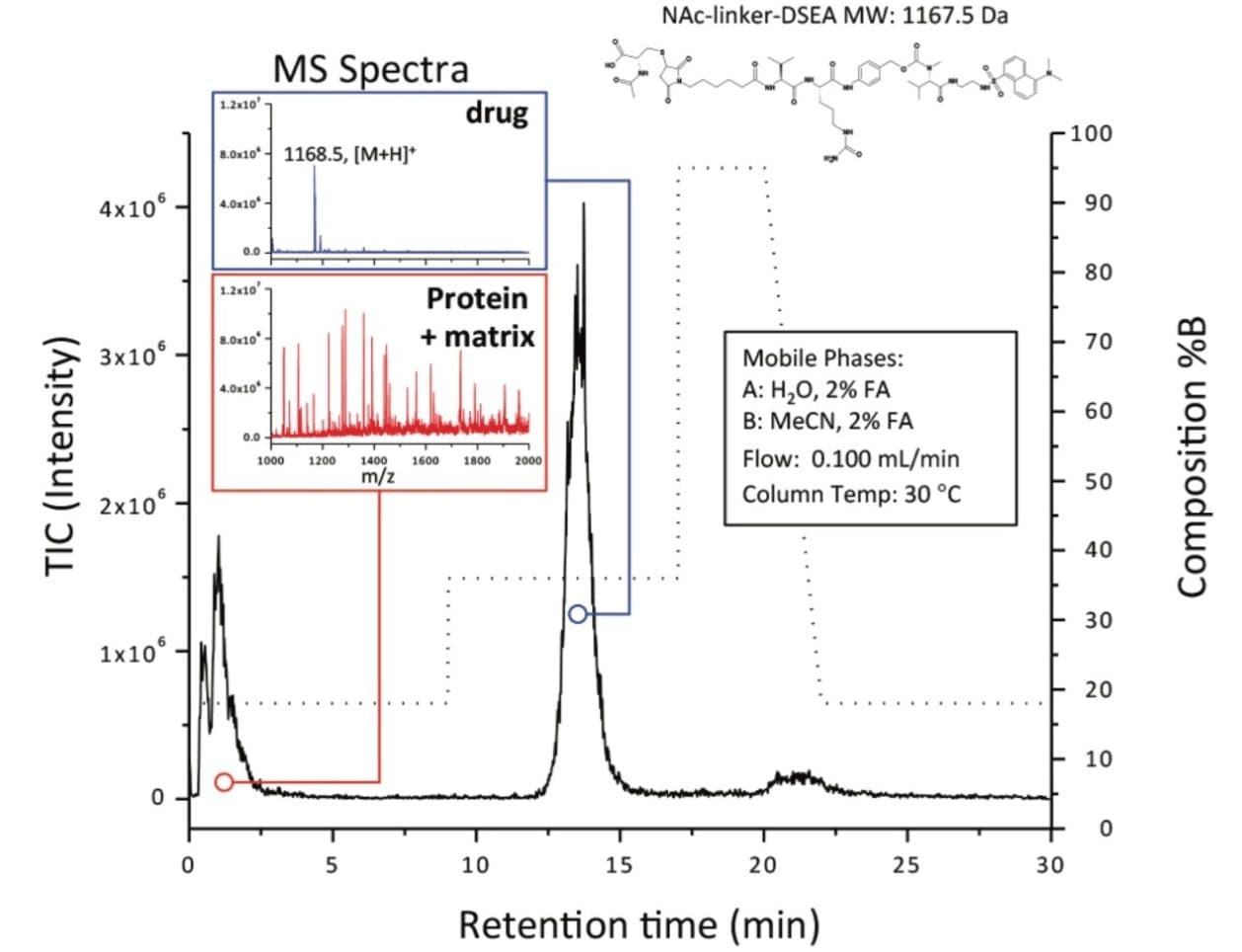
ADCs present a unique separation challenge in that free non-conjugated hydrophobic drugs require separation from their biological matrices to minimize column fouling and matrix effects in RPLC-based methods. Separation of contrasting compounds such as these is accomplished through the use of the Oasis SPE Chemistries which can be applied in an on-line format saving time and resources in the characterization of drug impurities in therapeutic biomolecules.
720006021, May 2017