This application note demonstrates that the GlycoWorks RapiFluor-MS N-glycan sample preparation kit is readily scalable from manual, to semi-automated, and fully automated workflows.
The purpose of this application note is to demonstrate the throughput flexibility and scalability of the GlycoWorks RapiFluor-MS N-glycan reagent kit options. These include; the GlycoWorks RapiFluor-MS N-Glycan Kit for low- to medium-throughput (semi-automated) sample preparation, and the GlycoWorks RapiFluor-MS N-Glycan Kit – Automation for fully automated high throughput sample preparation.
Since its launch, the GlycoWorks RapiFluor-MS N-Glycan Kit has offered scientists a fast and reproducible method for the manual preparation of glycoprotein samples for N-glycan analysis. The novel RapiFluor-MS label and associated chemistries provide marked benefits to bioanalytical scientists in terms of sample preparation efficiency and FLR and MS detection sensitivity. This manual sample preparation workflow is ideally suited to laboratories with relatively low sample throughput or laboratories which do not have access to automated liquid handling platforms.
For other laboratories, a further opportunity exists to enhance theefficiency of GlycoWorks using automation, in particular for medium-to-high-throughput requirements. In addition to gains in laboratory efficiency, the automation of complex workflows can provide a route to method simplification and standardization. Yet, a challenge for these laboratoriesis resourcing the adaptation of existing workflows to automation and the creation and validation of automation scripts.
To address these challenges, Waters has introduced the GlycoWorks RapiFluor-MS N-Glycan Kit – Automation and a range of semi-automated and fully automated workflows which combine the power of the existing rapid RapiFluor-MS and associated chemistries with the power of automation.
|
LC system: |
ACQUITY UPLC I-Class or ACQUITY UPLC H-Class Bio with ACQUITY UPLC FLR Fluorescence Detector |
|
Column: |
ACQUITY UPLC Glycan BEH Amide, 130Å, 1.7 μm, 2.1 x 150 mm (p/n: 186004742) |
|
Column temp. |
60 °C |
|
Sample temp. |
10 °C |
|
Detection |
Ex 265/Em 425 nm |
|
Data rate |
10 Hz |
|
Initial flow rate |
0.4 mL/min |
|
Mobile phase A: |
50 mM Ammonium formate solution, pH 4.4 (p/n: 186007081) along with LC-MS-grade water [Fisher p/n W6 or equivalent] is recommended.) |
|
Mobile phase B: |
100% acetonitrile (LC-MS-grade ACN is recommended, Fisher p/n A955 or equivalent) |
|
Seal wash and LC: |
70% LC-MS grade ACN (LC-MS-grade ACN is recommended, Fisher p/n A955 or equivalent.), 30% LC-MS grade water [Fisher p/n W6 or equivalent] is recommended.) (v/v) |
|
Injection volume: |
10 μL suggested (=30 μL, DMF/ACN diluted SPE eluate, 2.1 mm I.D. columns) |
|
Injector needle: |
A stainless steel needle is recommended when injecting samples containing DMF as co-solvent |
|
Time (min) |
Flow rate (mL/min) |
%A |
%B |
Curve |
|
0 |
0.4 |
25 |
75 |
6 |
|
35 |
0.4 |
46 |
54 |
6 |
|
36.5 |
0.2 |
100 |
0 |
6 |
|
39.5 |
0.2 |
100 |
0 |
6 |
|
43.1 |
0.2 |
25 |
75 |
6 |
|
47.6 |
0.4 |
25 |
75 |
6 |
|
55 |
0.4 |
25 |
75 |
6 |
Data management: Empower 3 CDS
The following protocol was executed for method comparison purposes.
Using manual, semi-automated (Andrew Alliance), and fully automated (Tecan) workflows; 15 N-glycan samples wereprepared from the same lot of glycoprotein (Waters Intact mAb Mass Check Standard (p/n: 186006552)) and using the same GlycoWorks RapiFluor-MS N-Glycan Kit – Automation lot (p/n:176004152). Samples were prepared using reagent concentrations specified in the instructions for each workflow:
RapiFluor-MS Glycan Performance Test Standard (p/n:186007983) and RapiFluor-MS Intact mAb Standard (p/n: 186008843) were reconstituted in 50 μL water for chromatographic benchmarking and system suitability.
