
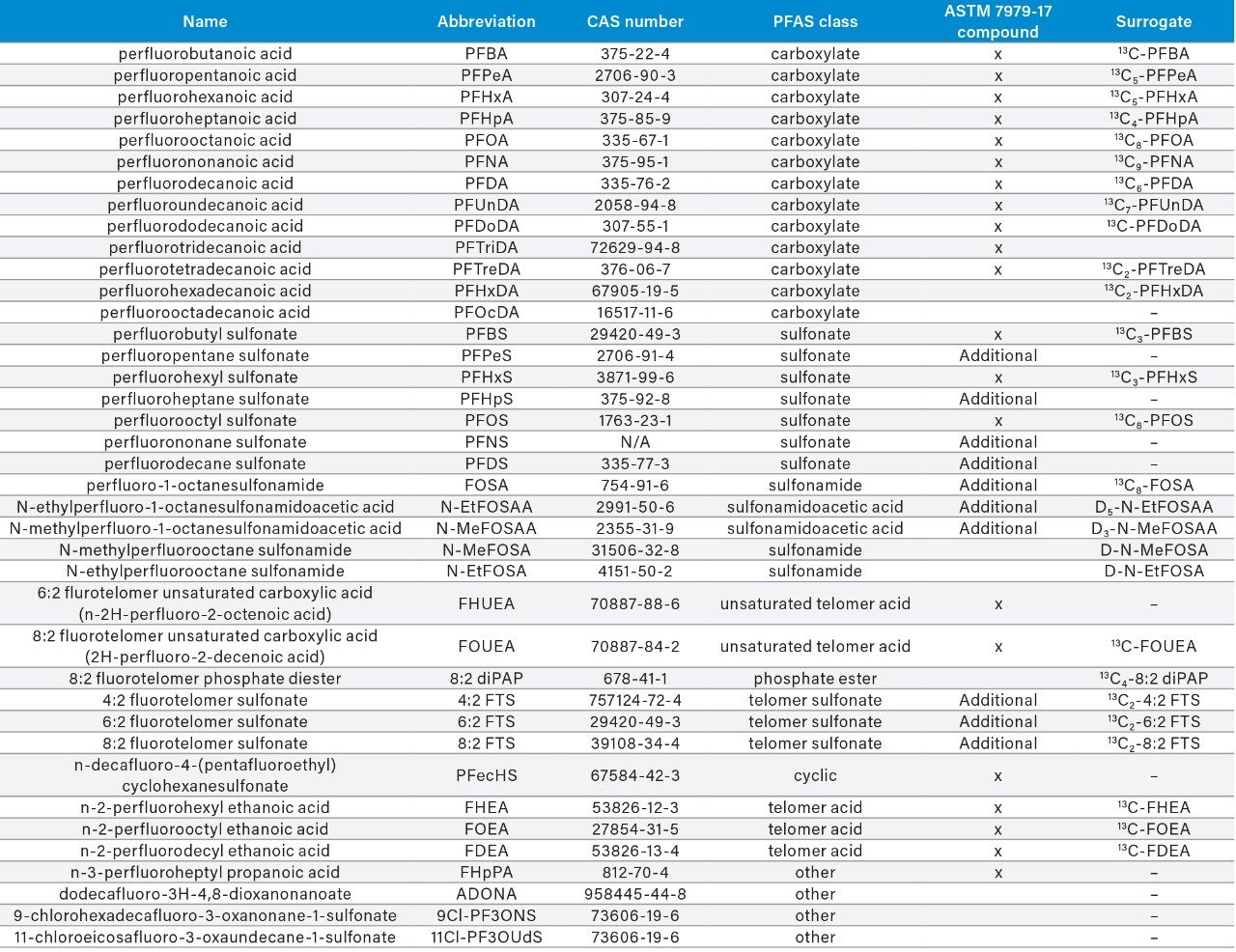
In this application note we describe the use of the recently developed ASTM 7979-17 method (EPA Region 5, Dr. Lawrence B. Zintek) to analyze PFASs of interest in environmental waters, not only as described by U.S. legislation, but also those of interest elsewhere, including newer compounds (ADONA, 9Cl-PF3ONS, and 11Cl-PF3OUdS).
Performing the ASTM 7979-17 method on the Xevo TQ-XS allows:
Perfluoroalkyl substances (PFASs) are a class of anthropogenic compounds that are found in a range of consumer goods and industrial processes due to their chemical properties. Common uses include firefighting foams, insecticide formulations, water-resistant coating, floor polishes, and oilresistant coatings for paper products approved for food contact. Due to their widespread use and subsequent leaching from materials, PFASs are so ubiquitous that they are frequently detected throughout the environment and in 2009, they were classified as persistent organic pollutants (POPs) within the Stockholm Convention.1 Due to their persistent, ubiquitous nature, and possible toxicity, most regulatory agencies worldwide closely monitor the use, occurrence, and impact of both traditional/common and newer, replacement short-chain PFASs.