All prepared samples were tested on the same ACQUITY UPLC H-Class Bio UPLC-FLR System.
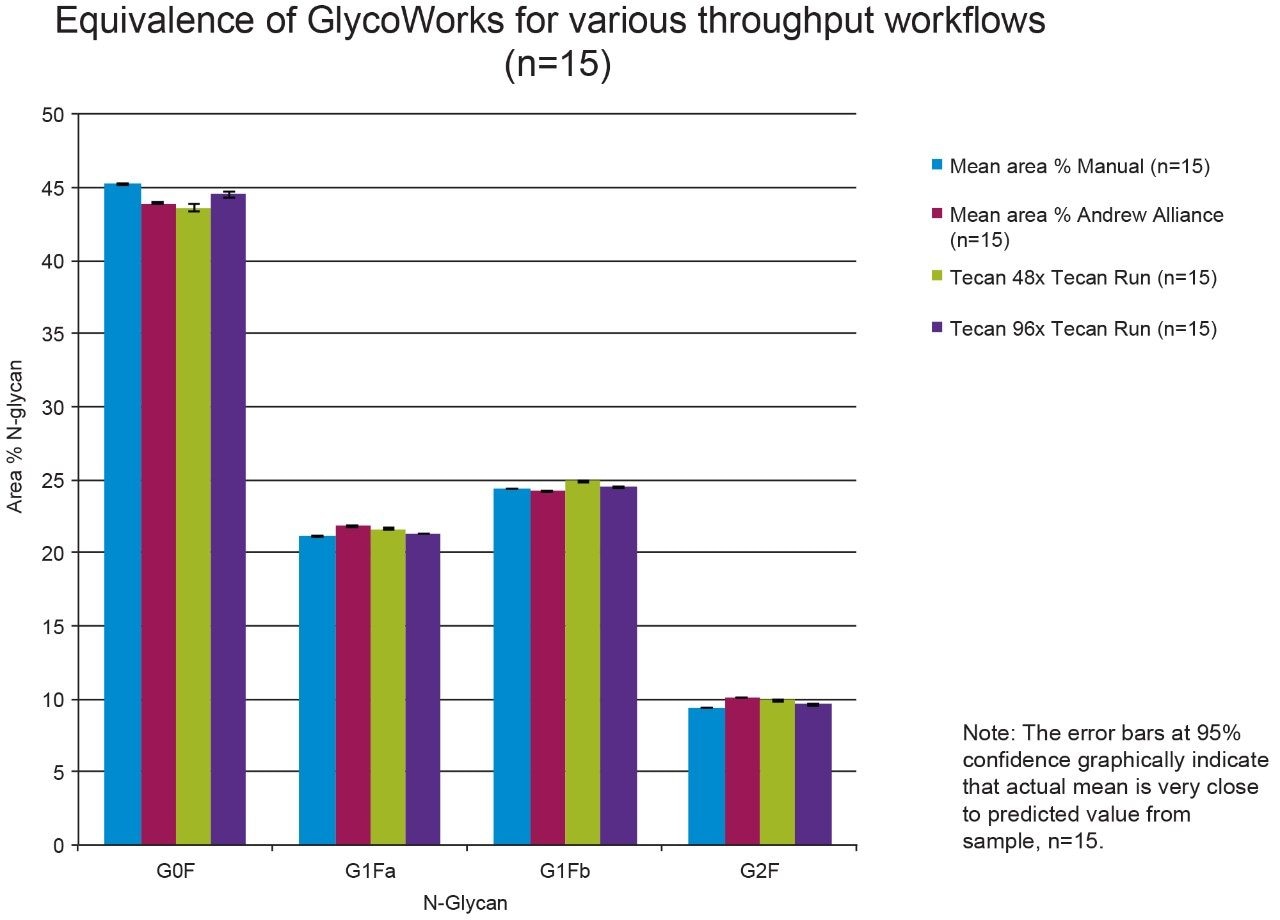
To qualitatively compare the sample preparation methods, graphical representations of the N-glycan mean area % data for selected peaks for each sample set (n = 15) was prepared (Figures 1 and 2).