For monitoring and research purposes, ng/L, or part-per-trillion (ppt), detection of PFASs is often required. Within the U.S. drinking water is regulated under the Safe Drinking Water Act, while other environmental waters are regulated under the Clean Water Act. In the third Unregulated Contaminant Monitoring rule (UCMR3)2 for drinking water, the U.S. EPA has required monitoring of six different PFAS compounds with a minimum reporting level in the range of 30 to 200 ng/L for each component. The U.S. EPA has also issued a health advisory3 acute level at 70 ng/L based on the best available peer-reviewed studies of PFAS effects. Within the EU, drinking water is regulated under the Drinking Water Directive, 98/83/EC, while other environmental waters are regulated under the EC Water Framework Directive (WFD), 2013/39/EU.4 In the WFD, PFOS is specifically identified as a “priority hazardous substance.”
In this application note we describe the use of the recently developed ASTM 7979-17 method (EPA Region 5, Dr. Lawrence B. Zintek)5 to analyze PFASs of interest in environmental waters, not only as described by U.S. legislation, but also those of interest elsewhere, including newer compounds (ADONA, 9Cl-PF3ONS, and 11Cl-PF3OUdS). Since many countries look to the U.S. EPA and other agencies for guidance, it was decided to include as many compounds in a single analysis as was feasible at relevant detection levels.
The ASTM 7979-17 currently covers the analysis of 21 PFAS compounds, with 10 additional compounds listed for consideration in the appendix of the method. For this analysis, eight additional compounds were added to the method to bring the total number of PFAS analytes to 39. Three of the compounds added to the method are emerging PFAS compounds of interest including ADONA, 9Cl-PF3ONS (the main component of F-53B), and 11Cl-PF3OUdS (minor component of F-53B). Table 1 contains the compound information for all of the PFAS compounds included in this method. All standards were obtained from Wellington Laboratories (Guelph, Ontario).
A Certified QC Standard (Cat #731) from ERA (Golden, CO), for use with ground and surface waters, was used as an instrumental QC check throughout the analysis. The standard contained a mix of 12 PFAS compounds. Certified values and QC Performance Acceptance Limits for each compound in the mix are provided with the standard, making instrumental QC evaluation fast and straightforward.
Since required detection limits are in the low ng/L range and as a result of the widespread use of PFASs, specific challenges must be addressed for sample collection, preparation, and analysis. There are many common sources of PFAS contamination in the field and laboratory. In the field, caution should be taken to avoid Teflon containing materials (such as waterproof clothing/jackets), plastic clipboards, waterproof notebooks, and chemical ice packs. In the lab, items to avoid include sticky notes, certain glass disposable pipettes, aluminum foil, vial caps with Teflon seals, and LDPE containers, to name a few. In fact, it is recommended that all laboratory supplies be checked for PFAS contamination before use, as is practical. Contamination is unavoidable from the chromatographic system. Therefore steps should be taken to minimize any system contribution, and as such, the Waters PFC Analysis Kit (p/n:176001744) for the UPLC system was utilized. The kit is comprised of PFAS-free components (such as PEEK tubing to replace the conventional Teflon coated solvent lines) and a PFC Isolator Column that helps to delay any residual background interferences from co-eluting with the analytical peak. Installation of the PFC Analysis Kit is straightforward and quick.6
Samples were provided by the U.S. EPA Region 5 through a Cooperative Research and Development Agreement (EPA CRADA #884-16). Provided samples included reagent water, surface (river) water, ground water, influent waste water, and effluent waste water. Each water sample was spiked with a low and high level of a selection of PFAS compounds (three replicates of each concentration) prior to being received in the lab. Two blanks of each sample were also received.
The entirety of each 5-mL water sample was used to avoid any compound loss. Each sample was spiked with 160 ng/L of isotopically labeled surrogates (see Table 1). The surrogates are added to the sample prior to any preparation in order to determine method recoveries. 5 mL of methanol was then added to each water sample and vortexed for 2 min. The entire 10 mL sample was filtered using a disposable polypropylene syringe with a glass filter (25 mm diameter, 1.0 μm pore size) stacked on top of a polypropylene GHP filter (25 mm diameter, 0.2 μm pore size). Following filtration, 10 μL of acetic acid was added to each sample. An aliquot of each sample was transferred to a polypropylene autosampler vial and sealed with a Polyethylene Cap (p/n: 186005230).
|
LC system: |
ACQUITY UPLC I-Class fitted with PFC Kit |
|
Column: |
ACQUITY UPLC CSH Phenyl Hexyl 1.7 μm , 2.1 × 100 mm |
|
Column temp.: |
35 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
30 μL |
|
Mobile phase A: |
95:5 Water:methanol + 2 mM ammonium acetate |
|
Mobile phase B: |
Methanol + 2 mM ammonium acetate |
|
Time (min) |
Flow rate (mL/min) |
%A |
%B |
|
0 |
0.3 |
100 |
0 |
|
1 |
0.3 |
80 |
20 |
|
6 |
0.3 |
55 |
45 |
|
13 |
0.3 |
20 |
80 |
|
14 |
0.4 |
5 |
95 |
|
17 |
0.4 |
5 |
95 |
|
18 |
0.3 |
100 |
0 |
|
22 |
0.3 |
100 |
0 |
|
MS system: |
Xevo TQ-XS |
|
Ionization mode |
ESI- |
|
Capillary voltage: |
1.0 kV |
|
Desolvation temp.: |
500 °C |
|
Desolvation gas flow: |
1100 L/hr |
|
Cone gas flow: |
150 L/hr |
|
Source temp.: |
120 °C |
|
Method events: |
Divert flow to waste from 15 to 21 min. |
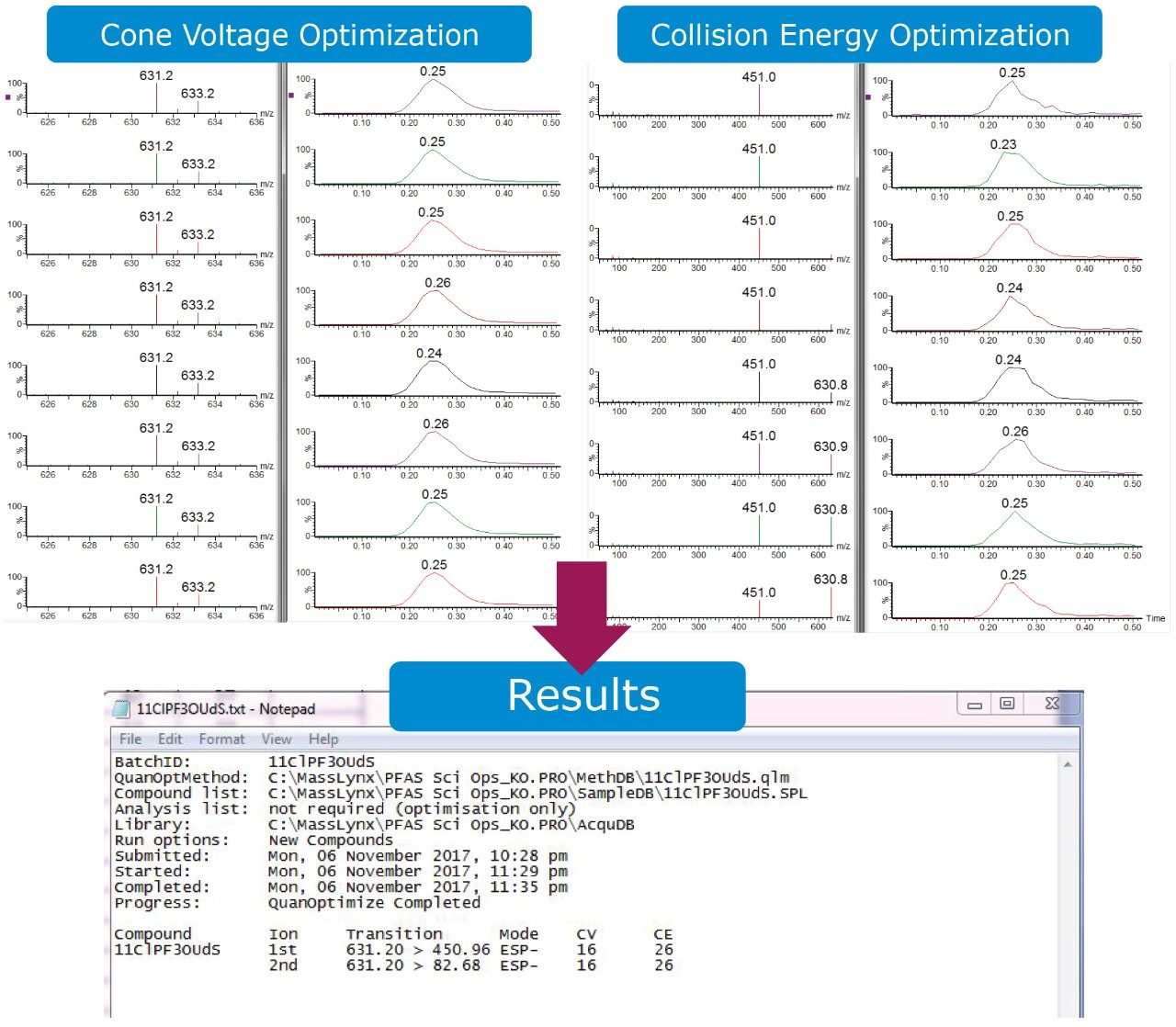
All MRM parameters for each compound were optimized using the QuanOptimize tool in MassLynx. QuanOptimize will automatically determine the parent ion, fragment ions, cone voltage, and collision energy required for each individual compound through injection. The MRMs generated from QuanOptimize for this method are detailed in Appendix Table A. By providing the mass or chemical formula in the MassLynx sample list, QuanOptimize will step through the cone voltages and collision energies designated in the QuanOptimize method. The software then automatically processes the results and generates a report with the MRM transition and corresponding cone voltage and collision energy (Figure 1). This tool also allows rapid and simple optimization of MRM method parameters for new compounds that may need to be added to the analysis method in the future.