Initial statistical analyses (t-test) of the method data sets indicated that the methods were not statistically equivalent (p<0.05). Additional analyses indicated that the test is limited by the high precision of each data set and a simulation using only minor adjustments to a small number of data point to values yielded p>0.05.
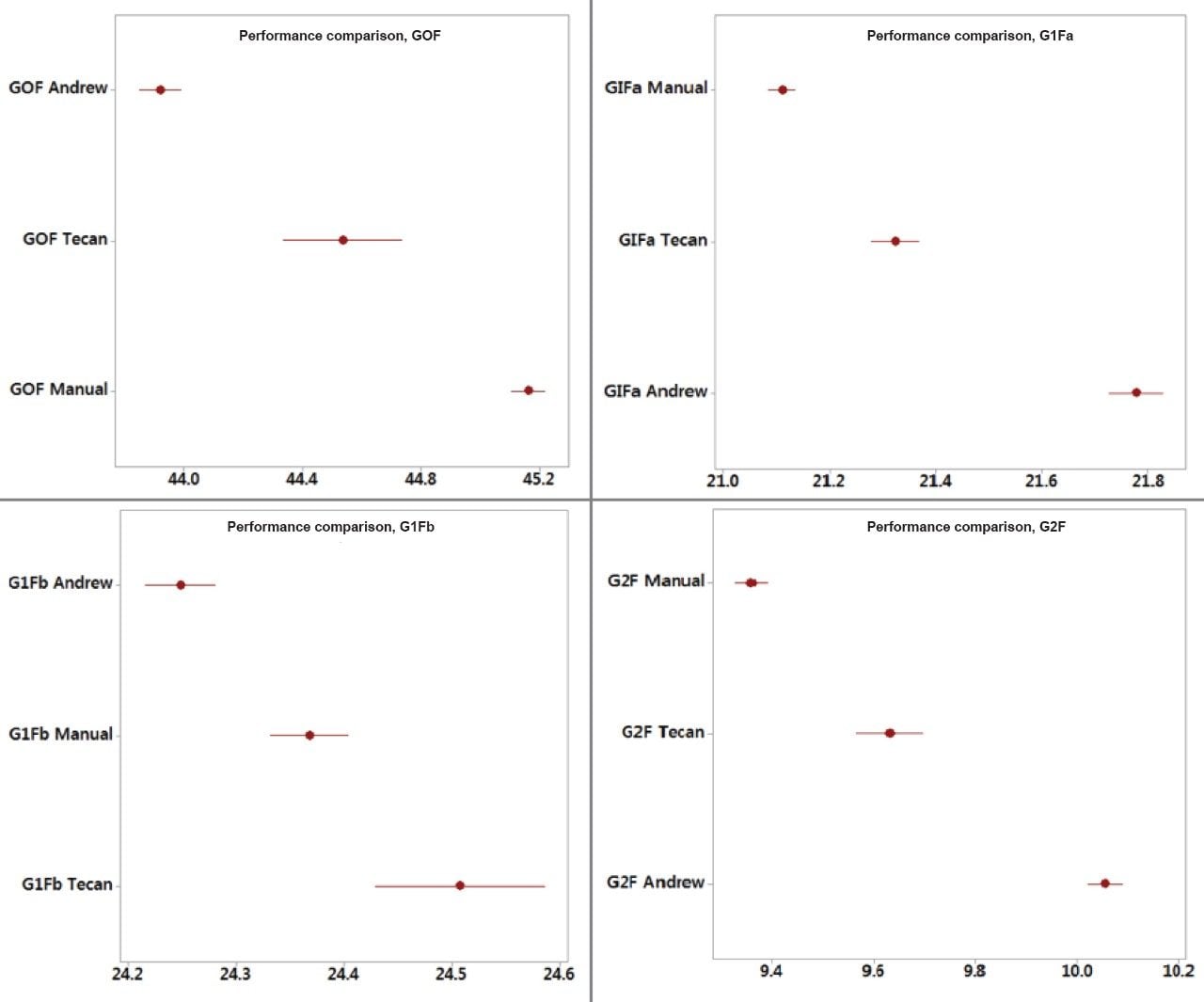
Further statistical analyses were performed to quantify the bias for the automated methods relative to the manual method and for the semi-automated and fully automated methods. Minimum to maximum values for each N-glycan area percent data set were tabulated for evaluation as well as calculations of mean %bias as estimates and to 95% confidence intervals (Tables 2–4).