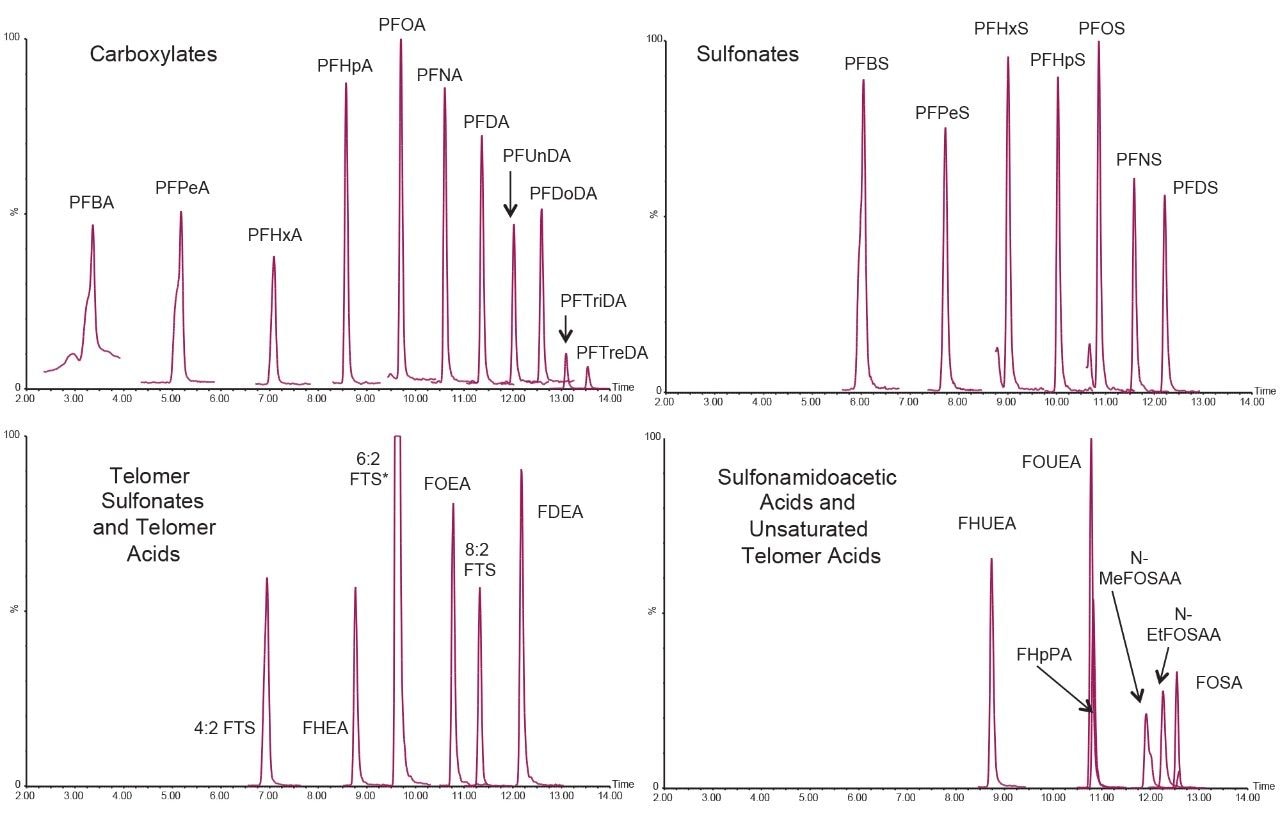
Sample analysis was performed as described in ASTM 7979-17. One minor change was made to the mobile phase composition. For this work, methanol was used in place of acetonitrile. Also, the concentration of ammonium acetate added to both mobile phases was reduced to 2 mM from the suggested 20 mM in the official method. Both changes were made due to solubility concerns of ammonium acetate in acetonitrile. These changes had no negative effects on method performance, such as peak resolution or response but made the LC method more robust. An overlaid chromatogram of all native compounds and isotope surrogates is demonstrated in Figure 2.

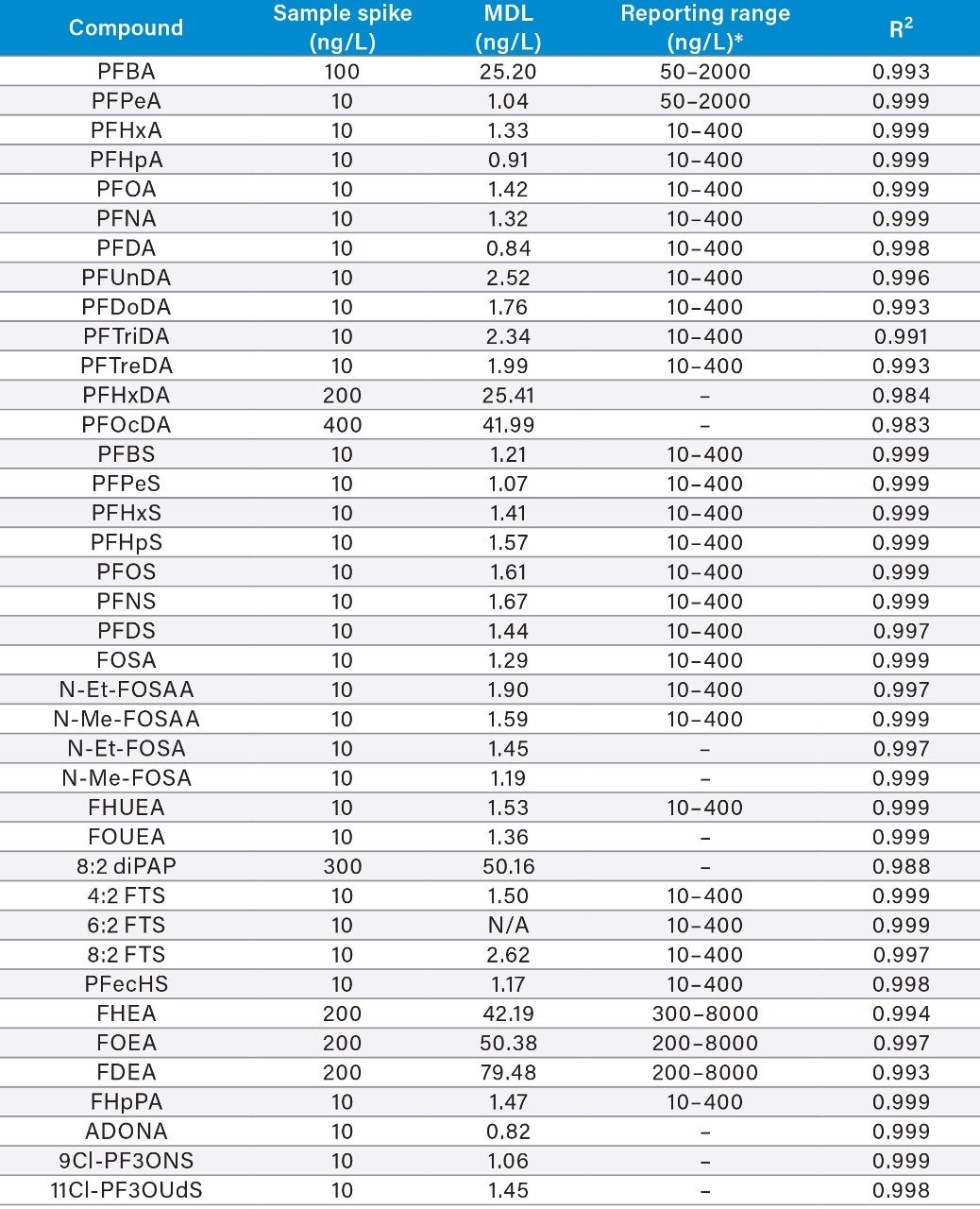
A Method Detection Limit (MDL) study was performed to assess the sensitivity of the sample analysis method. Nine replicate samples were prepared by spiking reagent water with various concentrations of the PFAS analytes (Table 2) and 80 ng/L of the surrogate standard solution. All samples were taken through the sample pre-treatment procedure prior to analysis. MDL values were calculated using the equation:
MDL = SD x tn-1 , where SD = standard deviation of n replicates and tn-1 = 2.896 (student t value for n-1 samples)
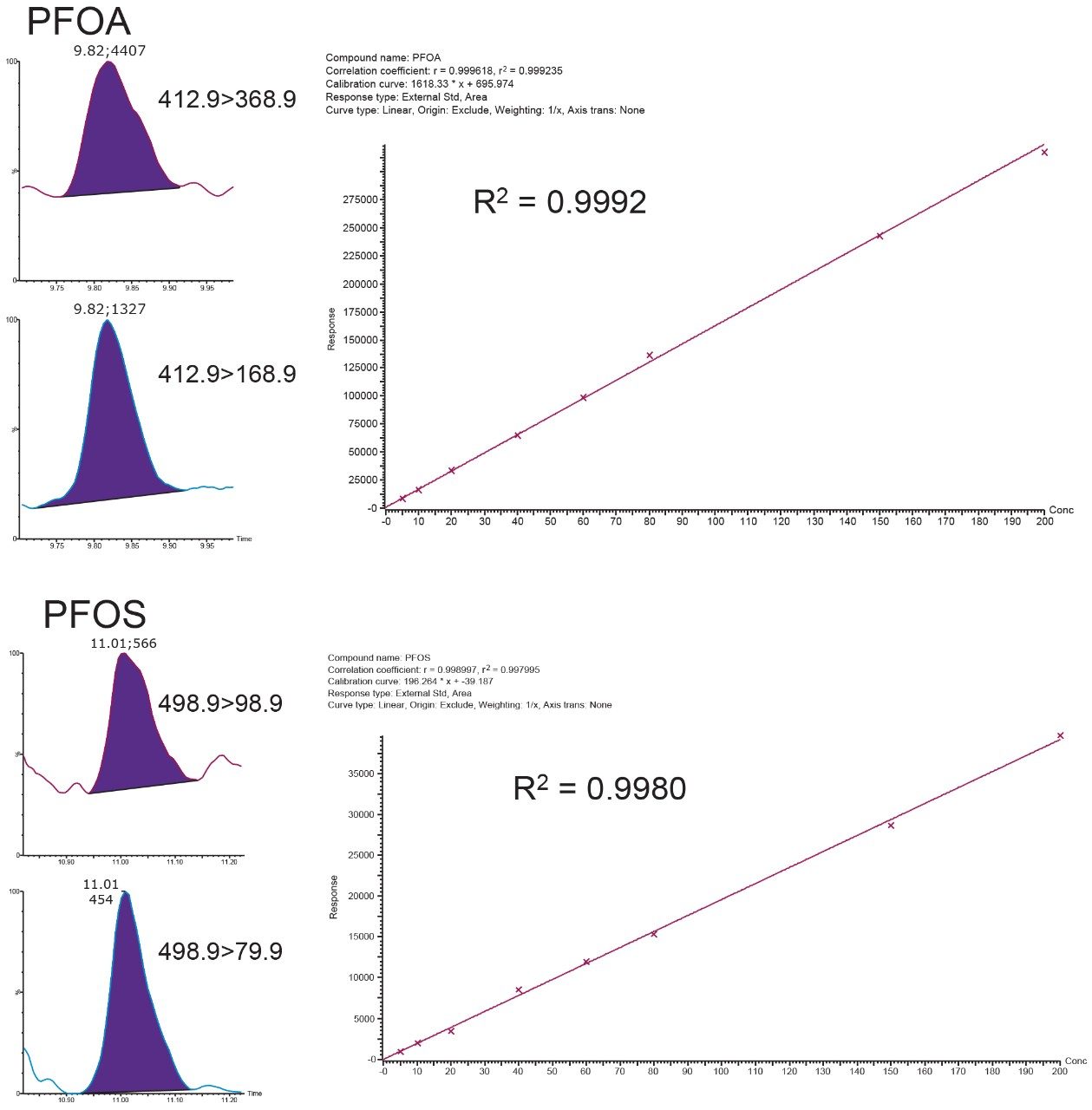
The MDL values were all well below the required reporting limits stated in the ASTM 7979 method, indicating that this method is more than suitable for this analysis. An MDL value could not be calculated for telomer sulfonate isomer 6:2 FTS due to