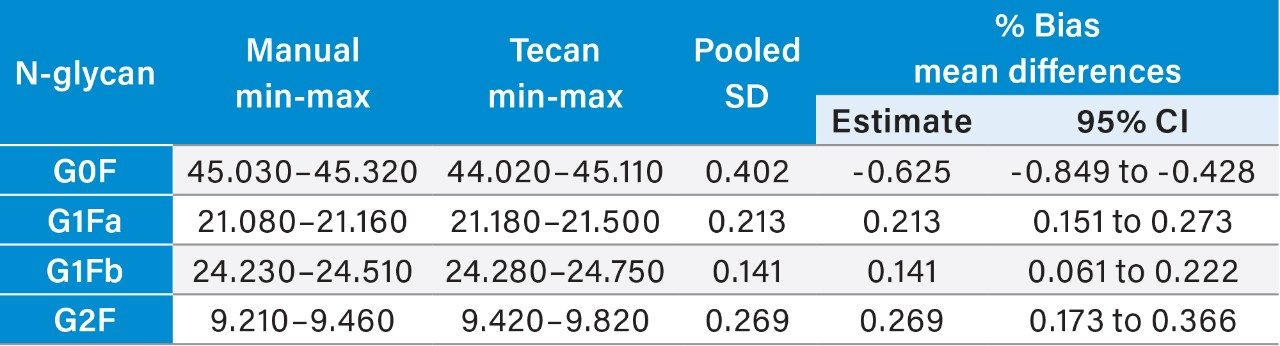
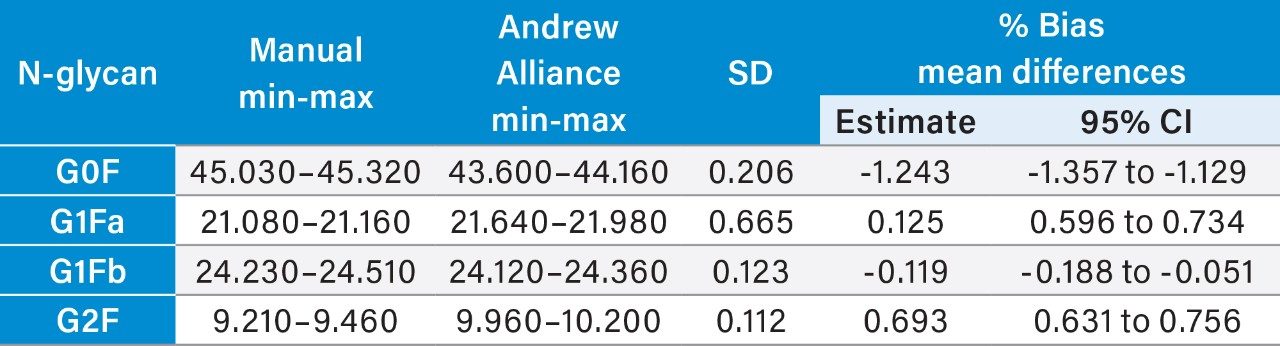
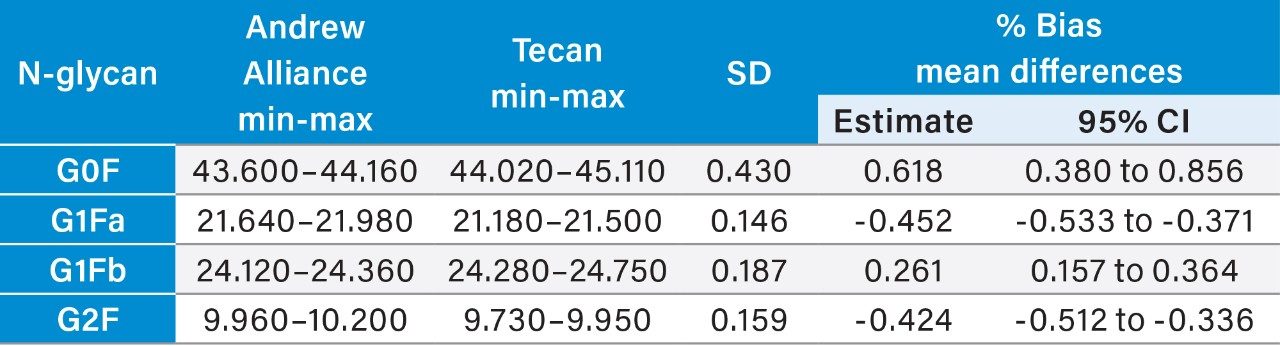
The semi-automated and fully automated sample preparation workflows developed by Waters for use with the GlycoWorks RapiFluor-MS N-Glycan Kit and GlycoWorks RapiFluor-MS N-Glycan Kit – Automation provide area percent N-glycan profile results without significant bias relative to the established manual GlycoWorks RapiFluor-MS N-glycan sample preparation workflow.