contamination of this compound in the solvents used for sample preparation. The remaining PFAS compounds experienced no background interference or contamination. Calibration curve requirements in ASTM 7979 require an R2 value of 0.98 or greater for linear regression fit. All compounds were within this requirement, as highlighted in Table 2. Example calibration curves are also shown in Figure 3 for PFOA and PFOS. Figure 3 also shows chromatograms for PFOA and PFOS injected at 2.5 ng/L. This demonstrates the sensitivity at half the required reporting limit for these two compounds.
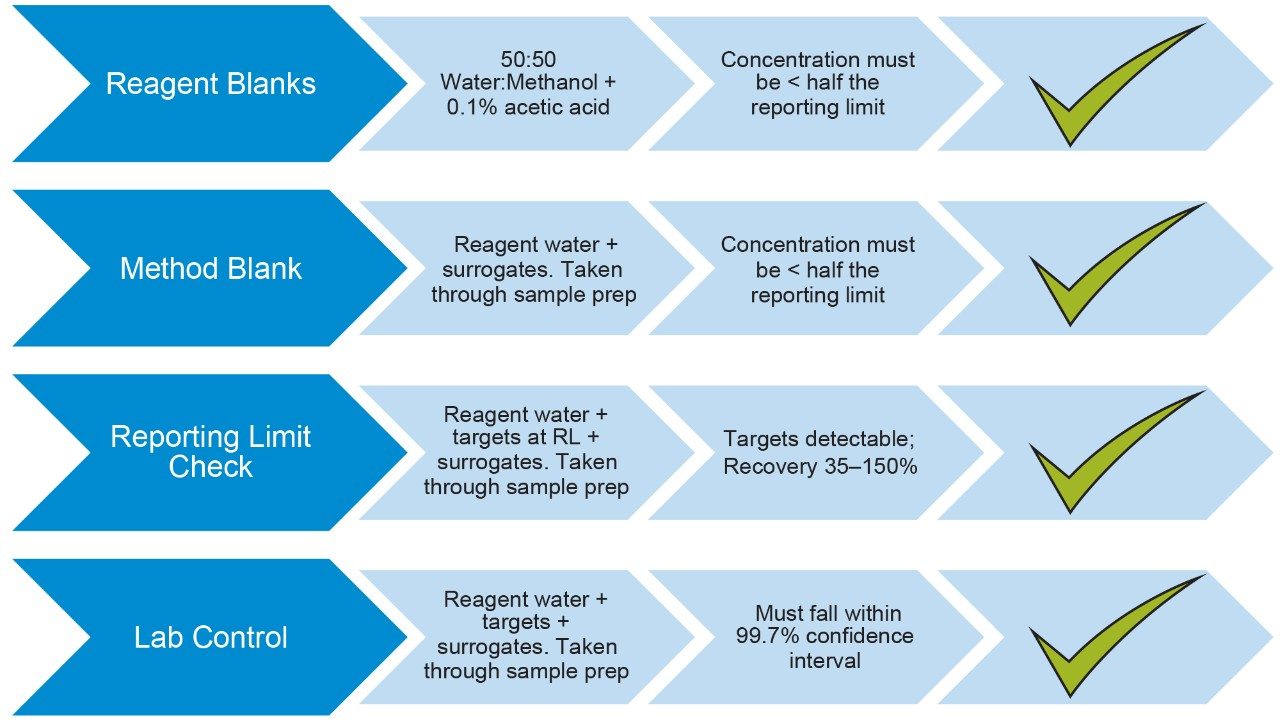
The ASTM 7979-17 method requires control samples to be run with the criteria they must pass as outlined in Figure 4. All compounds passed the control criteria, with the exception of 6:2 FTS due to solvent contamination of that compound.