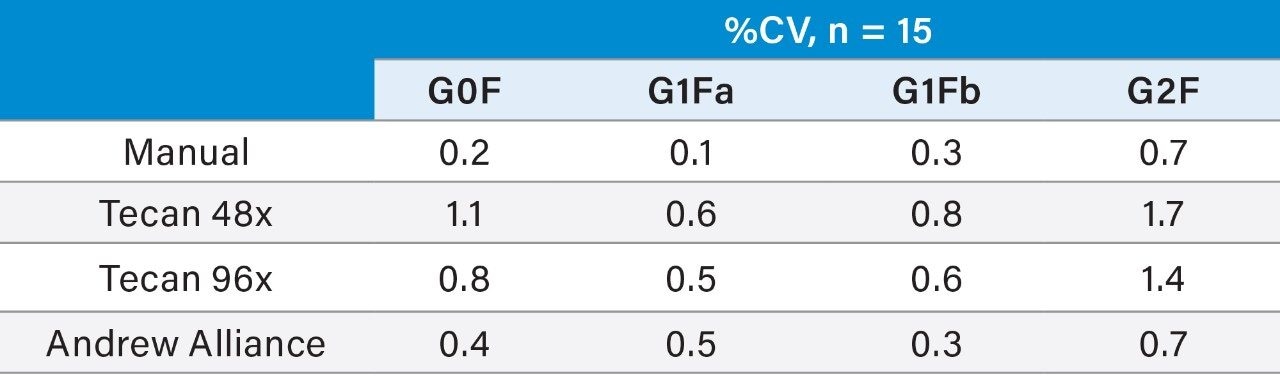
A high measurement precision of <2% CV is achievable for the semi-automated and fully automated methods (Table 1). Therefore, the GlycoWorks RapiFluor-MS N-Glycan Kit –Automation can be applied to highly efficient, automated, high-throughput N-glycan sample preparation without any significant loss inprecision performance. Further statistical analysis demonstrates expected measurement ranges (minimum-maximum) and measurement bias (Tables 2 and 3) for the semi-automated and fully automated methods.
This application note provides data to help biopharmaceutical scientists evaluate the analytical performance of each method.
Waters has introduced a range of validated products and automated N-glycan sample preparation workflows which enable a marked increase in efficiency and throughput flexibility for scientists. This application note demonstrates that the GlycoWorks RapiFluor-MS N-glycan sample preparation kit is readily scalable from manual, to semi-automated, and fully automated workflows.
The GlycoWorks RapiFluor-MS N-Glycan Kit – Automation is offered with a validated Tecan script for completely hands free preparation of N-glycan samples. The kit reagent quantities have been designed to be compatibl ewith the other major automated liquid handling manufacturers. The coupling of the highly efficient RapiFluor-MS label and associated chemistries ofthe GlycoWorks RapiFluor-MS N-Glycan Kit to the validated automation workflows and scripts provides an effective approach for laboratories wishing to leverage their automation resources.
The provision of these GlycoWorks automation resources completes a product family which offers maximum flexibility to suit the throughput requirements and automation resources of laboratories. Further resources for the manual workflow, semi-automated workflow on the Andrew Alliance, and the fully automated workflow on the Tecan can be found in corresponding application notes at http://wvmc.waters.com/glycans/.
720006322, June 2018