Triplicates of each spiked matrix at both concentrations were prepared and analyzed using the method according to ASTM 7979-17. Only the compounds currently written into the ASTM method were spiked into the water samples. All PFAS compounds that were spiked into the various water samples were detected at both the high and low concentration spike. PFBA and PFPeA were spiked at 300 ng/L and 1000 ng/L in the low and high spike samples, respectively. 4:2, 6:2, and 8:2 FTS were spiked at 1200 ng/L and 4000 ng/L in the low and high spike, respectively. All other PFAS compounds were spiked at 60 ng/L in the low spike samples and 200 ng/L in the high spike samples. Figure 5 shows an example of all the PFAS compounds spiked at the low concentration level in the surface (river) water sample.
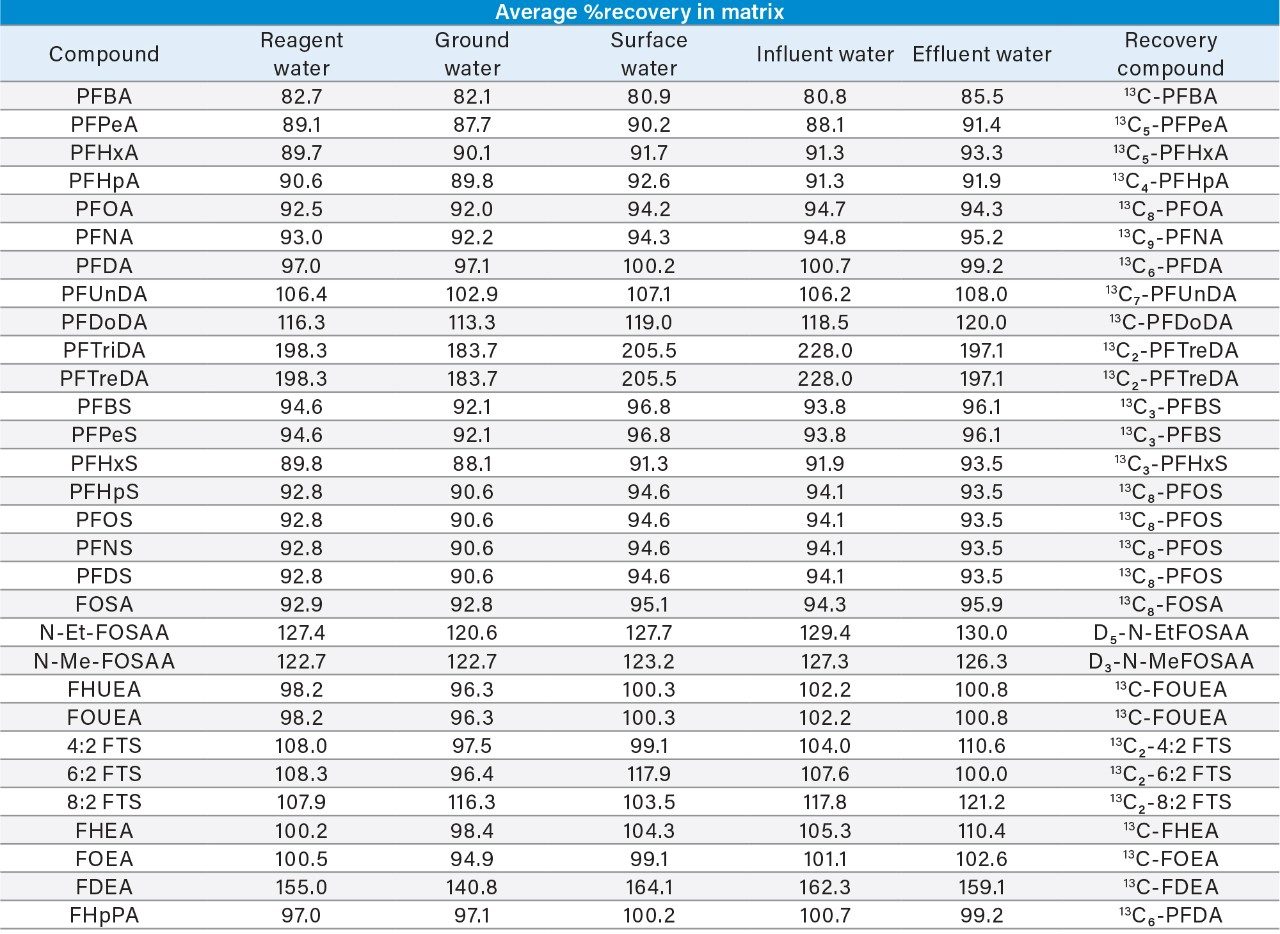
Recovery of the PFAS compounds was determined using isotope labelled surrogate standards that were spiked into the samples prior to sample pre-treatment and analysis. For compounds that did not have a surrogate available, a compound close in retention time and chemical structure was used as the surrogate. Table 3 demonstrates the percent recovery of all PFAS compounds spiked into the five water samples. ASTM 7979 requires percent recoveries to be in the range of 70% to 130%. All compounds included in the method were within this range with the exception of PFTreDA, PFTriDA, and FDEA. These compounds exhibited an enhancement effect when analyzed from a prepared sample compared to the response exhibited in solvent standards. The enhancement could be associated with co-elution of these compounds with matrix components in the sample. Correction of sample concentration can be performed if necessary based on the percent recovery exhibited by the surrogate standard or by quantification using matrix matched calibration curves.
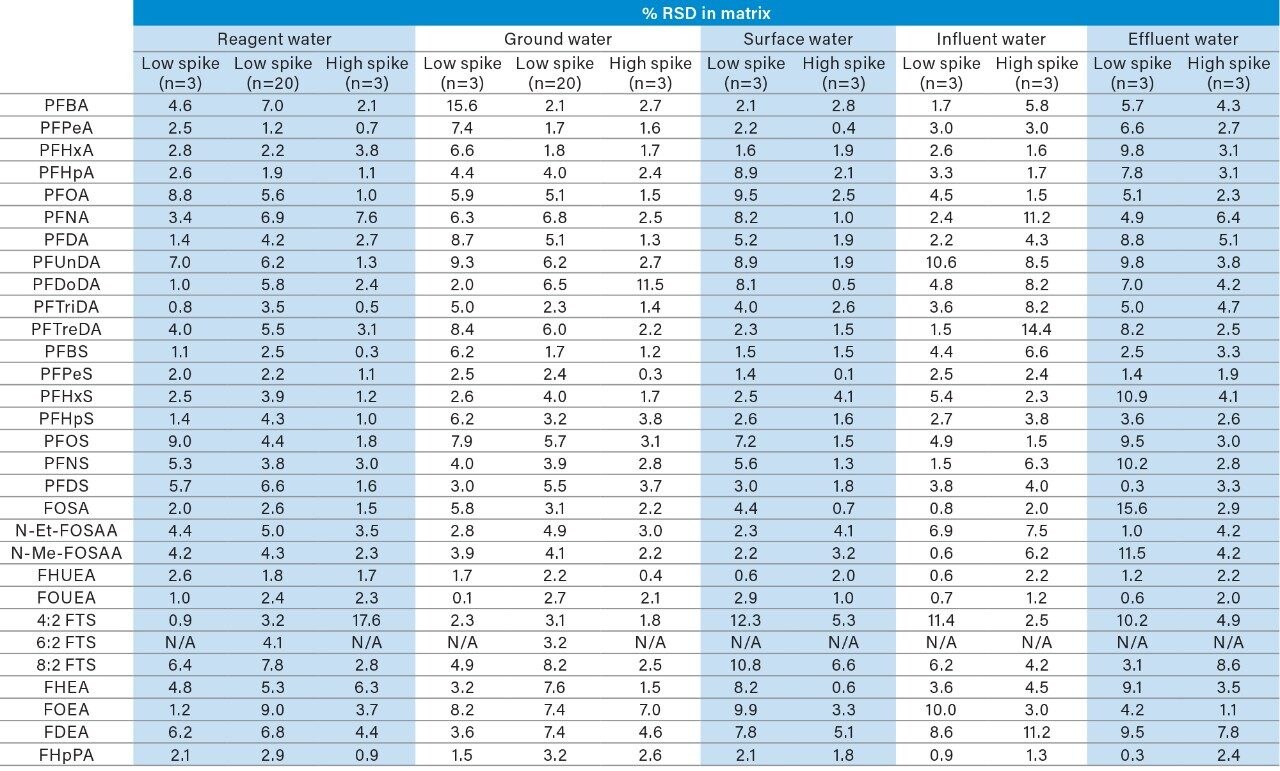
The method proved to be repeatable as well, demonstrated by the %RSD values highlighted in Appendix Table B. All matrix samples were processed in triplicate, which is demonstrated by the n=3 RSD values. These values represent the full method reproducibility. A single sample of reagent water and ground water were also injected 20 times to produce instrument repeatability data (%RSD values in Appendix Table B). Again, due to solvent contamination of 6:2 FTS, an accurate %RSD value for the method replicates could not be calculated. For the most part, the %RSD values fell below 10%, with a majority of compounds exhibiting RSDs below 5%.

720006329, June 2018